Advanced Gold-Catalyzed Synthesis of 4-Fluoro Pyrazole Derivatives for Commercial Scale-Up
Advanced Gold-Catalyzed Synthesis of 4-Fluoro Pyrazole Derivatives for Commercial Scale-Up
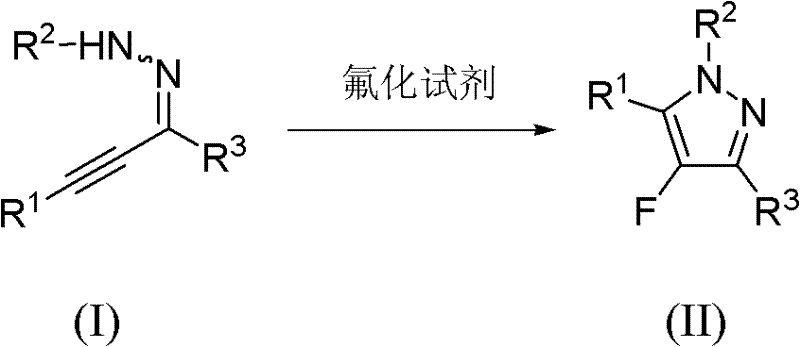
The pharmaceutical and agrochemical industries continuously demand efficient pathways to access fluorinated heterocycles, particularly 4-fluoro pyrazole derivatives, due to their profound impact on metabolic stability and bioactivity. Patent CN102285919A discloses a groundbreaking methodology that addresses the longstanding challenges in synthesizing these valuable scaffolds. By leveraging a homogeneous gold-catalyzed intermolecular amination and fluorination cascade, this technology enables the direct conversion of readily available alkynyl hydrazine substrates into high-purity 4-fluoro pyrazoles. The process operates under exceptionally mild conditions, typically at room temperature, utilizing Selectfluor as a robust electrophilic fluorinating source. This innovation represents a significant leap forward for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering complex fluorinated building blocks with superior efficiency and safety profiles compared to legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fluorinated pyrazole rings has been fraught with significant technical and economic hurdles that impede large-scale manufacturing. Traditional approaches often rely on the direct fluorination of pre-formed pyrazole rings using aggressive electrophilic fluorinating agents, which frequently suffer from poor regioselectivity and the formation of difficult-to-separate polyfluorinated byproducts. Alternative strategies involving the condensation of fluorinated diketones with hydrazines are limited by the scarcity and high cost of specific fluorinated carbonyl precursors. Furthermore, methods employing diazotization of amino-pyrazoles followed by thermal decomposition of tetrafluoroborate salts introduce severe safety risks due to the explosive nature of diazonium intermediates. These conventional pathways often necessitate harsh reaction conditions, multiple synthetic steps, and extensive purification protocols, resulting in low overall yields and substantial waste generation that negatively impacts the cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a streamlined, one-pot cyclization strategy driven by gold catalysis. This method bypasses the need for pre-functionalized fluorinated starting materials by introducing the fluorine atom directly during the ring-closing event. The reaction employs alkynyl hydrazines, which are easily synthesized from commercial alkynes, reacting with stable fluorinating reagents like N-fluorobenzenesulfonimide or Selectfluor. The use of a Lewis acid catalyst, specifically homogeneous gold complexes such as Ph3PAuNTf2, activates the alkyne moiety towards nucleophilic attack by the hydrazine nitrogen, followed by electrophilic fluorination. This tandem process occurs efficiently at temperatures ranging from 0°C to 100°C, with optimal results at room temperature, thereby eliminating the energy costs associated with high-temperature reactors and ensuring a much safer operational environment for plant personnel.
Mechanistic Insights into Ph3PAuNTf2-Catalyzed Cyclization
The core of this technological advancement lies in the unique ability of the cationic gold catalyst to activate the carbon-carbon triple bond of the alkynyl hydrazine substrate. Upon coordination with the gold center, the alkyne becomes highly electrophilic, facilitating an intramolecular or intermolecular nucleophilic attack by the adjacent hydrazine nitrogen atom. This initial amination step forms a vinyl-gold intermediate, which is then trapped by the electrophilic fluorine source present in the reaction mixture. The specific choice of Ph3PAuNTf2 as the catalyst is critical, as the non-coordinating NTf2 anion enhances the Lewis acidity of the gold center, promoting faster turnover rates and higher conversion efficiencies. This mechanistic pathway ensures excellent chemical and regioselectivity, directing the fluorine atom specifically to the 4-position of the pyrazole ring, a feat that is notoriously difficult to achieve with high precision using other transition metal catalysts or uncatalyzed thermal methods.
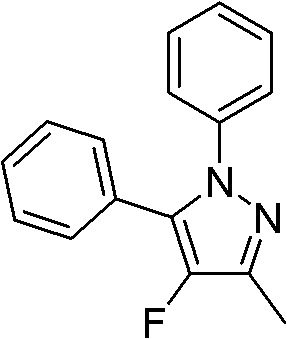
Furthermore, the reaction mechanism inherently suppresses the formation of common impurities associated with radical fluorination pathways. The controlled ionic nature of the gold-catalyzed cycle minimizes side reactions such as polymerization of the alkyne or over-fluorination of the aromatic rings. The presence of a mild inorganic base, such as sodium bicarbonate or sodium carbonate, plays a pivotal role in neutralizing the acid byproducts generated during the fluorination step, thereby maintaining the stability of the catalyst and preventing the decomposition of sensitive functional groups on the substrate. This delicate balance between catalyst activation, nucleophilic cyclization, and electrophilic quenching allows for the synthesis of complex molecules with diverse substituents, including halogens and electron-withdrawing groups, without compromising the integrity of the final product structure.
