Industrial Scale-Up Of Chiral Diphenyl Prolinol Via Novel One-Pot Sulfuric Acid Catalysis
Introduction To The Breakthrough In Chiral Catalyst Manufacturing
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral building blocks that enable the production of complex pharmaceutical agents. A pivotal development in this field is detailed in patent CN109265385B, which outlines a revolutionary low-cost and high-efficiency synthesis process for chiral diphenyl prolinol and its hydrochloride salt. This compound serves as a critical precursor for the renowned Corey-Bakshi-Shibata (CBS) catalysts, which are indispensable for the asymmetric reduction of ketones and other carbonyl compounds in modern drug discovery. The patent introduces a novel methodology that replaces hazardous reagents like phosgene with environmentally benign concentrated sulfuric acid, facilitating a streamlined one-pot operation that achieves total yields exceeding 80% and optical purities greater than 99.5%. This technological leap addresses long-standing industrial pain points regarding safety, scalability, and cost-effectiveness.
For R&D directors and procurement specialists seeking a reliable chiral catalyst supplier, understanding the mechanistic underpinnings and commercial viability of this new route is essential. The ability to produce such high-value intermediates with minimal environmental impact and superior consistency positions this technology as a benchmark for future manufacturing standards in the fine chemical sector.
The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
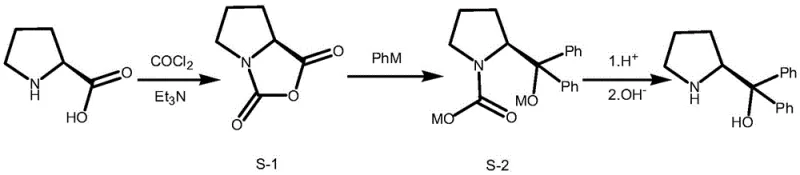
Historically, the synthesis of chiral diphenyl prolinol has been plagued by significant safety hazards and operational inefficiencies. Early methodologies, such as the route reported by Mathre et al. in 1991, relied heavily on the use of phosgene or triphosgene to introduce protecting groups. As illustrated in the reaction schemes of prior art, these processes involve the formation of unstable anhydride intermediates (Compound S-1) that polymerize rapidly even at low temperatures, releasing carbon dioxide and posing severe risks for industrial scale-up. Furthermore, the reliance on toxic reagents necessitates expensive containment systems and rigorous waste treatment protocols, drastically inflating the cost reduction in pharmaceutical intermediates manufacturing. Other approaches, such as those utilizing ethyl chloroformate or trimethylchlorosilane, suffer from similar drawbacks, including the use of irritating reagents, harsh low-temperature conditions for Grignard reactions (often below -10°C), and complex multi-step purification procedures that erode overall yield.
The Novel Approach
In stark contrast, the process disclosed in CN109265385B offers a paradigm shift by utilizing concentrated sulfuric acid as a catalyst for the initial esterification step. This modification not only eliminates the need for dangerous acyl chlorides but also simplifies the reaction matrix. The novel approach enables the direct conversion of chiral proline into proline methyl ester sulfate, which is then subjected to Boc protection without isolation. Crucially, the subsequent Grignard reaction with phenylmagnesium chloride can be conducted at a much milder temperature range of 10-60°C, avoiding the energy-intensive cryogenic conditions required by legacy methods. This robustness allows for the commercial scale-up of complex chiral intermediates with unprecedented ease, ensuring that the supply chain remains resilient against technical failures and raw material volatility.
Mechanistic Insights Into Sulfuric Acid Catalyzed One-Pot Synthesis
The core innovation of this patented process lies in its elegant integration of esterification, protection, and nucleophilic addition into a cohesive sequence that minimizes intermediate handling. The mechanism begins with the protonation of the carboxylic acid group of chiral proline by concentrated sulfuric acid in methanol, driving the equilibrium towards the formation of the methyl ester sulfate salt. Unlike traditional methods that require neutralization and extraction before protection, this crude sulfate salt is directly treated with an inorganic base, such as sodium carbonate or potassium carbonate, to adjust the pH. This in situ neutralization generates the free amine necessary for the immediate reaction with di-tert-butyl dicarbonate ((Boc)2O). The inventors discovered that residual inorganic salts from this neutralization step do not interfere with the subsequent Boc protection or the Grignard addition, a finding that fundamentally alters the process economics by removing filtration and drying steps.
