Scalable Synthesis of 5,8-Diamino-3,4-dihydro-2H-1-naphthalenone for ADC Manufacturing
The rapidly evolving landscape of oncology therapeutics has placed Antibody-Drug Conjugates (ADCs) at the forefront of pharmaceutical innovation, with topoisomerase I inhibitors serving as potent payloads for targeted cancer therapy. A critical bottleneck in the manufacturing of these life-saving drugs, such as the widely recognized Sacituzumab govitecan and Trastuzumab deruxtecan, lies in the efficient production of their core small-molecule warheads. Specifically, the synthesis of 5,8-diamino-3,4-dihydro-2H-1-naphthalenone, referred to herein as Compound I, represents a pivotal challenge due to the complexity of establishing the correct substitution pattern on the naphthalene ring. The recent disclosure in patent CN115703712A introduces a transformative synthetic methodology that circumvents traditional limitations, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize ADC payload production. This technological breakthrough not only addresses the structural intricacies of camptothecin derivatives but also aligns with the stringent quality and scalability demands of modern GMP manufacturing environments.
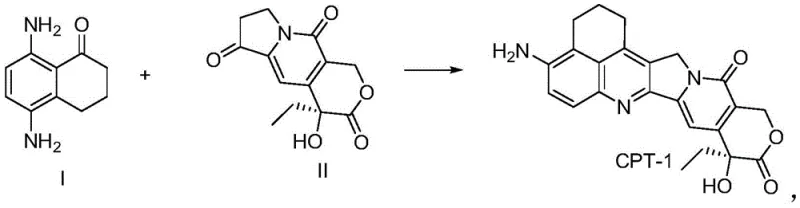
Historically, the preparation of Compound I has been plagued by inefficient synthetic strategies that rely heavily on electrophilic aromatic substitution reactions, particularly nitration. As illustrated in the prior art, conventional routes often involve the nitration of tetralone derivatives, a process inherently prone to generating regioisomers that are chemically similar and notoriously difficult to separate. For instance, existing literature describes pathways where the total reaction yield hovers between a mere 15.6% and 33.2%, primarily due to the loss of material during extensive purification steps such as multi-step column chromatography. These legacy methods, depicted in older synthetic schemes, necessitate harsh oxidative conditions and generate significant chemical waste, posing both environmental hazards and safety risks when attempting to scale up to metric ton quantities. The presence of isomeric impurities not only complicates the downstream synthesis of the final ADC payload but also threatens the critical purity specifications required for clinical-grade materials, making these traditional approaches economically and technically unsustainable for large-scale commercial operations.
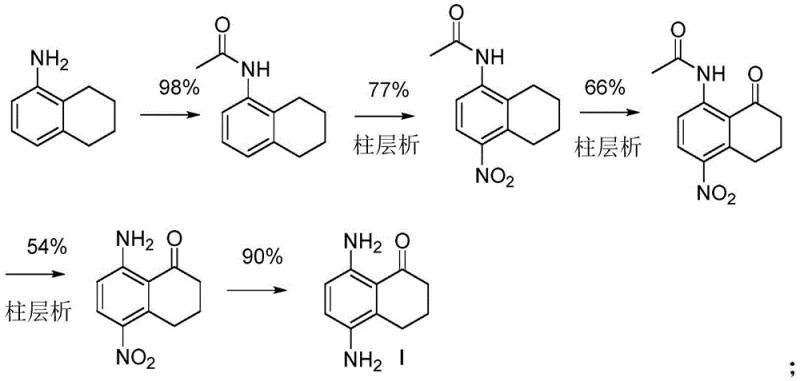
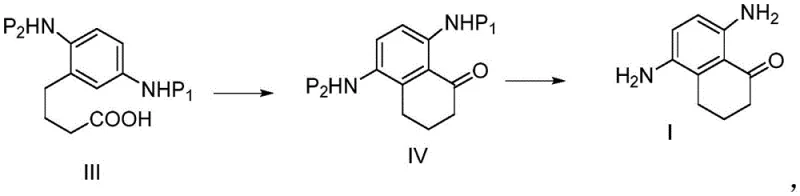
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent utilizes a strategic Friedel-Crafts cyclization of a pre-functionalized linear precursor, effectively bypassing the need for direct ring nitration. The core innovation involves the construction of the naphthalene skeleton from a 2,5-di-protected aminobenzenebutyric acid derivative, designated as Compound III in the patent documentation. By installing the amino protecting groups (such as acetyl groups) prior to the ring-closing step, the synthesis achieves exceptional regiocontrol, ensuring that the amino functionalities end up in the desired 5 and 8 positions without the formation of unwanted isomers. This methodological shift transforms the synthesis from a purification-heavy operation into a streamlined sequence of coupling and cyclization reactions. The avoidance of nitration not only simplifies the workup procedures but also eliminates the safety concerns associated with handling large quantities of nitrating agents and nitro-intermediates, thereby facilitating a smoother transition from laboratory benchtop to industrial reactor vessels.
