Advanced Pyrimidotriazole Synthesis: A Non-Corrosive Route for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic routes that balance chemical efficiency with operational safety and cost-effectiveness. A significant breakthrough in this domain is documented in patent CN112225761B, which details a novel synthesis method for Pyrimidotriazole, a critical scaffold in medicinal chemistry particularly relevant for treating hyperthyroidism. This patent introduces a transformative two-step process that fundamentally shifts the manufacturing paradigm from harsh, corrosive conditions to a mild, room-temperature protocol. By utilizing 4-(O-(2,4,6-triisopropyl)benzenesulfonyl)-uridine as a key starting material and employing formic acid for the final cyclization, this method eliminates the need for aggressive chlorinating agents like phosphorus oxychloride (POCl3) or phosphorus pentachloride (PCl5). For R&D Directors and Supply Chain Heads, this represents a pivotal opportunity to optimize production lines, reduce equipment degradation, and enhance the overall sustainability of the manufacturing process without compromising on the purity or yield of the final pharmaceutical intermediate.
The strategic value of this patent extends beyond mere chemical novelty; it addresses critical pain points in the supply chain of complex heterocyclic compounds. Traditional methods often impose severe constraints on facility infrastructure due to the generation of corrosive gases and the requirement for specialized containment systems. In contrast, the approach outlined in CN112225761B allows for synthesis under ambient conditions, drastically lowering the barrier to entry for scale-up. This means that existing general-purpose chemical reactors can be utilized without the need for expensive, corrosion-resistant linings or extensive gas scrubbing systems. For a reliable pharmaceutical intermediate supplier, adopting such a methodology translates directly into improved asset longevity and reduced downtime, ensuring a more consistent and reliable supply of high-purity materials to downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the triazole-pyrimidine fused ring system has relied on chemically aggressive pathways that pose significant operational challenges. As illustrated in the prior art, conventional routes frequently involve the chlorination of uracil derivatives using POCl3 or PCl5, followed by reaction with hydrazine and subsequent ring closure with trimethyl orthoformate. 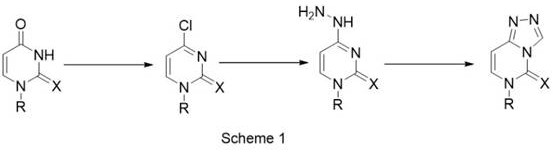 These legacy processes are fraught with inefficiencies; the use of POCl3 generates substantial quantities of hydrogen chloride gas, necessitating robust ventilation and neutralization systems to protect personnel and infrastructure. Furthermore, the requirement for reflux conditions in certain steps introduces thermal stress and potential safety hazards, including the risk of explosion when handling hydrazine hydrate at elevated temperatures. The reliance on trifluoroacetic acid (TFA) as a catalyst or solvent in alternative methods further exacerbates equipment corrosion, leading to frequent maintenance cycles and the potential for metal contamination in the final product. These factors collectively drive up the cost of goods sold (COGS) and complicate the regulatory compliance landscape for manufacturers.
These legacy processes are fraught with inefficiencies; the use of POCl3 generates substantial quantities of hydrogen chloride gas, necessitating robust ventilation and neutralization systems to protect personnel and infrastructure. Furthermore, the requirement for reflux conditions in certain steps introduces thermal stress and potential safety hazards, including the risk of explosion when handling hydrazine hydrate at elevated temperatures. The reliance on trifluoroacetic acid (TFA) as a catalyst or solvent in alternative methods further exacerbates equipment corrosion, leading to frequent maintenance cycles and the potential for metal contamination in the final product. These factors collectively drive up the cost of goods sold (COGS) and complicate the regulatory compliance landscape for manufacturers.