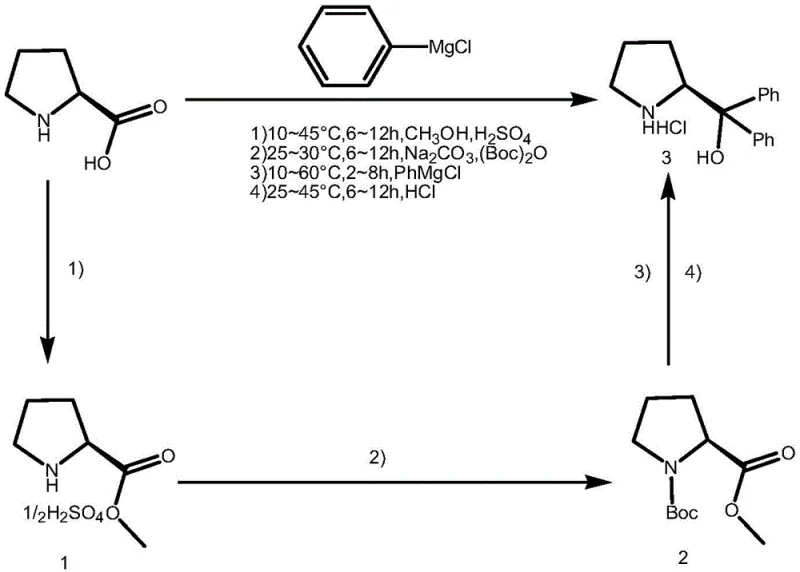
Following Boc protection, the reaction mixture is simply distilled to remove solvents, and the crude residue is redissolved in tetrahydrofuran for the formatting reaction. The addition of phenylmagnesium chloride proceeds smoothly at ambient to moderately elevated temperatures (10-60°C), attacking the ester carbonyl to form the tertiary alcohol structure characteristic of diphenyl prolinol. The final step involves deprotection using concentrated hydrochloric acid, which cleaves the Boc group and simultaneously forms the stable hydrochloride salt of the product. Due to the low solubility of the hydrochloride salt in the THF/water mixture at low temperatures (-5°C), the product precipitates out and can be isolated via centrifugation. This crystallization-driven purification ensures high optical purity (≥99.5% ee) and chemical content (>99.0%) without the need for chromatography, effectively controlling impurity profiles and guaranteeing batch-to-batch consistency.
How To Synthesize Chiral Diphenyl Prolinol Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and safety, yet it is designed to be operationally straightforward for industrial reactors. The process leverages the stability of the intermediates to allow for a continuous workflow within a single vessel, reducing the footprint and equipment requirements for production facilities. Operators must monitor the exothermic nature of the base addition and Grignard reaction, although the wider temperature window compared to prior art provides a significant safety margin. For detailed technical specifications and standard operating procedures regarding reagent grades and mixing rates, please refer to the structured guide below.
- Esterify chiral proline with methanol using concentrated sulfuric acid at 10-45°C to form proline methyl ester sulfate.
- Adjust pH with inorganic base and protect the amino group with di-tert-butyl dicarbonate (Boc2O) without purifying the intermediate.
- React the crude Boc-protected ester with phenylmagnesium chloride at 10-60°C, followed by deprotection with HCl to isolate the hydrochloride salt.
Commercial Advantages For Procurement And Supply Chain Teams
From a strategic sourcing perspective, the adoption of this sulfuric acid-catalyzed process offers transformative benefits for procurement managers and supply chain heads tasked with securing high-purity chiral intermediates. The elimination of phosgene and other hazardous gases removes a major regulatory burden and liability risk, simplifying the permitting process for manufacturing sites and ensuring uninterrupted production schedules. Moreover, the one-pot nature of the synthesis drastically reduces the consumption of solvents and auxiliary materials, which are often significant cost drivers in fine chemical production. By minimizing the number of unit operations—specifically removing intermediate isolation and purification steps—the process enhances labor efficiency and throughput, allowing suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction In Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive and toxic reagents like phosgene and triphosgene. Replacing these with commodity chemicals like sulfuric acid and sodium carbonate significantly lowers the raw material bill of goods. Additionally, the ability to skip purification of the Boc-protected intermediate reduces solvent usage and waste disposal costs, while the higher overall yield (>80%) means less starting material is required per kilogram of final product, creating a compounding effect on cost savings throughout the value chain.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the complexity of multi-step syntheses. This new route relies on widely available, commodity-grade raw materials such as L-proline or D-proline, methanol, and phenylmagnesium chloride, which are sourced from a robust global market. The simplified process flow reduces the likelihood of bottlenecks at intermediate stages, ensuring that lead times for high-purity chiral catalysts can be consistently met even during periods of high demand, thereby stabilizing the supply chain for downstream API manufacturers.
- Scalability And Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, particularly for exothermic reactions. The milder reaction conditions (10-60°C) of this Grignard addition make it inherently safer and easier to scale from pilot plants to multi-ton commercial reactors compared to cryogenic alternatives. Furthermore, the avoidance of heavy metals and persistent organic pollutants aligns with increasingly stringent global environmental regulations, facilitating smoother audits and certifications for green chemistry initiatives, which is a critical factor for long-term partnerships with top-tier pharmaceutical companies.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis technology, we have compiled answers to common inquiries based on the patent data. These insights address concerns regarding purity, safety, and process robustness, providing a clear picture of why this method represents a superior choice for industrial applications. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader manufacturing strategies.
Q: What are the key advantages of the sulfuric acid catalyzed route over phosgene methods?
A: The sulfuric acid route eliminates the use of highly toxic phosgene or triphosgene, significantly improving operational safety and reducing environmental compliance costs associated with hazardous gas handling.
Q: How does this process achieve cost reduction in manufacturing?
A: By employing a one-pot strategy that skips purification of the Boc-protected intermediate, the process reduces solvent consumption, labor hours, and material loss, leading to substantial overall cost savings.
Q: What optical purity can be expected from this synthesis method?
A: The patented process consistently delivers chiral diphenyl prolinol with an optical purity (ee value) of not less than 99.5%, meeting the stringent requirements for asymmetric catalysis applications.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable Chiral Diphenyl Prolinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN109265385B can be fully realized in a GMP-compliant environment. We are committed to delivering chiral diphenyl prolinol with stringent purity specifications, leveraging our rigorous QC labs to verify optical purity and chemical content for every batch, thus guaranteeing the performance of your downstream asymmetric synthesis reactions.
We invite you to collaborate with us to optimize your supply chain for chiral catalysts. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and production goals efficiently and sustainably.
