Advanced Manufacturing of R-Biphenylalaninol: A Scalable Route for Sacubitril Intermediates
Introduction to Novel Sacubitril Intermediate Technology
The pharmaceutical landscape for cardiovascular treatments has been significantly reshaped by the introduction of LCZ696 (Entresto), a dual-acting angiotensin receptor-neprilysin inhibitor. Central to the manufacturing of this blockbuster drug is the critical chiral intermediate known as R-biphenylalaninol. Patent CN107540574B discloses a groundbreaking preparation method that addresses long-standing inefficiencies in the supply chain of this vital molecule. By utilizing a D-tyrosine derivative as the chiral starting material, the invention circumvents the need for complex asymmetric catalysis or difficult resolution processes typically associated with biphenyl structures. This approach not only streamlines the synthetic pathway but also ensures high optical purity from the outset, leveraging the inherent chirality of natural amino acids.
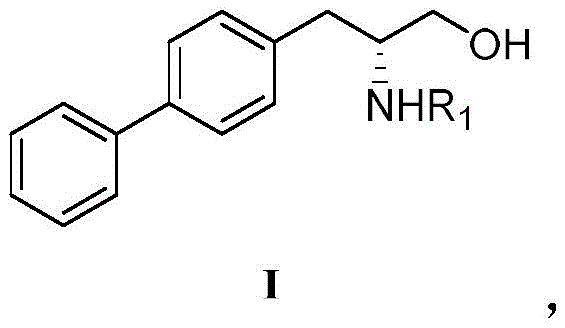
The structural complexity of R-biphenylalaninol, featuring a biphenyl system attached to a chiral amino alcohol backbone, presents unique challenges for large-scale synthesis. Traditional methods often struggle with the low reactivity of the phenyl group or the high cost of introducing chirality at a late stage. The disclosed technology offers a robust solution by first activating the phenolic hydroxyl group of the tyrosine derivative through sulfonylation. This activation creates a versatile intermediate capable of undergoing efficient cross-coupling reactions. For global procurement teams and R&D directors, this represents a shift towards more reliable and cost-stable sourcing strategies for heart failure medication components, ensuring consistent quality and supply continuity for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing R-biphenylalaninol have been plagued by significant economic and safety hurdles that hinder industrial scalability. One prominent prior art method, described in literature such as J. Med. Chem. (1995), relies on the activation of D-tyrosine using trifluoromethanesulfonic anhydride (triflic anhydride). While chemically effective, triflic anhydride is notoriously expensive, highly corrosive, and flammable, posing severe safety risks and requiring specialized, corrosion-resistant equipment that drives up capital expenditure. Furthermore, alternative strategies starting from biphenyl aldehyde often necessitate the use of precious metal catalysts like rhodium for asymmetric hydrogenation.
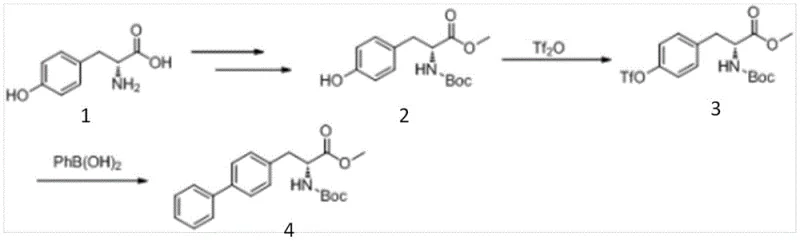
These rhodium-catalyzed routes, while capable of producing chiral intermediates, involve long reaction sequences including esterification, hydrogenation, reduction, and hydrogenolysis. The reliance on expensive phosphine ligands and noble metals drastically inflates the cost of goods sold (COGS). Additionally, methods involving enzymatic resolution or traditional chemical splitting of racemic mixtures suffer from theoretical yield limits of 50% and require rigorous control of reaction parameters like pH and substrate concentration. These factors collectively result in a fragile supply chain vulnerable to raw material price fluctuations and operational bottlenecks, making them less attractive for the high-volume production demands of modern cardiovascular therapeutics.