The Novel Approach
The methodology presented in CN112225761B offers a decisive break from these traditional constraints by leveraging a mild, nucleophilic substitution strategy followed by an acid-catalyzed cyclization that avoids corrosive byproducts. Instead of forcing the reaction with harsh chlorinating agents, this novel route utilizes a bulky 2,4,6-triisopropylbenzenesulfonyl leaving group, which facilitates the displacement by N-tert-butyloxycarbonyl hydrazine under gentle conditions. The subsequent cyclization is achieved using aqueous formic acid at room temperature, a reagent that is significantly less corrosive than TFA or mineral acids and does not evolve hazardous gases. This shift not only simplifies the work-up procedure but also preserves the integrity of the reaction vessel, allowing for longer campaign runs without the fear of equipment failure. For procurement managers, this translates to cost reduction in pharmaceutical intermediate manufacturing through lower maintenance overheads and reduced waste disposal costs associated with neutralizing corrosive effluents.
Mechanistic Insights into Formic Acid-Mediated Cyclization
At the heart of this synthesis is a sophisticated understanding of nucleophilic aromatic substitution and intramolecular cyclization dynamics. The first step involves the activation of the uridine scaffold through the installation of the triisopropylbenzenesulfonyl group, which serves as an excellent leaving group due to its steric bulk and electron-withdrawing properties. When reacted with N-tert-butyloxycarbonyl hydrazine, the hydrazine nitrogen attacks the C4 position of the pyrimidine ring, displacing the sulfonate ester to form the key hydrazino-intermediate. This step is critical as it sets the stage for the ring closure without requiring high thermal energy. The use of acetonitrile as a solvent ensures optimal solubility of the organic substrates while maintaining a polar environment conducive to the substitution reaction. The mildness of this step is paramount for preserving sensitive functional groups on the ribose sugar moiety, ensuring that the final product retains the necessary stereochemistry and structural integrity required for biological activity.
The second step, the cyclization, is where the true elegance of the process shines. By dissolving the hydrazino-intermediate in aqueous formic acid, the reaction proceeds through a mechanism where the formic acid acts as both a solvent and a catalyst. The acidic environment protonates the hydrazine nitrogen, increasing its nucleophilicity towards the adjacent carbonyl or imine carbon, facilitating the closure of the triazole ring. 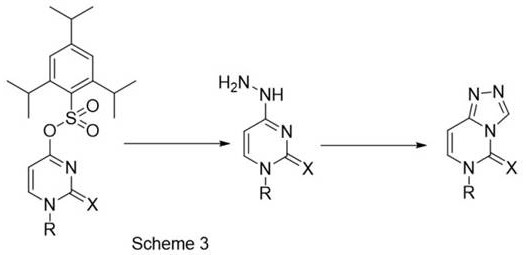 Crucially, this cyclization occurs at room temperature over a period of 8-12 hours, eliminating the need for thermal input that could degrade the product or generate impurities. The aqueous nature of the formic acid solution also aids in the solubility of polar intermediates and simplifies the subsequent isolation process. From an impurity control perspective, the absence of harsh reagents means fewer side reactions, such as over-chlorination or degradation of the sugar backbone, resulting in a cleaner crude product that requires less intensive purification. This mechanistic efficiency is a key driver for achieving the high-purity pharmaceutical intermediates demanded by regulatory bodies.
Crucially, this cyclization occurs at room temperature over a period of 8-12 hours, eliminating the need for thermal input that could degrade the product or generate impurities. The aqueous nature of the formic acid solution also aids in the solubility of polar intermediates and simplifies the subsequent isolation process. From an impurity control perspective, the absence of harsh reagents means fewer side reactions, such as over-chlorination or degradation of the sugar backbone, resulting in a cleaner crude product that requires less intensive purification. This mechanistic efficiency is a key driver for achieving the high-purity pharmaceutical intermediates demanded by regulatory bodies.
How to Synthesize Pyrimidotriazole Efficiently
Implementing this synthesis route in a production environment requires careful attention to the stoichiometry and mixing parameters to ensure consistent quality. The process begins with the preparation of the activated uridine derivative, followed by the controlled addition of the hydrazine reagent to manage exotherms, although the reaction is largely thermally neutral. The subsequent cyclization step is operationally simple, requiring only the preparation of the formic acid solution and adequate stirring time to ensure complete conversion. Detailed standard operating procedures (SOPs) would specify the exact grades of reagents and the parameters for the column chromatography separation to maximize yield. The simplicity of these steps makes the technology highly transferable between different manufacturing sites, reducing the risk associated with technology transfer projects.
