Revolutionizing Pomalidomide Intermediate Production: A Green, High-Yield Synthesis of 3-Aminopiperidine-2,6-Dione Hydrochloride
Introduction: Strategic Innovation in Oncology Intermediate Synthesis
The pharmaceutical landscape for multiple myeloma treatment has been significantly shaped by the approval of Pomalidomide, a potent immunomodulatory imide drug. As demand for this critical oncology therapy grows, the efficiency of its supply chain becomes paramount, particularly regarding the synthesis of its key precursor, 3-aminopiperidine-2,6-dione hydrochloride. Patent CN111675648B introduces a transformative synthetic methodology that fundamentally alters the production economics and environmental footprint of this vital intermediate. By shifting the starting material from costly glutamine to the abundant and inexpensive L-glutamic acid, this invention addresses long-standing bottlenecks in pharmaceutical manufacturing. The disclosed process not only streamlines the reaction sequence by removing complex protection and deprotection steps but also achieves exceptional purity levels exceeding 98.7 percent, ensuring a robust foundation for downstream API synthesis. This technical breakthrough offers a compelling value proposition for global pharmaceutical manufacturers seeking to optimize their procurement strategies and enhance supply chain resilience.
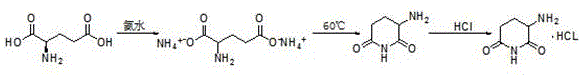
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-aminopiperidine-2,6-dione hydrochloride has relied heavily on L-glutamine as the primary starting material. This conventional pathway, while chemically feasible, is fraught with significant operational and economic inefficiencies that burden modern supply chains. The traditional route necessitates a multi-step sequence involving the protection of the amino group using tert-butyloxycarbonyl (Boc) anhydride, followed by cyclization and a subsequent deprotection step utilizing trifluoroacetic acid (TFA). The reliance on TFA is particularly problematic; it is a corrosive, toxic substance that poses severe safety risks to personnel and requires specialized, corrosion-resistant equipment, thereby inflating capital expenditure. Furthermore, the generation of fluorinated waste streams creates substantial environmental compliance challenges and disposal costs. The complexity of managing protecting groups inherently increases the number of unit operations, leading to lower overall yields and higher production costs due to the consumption of expensive reagents and solvents.
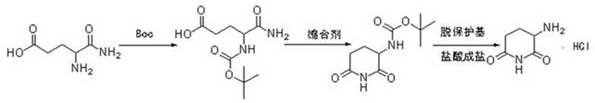
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach detailed in the patent leverages the intrinsic reactivity of L-glutamic acid to achieve direct cyclization. This innovative strategy bypasses the need for any amino protection entirely, collapsing what was once a multi-stage process into a concise, efficient sequence. The reaction initiates with the formation of a diammonium salt in aqueous media, followed by a controlled thermal treatment that induces intramolecular cyclization with the elimination of ammonia. This elegant simplification not only drastically reduces the number of processing steps but also eliminates the usage of hazardous organic solvents and toxic deprotecting agents during the core synthesis phase. The result is a cleaner reaction profile with fewer by-products, facilitating easier purification and yielding a product of superior quality. By utilizing water as the primary reaction medium for the cyclization step, the process aligns perfectly with green chemistry principles, offering a sustainable alternative that minimizes the ecological impact of pharmaceutical manufacturing.
Mechanistic Insights into Thermal Cyclization and Salt Formation
The core of this synthetic advancement lies in the precise manipulation of the L-glutamic acid diammonium salt under thermal conditions. Upon reacting L-glutamic acid with ammonia water at controlled temperatures between 15°C and 30°C, the carboxylic acid groups are neutralized to form the soluble diammonium species. This intermediate is stable under ambient conditions but becomes highly reactive upon heating. When the temperature is elevated to the range of 60°C to 80°C, the molecule undergoes a thermodynamic driving force towards cyclization. The mechanism involves the nucleophilic attack of the alpha-amino group on one of the activated carbonyl carbons of the side chain, facilitated by the loss of a water molecule and the elimination of ammonia gas. This intramolecular condensation effectively closes the piperidine ring, forming the 3-aminopiperidine-2,6-dione scaffold. The simplicity of this mechanism is its greatest strength; it relies on fundamental thermodynamic stability rather than kinetic control via protecting groups, which inherently reduces the formation of complex impurities often associated with incomplete protection or deprotection cycles.