The Novel Approach
The innovative methodology presented in patent CN107540574B fundamentally reengineers the synthesis by adopting a "chiral pool" strategy combined with modern cross-coupling chemistry. Instead of building chirality onto a biphenyl scaffold, the process starts with readily available D-tyrosine derivatives, which already possess the required stereochemistry. The key breakthrough lies in the conversion of the phenolic hydroxyl group into a sulfonate ester (Compound III) using benign sulfonyl chlorides or anhydrides. This transformation activates the aromatic ring for subsequent palladium or nickel-catalyzed coupling with phenylboronic acid derivatives, effectively constructing the biphenyl motif under mild conditions.
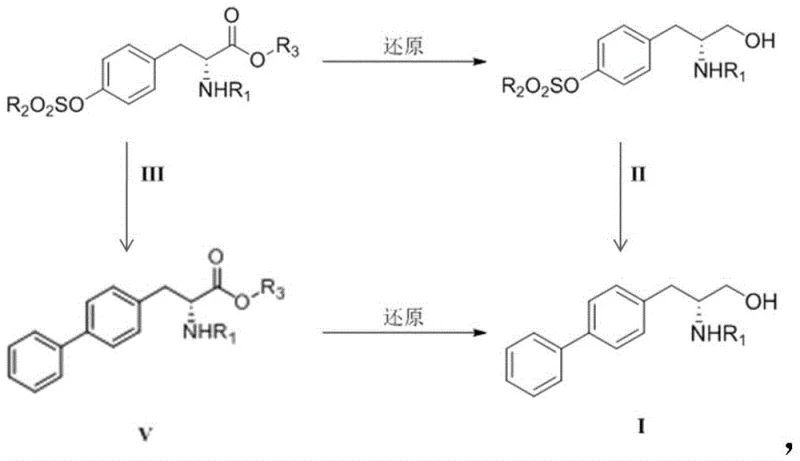
This novel approach offers two flexible synthetic routes: one where the sulfonate intermediate is reduced to an alcohol before coupling, and another where the coupling occurs prior to reduction. Both pathways eliminate the need for hazardous triflic anhydride and expensive rhodium catalysts. The use of earth-abundant nickel or standard palladium catalysts significantly lowers material costs. Moreover, the reaction conditions are optimized for industrial feasibility, operating at moderate temperatures between 30°C and 130°C with common solvents like morpholine or dioxane. This strategic pivot not only simplifies the operational workflow but also enhances the overall safety profile of the manufacturing process, making it highly suitable for multi-ton scale production.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
The core chemical transformation enabling this efficient synthesis is the Suzuki-Miyaura cross-coupling reaction, which links the activated tyrosine derivative with a phenyl boron species. The patent details the use of advanced palladium catalysts, specifically N-heterocyclic carbene (NHC) complexes such as [(IMes)Pd(Py)Cl2] or [(IPr)Pd(cin)Cl]. These catalysts are renowned for their high turnover numbers and stability, allowing the reaction to proceed efficiently even with sterically hindered substrates. The mechanism involves the oxidative addition of the palladium catalyst to the carbon-oxygen bond of the sulfonate intermediate (Compound III or II), followed by transmetallation with the phenylboronic acid and subsequent reductive elimination to form the carbon-carbon bond of the biphenyl system.
Crucially, the choice of ligand and base plays a pivotal role in maintaining the integrity of the chiral center during this transformation. The patent specifies the use of bases like potassium carbonate or potassium tert-butoxide in solvents such as morpholine or toluene to facilitate the activation of the boron species. By carefully controlling the reaction temperature between 110°C and 130°C and maintaining an inert argon atmosphere, the process minimizes side reactions such as homocoupling or protodeboronation. This precise control ensures that the stereochemical information from the D-tyrosine starting material is preserved throughout the coupling event, resulting in the final product with an enantiomeric excess (ee) value consistently exceeding 99%, which is critical for meeting stringent regulatory standards for pharmaceutical intermediates.