- React 4-(O-(2,4,6-triisopropyl)benzenesulfonyl)-uridine with N-tert-butyloxycarbonyl hydrazine in acetonitrile at room temperature to form the hydrazine intermediate.
- Dissolve the intermediate in an aqueous formic acid solution and stir at room temperature for 8-12 hours to effect cyclization.
- Concentrate the reaction mixture and purify the final Pyrimidotriazole product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this patent-protected methodology offers substantial strategic benefits. The elimination of corrosive reagents like POCl3 and TFA directly impacts the total cost of ownership for manufacturing assets. Equipment used in traditional processes suffers from accelerated wear and tear, leading to frequent replacements and unplanned shutdowns. By switching to this mild formic acid-based route, manufacturers can significantly extend the lifecycle of their reactors and piping systems. This reduction in capital expenditure on maintenance and replacement parts contributes to a more stable cost structure. Furthermore, the safety profile of the process is markedly improved; the absence of hydrogen chloride gas evolution removes the need for complex gas scrubbing infrastructure, lowering both initial setup costs and ongoing operational expenses related to environmental compliance and waste treatment.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive and hazardous reagents. Traditional methods require stoichiometric amounts of chlorinating agents and large volumes of corrosive solvents, all of which incur high procurement and disposal costs. By utilizing formic acid and avoiding heavy metal catalysts or aggressive chlorination, the material cost per kilogram of product is inherently lower. Additionally, the simplified work-up procedure reduces the consumption of purification materials and solvents. The qualitative reduction in hazardous waste generation also lowers the fees associated with environmental disposal, creating a leaner and more economically efficient production model that enhances margin potential without compromising quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is often threatened by the complexity of sourcing specialized, hazardous chemicals and the downtime associated with equipment maintenance. This synthesis route relies on commodity chemicals like formic acid and acetonitrile, which are readily available from multiple global suppliers, reducing the risk of raw material shortages. The mild reaction conditions also mean that production is less susceptible to disruptions caused by equipment failure or safety incidents. A manufacturing process that does not require specialized corrosion-resistant alloys allows for greater flexibility in choosing contract manufacturing organizations (CMOs), as more facilities are equipped to handle general organic synthesis. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates by widening the pool of qualified manufacturing partners.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this method mitigates those risks through its inherent mildness. The room temperature operation eliminates the energy costs associated with heating and cooling large reactors, contributing to a lower carbon footprint. The absence of corrosive gas emissions simplifies the permitting process for new production lines and ensures ongoing compliance with increasingly stringent environmental regulations. The process generates less hazardous waste, aligning with green chemistry principles and corporate sustainability goals. This environmental compatibility makes the process future-proof against regulatory tightening, ensuring long-term viability for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D and procurement teams evaluating the feasibility of adopting this route for their specific supply chains. The answers are derived directly from the technical disclosures and beneficial effects outlined in the patent documentation, providing a factual basis for decision-making. These insights help clarify the operational advantages and technical robustness of the method.
Q: What are the primary equipment advantages of this Pyrimidotriazole synthesis method?
A: Unlike conventional methods requiring POCl3 or PCl5 which release corrosive hydrogen chloride gas, this process operates at room temperature with formic acid, significantly reducing equipment corrosion and maintenance requirements.
Q: How does this method improve safety compared to prior art?
A: The elimination of reflux conditions and corrosive gases removes explosion risks and hazardous vapor exposure, creating a safer operational environment for chemical manufacturing teams.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the mild reaction conditions and lack of specialized high-pressure or corrosion-resistant equipment requirements make this route highly scalable for industrial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidotriazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the potential of the CN112225761B process and is prepared to leverage this technology to deliver superior value to our partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for API intermediates, guaranteeing that every batch meets the highest international standards. We are committed to translating the theoretical benefits of this mild synthesis route into tangible supply chain advantages for our clients.
We invite R&D Directors and Procurement Managers to explore how this optimized synthesis can enhance your product portfolio. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this non-corrosive method for your projects. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to build a more efficient, safe, and cost-effective supply chain for your critical pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