How to Synthesize 4-Fluoro Pyrazole Derivatives Efficiently
To implement this high-efficiency synthesis route in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and environmental controls to maximize yield and purity. The process begins with the preparation of the alkynyl hydrazine substrate, which can be sourced from existing literature or synthesized via standard coupling reactions. The reaction is conducted under an inert nitrogen atmosphere to prevent moisture-induced deactivation of the gold catalyst. A typical procedure involves dissolving the substrate in dry acetonitrile, followed by the sequential addition of the Ph3PAuNTf2 catalyst (2.0-3.0 mol%), the Selectfluor reagent (2.0 equivalents), and the base (2.0 equivalents). The mixture is stirred at ambient temperature for approximately 2 hours, with progress monitored by thin-layer chromatography. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance.
- Prepare the reaction mixture by combining the alkynyl hydrazine substrate, Ph3PAuNTf2 catalyst, Selectfluor fluorinating reagent, and a base such as sodium bicarbonate in acetonitrile solvent under nitrogen protection.
- Stir the mixture at room temperature (approximately 25°C) for about 2 hours, monitoring the reaction progress via TLC until the starting material is consumed.
- Upon completion, add silica gel to the mother liquor, remove the solvent under reduced pressure, and purify the crude product using column chromatography with a petroleum ether and ethyl acetate eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gold-catalyzed fluorination technology offers transformative benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials; since the method utilizes commercially available alkynes and stable fluorinating reagents, it removes the dependency on scarce, custom-synthesized fluorinated building blocks that often have long lead times and volatile pricing. This shift to commodity-grade starting materials significantly enhances supply chain reliability and reduces the risk of production stoppages due to raw material shortages. Additionally, the mild reaction conditions eliminate the need for specialized high-pressure or cryogenic equipment, allowing the process to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and hazardous reagents typically required for direct fluorination or diazotization. By operating at room temperature, the method achieves substantial energy savings compared to thermal cyclization processes that require prolonged heating. The high regioselectivity of the gold catalyst minimizes the formation of isomeric byproducts, which drastically reduces the complexity and cost of downstream purification steps such as chromatography or recrystallization. Furthermore, the catalyst loading is kept low (2.0-3.0 mol%), and the potential for catalyst recovery or the use of cheaper alternatives like copper or iron salts for less sensitive substrates provides additional avenues for optimizing the cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against varying substrate electronic properties ensures consistent production output regardless of batch-to-batch variations in starting material quality. The tolerance for halogens and heterocycles means that a single platform technology can be used to manufacture a wide library of derivatives, reducing the need for multiple dedicated production lines. This flexibility allows manufacturers to respond quickly to changing market demands for specific API intermediates without retooling. The use of stable, shelf-stable reagents like Selectfluor also simplifies inventory management and reduces the safety hazards associated with storing unstable diazonium salts or gaseous fluorine sources.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process generates minimal hazardous waste. The absence of heavy metal contaminants often associated with palladium-catalyzed cross-couplings simplifies the meeting of stringent residual metal specifications required for pharmaceutical ingredients. The reaction produces benign inorganic salts as byproducts, which are easier to treat in standard wastewater facilities. The simplicity of the workup procedure, involving basic silica gel treatment and solvent evaporation, facilitates easy scale-up from gram to ton quantities. This scalability ensures that the technology can support commercial production volumes ranging from 100 kgs to 100 MT annually, providing a secure supply base for long-term commercial agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this gold-catalyzed method over traditional fluorination routes?
A: Unlike traditional methods that often require harsh conditions, dangerous diazonium salts, or multi-step sequences, this patent-disclosed method operates under mild room temperature conditions with high regioselectivity. It utilizes commercially available alkynyl substrates and achieves high yields in a single pot, significantly simplifying the purification process and reducing safety risks associated with hazardous reagents.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly suitable for scale-up. The use of homogeneous gold catalysts at low loading (2.0-3.0 mol%), combined with inexpensive inorganic bases and common organic solvents like acetonitrile, makes the process economically viable. The mild reaction temperature eliminates the need for complex heating or cooling infrastructure, facilitating easier transition from laboratory to commercial manufacturing scales.
Q: What is the substrate scope regarding functional group tolerance?
A: The method demonstrates excellent substrate universality. It tolerates a wide range of substituents on the aromatic rings, including electron-donating groups like methoxy, electron-withdrawing groups like nitro, and halogens such as chlorine and bromine. Furthermore, heterocyclic substrates like thiophene are also compatible, making it a versatile platform for synthesizing diverse fluorinated pyrazole libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Fluoro Pyrazole Derivatives Supplier
As the global demand for fluorinated pharmaceutical intermediates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies backed by rigorous quality standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 4-fluoro pyrazole derivatives meets the highest international regulatory requirements. We understand the critical nature of these intermediates in the drug development pipeline and are committed to delivering materials that accelerate your timeline to market.
We invite you to engage with our technical procurement team to discuss how this advanced gold-catalyzed synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this more efficient manufacturing route. We encourage potential partners to contact us directly to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that demonstrate our capability to handle complex fluorination chemistries safely and effectively.