How to Synthesize R-Biphenylalaninol Efficiently
The synthesis of R-biphenylalaninol via this patented route is designed for operational simplicity and high yield, making it an ideal candidate for technology transfer and commercial scale-up. The process begins with the protection and sulfonylation of D-tyrosine, followed by a divergent strategy where the order of reduction and coupling can be selected based on specific facility capabilities. The standardized protocol utilizes common laboratory and plant equipment, avoiding the need for exotic high-pressure reactors often required for hydrogenation steps in alternative routes. Detailed experimental data in the patent demonstrates yields ranging from 46% to 74% for the coupling step and over 90% for the reduction step, indicating a robust and forgiving process window.
- React D-tyrosine derivative with sulfonyl chloride or anhydride to form the key sulfonate intermediate (Compound III).
- Perform a Suzuki-Miyaura coupling reaction using phenylboronic acid and a palladium catalyst to introduce the biphenyl moiety.
- Reduce the ester or carboxylic acid group using lithium aluminum hydride to yield the final R-biphenylalaninol alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic reduction of raw material costs associated with the elimination of triflic anhydride and rhodium catalysts. By substituting these high-value reagents with commodity chemicals like benzenesulfonyl chloride and palladium or nickel salts, the overall cost of manufacturing is significantly lowered. This cost efficiency is further amplified by the high atom economy of the coupling reaction and the ability to recycle solvents, contributing to a more sustainable and economically viable production model that can withstand market volatility.
- Cost Reduction in Manufacturing: The replacement of corrosive and expensive activating agents with stable sulfonyl chlorides directly reduces the expenditure on hazardous material handling and waste disposal. Furthermore, the avoidance of precious metal catalysts like rhodium removes a major variable cost driver, allowing for more predictable budgeting and pricing strategies for the final API. The simplified workup procedures, which often involve standard filtration and crystallization rather than complex chromatographic separations, also contribute to lower labor and utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Starting from D-tyrosine, a fermentation-derived amino acid available in bulk quantities from multiple global suppliers, de-risks the supply chain compared to relying on specialized chiral ligands or custom-synthesized biphenyl aldehydes. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram to multi-gram scales in the patent examples without loss of efficiency. The use of less toxic reagents and the generation of fewer hazardous byproducts align with green chemistry principles, simplifying environmental compliance and permitting processes. The ability to run reactions at atmospheric pressure and moderate temperatures reduces energy consumption and equipment stress, facilitating easier scale-up from pilot plant to commercial manufacturing suites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this R-biphenylalaninol synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN107540574B, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement teams assessing supplier capabilities. The clarity provided here aims to bridge the gap between academic patent language and practical industrial application requirements.
Q: What are the advantages of using D-tyrosine over biphenyl aldehyde for R-biphenylalaninol synthesis?
A: Using D-tyrosine leverages the chiral pool strategy, eliminating the need for expensive asymmetric hydrogenation or resolution steps required when starting from achiral biphenyl aldehydes. This significantly reduces catalyst costs and simplifies the purification process.
Q: Does this method avoid the use of hazardous triflic anhydride?
A: Yes, the patented method replaces highly corrosive and expensive trifluoromethanesulfonic anhydride with safer and more cost-effective benzenesulfonyl chlorides or anhydrides, improving operational safety and equipment longevity.
Q: What level of optical purity can be achieved with this synthetic route?
A: The process maintains the stereochemistry of the starting D-tyrosine material, consistently achieving optical purity values greater than 99% ee without the need for additional chiral resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Biphenylalaninol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving cardiovascular medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this patented route, including the safe handling of boronic acids and palladium catalysts. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of R-biphenylalaninol meets the highest international standards for optical purity and chemical identity.
We invite you to collaborate with us to leverage this advanced synthetic technology for your Sacubitril supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your overall manufacturing economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of this essential pharmaceutical intermediate.