Following the cyclization, the isolation of the product is achieved through a highly efficient salting-out procedure. The crude 3-aminopiperidine-2,6-dione, which precipitates from the aqueous reaction mixture, is redissolved in ethanol to ensure homogeneity before the introduction of hydrochloric acid gas. The use of gaseous HCl allows for precise control over the pH and stoichiometry of the salt formation, preventing local over-acidification that could lead to degradation. Maintaining the system temperature below 15°C during this exothermic gas absorption is critical to maximizing crystal purity and yield. The resulting hydrochloride salt exhibits low solubility in the ethanol medium, prompting immediate crystallization. This crystallization step acts as a powerful purification tool, excluding non-ionic impurities and residual starting materials from the crystal lattice. The final drying at 70°C ensures the removal of residual solvents without compromising the thermal stability of the aminopiperidine ring, delivering a free-flowing powder with consistent particle characteristics suitable for subsequent coupling reactions in Pomalidomide synthesis.
How to Synthesize 3-Aminopiperidine-2,6-Dione Hydrochloride Efficiently
The implementation of this synthesis protocol requires careful attention to temperature gradients and reagent addition rates to maximize the yield, which has been demonstrated to reach upwards of 82 percent in pilot examples. The process begins with the dispersion of L-glutamic acid in water, followed by the dropwise addition of ammonia water to form the diammonium salt precursor. Subsequent heating triggers the cyclization event, after which the solid intermediate is filtered and subjected to salt formation in ethanol. This standardized approach minimizes variability and ensures reproducible high-purity outcomes essential for GMP manufacturing environments. For detailed operational parameters, stoichiometric ratios, and specific workup procedures, please refer to the comprehensive guide below.
- React L-glutamic acid with ammonia water at 15-30°C to form the diammonium salt.
- Heat the mixture to 60-80°C to induce cyclization and eliminate ammonia, precipitating the diketone.
- Dissolve the crude product in ethanol and introduce hydrochloric acid gas below 15°C to form the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-glutamic acid-based route represents a significant opportunity to optimize cost structures and mitigate supply risks. The shift away from protected amino acids and hazardous reagents translates directly into tangible economic benefits and operational flexibility. By simplifying the chemical architecture of the synthesis, manufacturers can reduce their dependency on volatile specialty chemical markets and streamline their logistics.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the substitution of raw materials. L-glutamic acid is a commodity fermentation product available in massive quantities at a fraction of the cost of L-glutamine or protected derivatives. Furthermore, the complete elimination of Boc-anhydride and trifluoroacetic acid removes two of the most expensive line items from the bill of materials. The reduction in solvent usage, particularly the shift towards water and ethanol rather than chlorinated or aromatic solvents, further drives down utility and waste disposal costs. This leaner material profile ensures a substantially lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations for the final API.
- Enhanced Supply Chain Reliability: Relying on commodity-grade starting materials significantly de-risks the supply chain. L-glutamic acid is produced globally by numerous suppliers for the food and feed industries, ensuring a robust and diversified supply base that is less susceptible to the shortages often seen with fine chemical intermediates. Additionally, the simplified process flow reduces the number of critical path steps, shortening the overall manufacturing cycle time. This agility allows for faster response to market demand fluctuations and reduces the inventory holding costs associated with work-in-progress materials, thereby enhancing the overall resilience of the pharmaceutical supply network.
- Scalability and Environmental Compliance: From a facility perspective, the absence of corrosive TFA and toxic organic solvents simplifies the engineering requirements for production vessels and waste treatment systems. This 'green' profile facilitates easier regulatory approvals and reduces the burden of environmental monitoring and reporting. The process is inherently safer, lowering insurance premiums and operational risks associated with handling hazardous chemicals. The high purity of the crude product prior to final crystallization also implies that downstream purification loads are lighter, allowing existing equipment to process larger batches more efficiently, thus supporting seamless commercial scale-up from kilogram to multi-ton production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical implications of adopting this technology for large-scale manufacturing.
Q: Why is the L-glutamic acid route superior to the traditional glutamine route?
A: The L-glutamic acid route eliminates the need for expensive Boc protection and toxic trifluoroacetic acid (TFA) deprotection steps, significantly reducing raw material costs and environmental hazards while simplifying the process to a single pot cyclization.
Q: What represents the critical quality attribute in this synthesis?
A: The critical quality attribute is the purity of the final hydrochloride salt, which consistently achieves over 98.7% purity through controlled crystallization and the avoidance of complex side reactions associated with protecting group chemistry.
Q: Is this process scalable for commercial API production?
A: Yes, the process utilizes commodity chemicals like L-glutamic acid and ammonia water, operates under mild thermal conditions (60-80°C), and avoids hazardous reagents, making it highly suitable for multi-ton commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopiperidine-2,6-Dione Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the global oncology market. Our technical team has thoroughly analyzed the potential of the L-glutamic acid cyclization method and is fully prepared to support partners in integrating this technology into their supply chains. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, GMP-compliant manufacturing. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 3-aminopiperidine-2,6-dione hydrochloride meets the exacting standards required for Pomalidomide synthesis, minimizing the risk of downstream failures.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these cost-saving innovations. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and security for your critical drug pipelines.