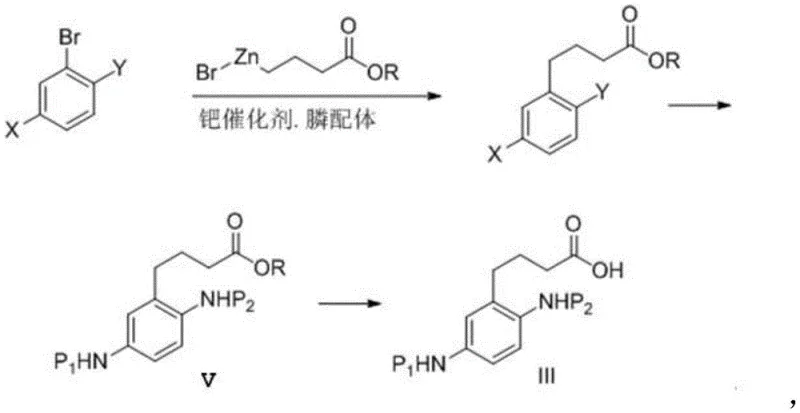
The mechanistic elegance of this new pathway is best exemplified by the palladium-catalyzed cross-coupling strategy employed to construct the carbon backbone of the precursor. The process initiates with the preparation of an organozinc reagent, specifically 4-ethoxy-4-oxobutyl zinc bromide, which serves as a nucleophilic partner in the subsequent coupling reaction. When reacted with a protected bromoaniline derivative in the presence of a palladium catalyst system—typically comprising palladium acetate and a bulky phosphine ligand like S-PHOS—the reaction proceeds with high efficiency to extend the carbon chain. This step is crucial as it installs the four-carbon tether required for the eventual cyclization. The use of zinc reagents offers a distinct advantage over other organometallic species due to their tolerance of various functional groups, including the protected amino moieties, allowing for a convergent synthesis that minimizes side reactions. Furthermore, the subsequent reduction of nitro groups (if the starting material contains them) and the careful management of protecting groups ensure that the final cyclization substrate is perfectly poised for ring closure.
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages for procurement and supply chain teams tasked with securing high-volume supplies of complex pharmaceutical intermediates. The most significant benefit lies in the drastic simplification of the purification workflow; by eliminating the need for repetitive column chromatography, the process becomes amenable to standard crystallization and extraction techniques that are far more cost-effective and scalable. This reduction in downstream processing directly translates to substantial cost savings in manufacturing, as it lowers solvent consumption, reduces labor hours, and increases the overall throughput of the production facility. Moreover, the avoidance of hazardous nitration steps enhances the safety profile of the manufacturing site, reducing regulatory burdens and insurance costs associated with handling energetic materials. For supply chain heads, this means a more reliable source of supply with reduced risk of batch failures or delays caused by complex purification bottlenecks, ensuring consistent availability of this critical ADC building block.
How to Synthesize 5,8-Diamino-3,4-dihydro-2H-1-naphthalenone Efficiently
The implementation of this advanced synthetic protocol requires precise control over reaction conditions and reagent stoichiometry to maximize yield and purity. The process generally begins with the protection of the starting aniline, followed by the generation of the organozinc species under strictly anhydrous conditions to prevent decomposition. The coupling reaction is typically conducted in polar aprotic solvents like DMF at moderate temperatures, ensuring complete conversion before quenching. Subsequent steps involve the hydrolysis of the ester functionality to the corresponding carboxylic acid, which then undergoes the critical Friedel-Crafts cyclization mediated by polyphosphoric acid or Lewis acids like aluminum chloride.
- Protect the amino group of the starting bromoaniline derivative using acetic anhydride to form the acetamide intermediate.
- Prepare the organozinc reagent (4-ethoxy-4-oxobutyl zinc bromide) by reacting ethyl 4-bromobutyrate with activated zinc powder in anhydrous DMF.
- Perform the palladium-catalyzed cross-coupling reaction between the protected bromoaniline and the zinc reagent using Pd(OAc)2 and S-PHOS ligand.
- Reduce the nitro group (if present) using Pd/C hydrogenation, followed by acetylation to secure the second amino protecting group.
- Hydrolyze the ester to the acid, then perform Friedel-Crafts cyclization using polyphosphoric acid (PPA) or AlCl3 to close the ring, followed by deprotection.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its feasibility for their specific production needs. The following questions address common inquiries regarding the chemical robustness, scalability, and regulatory implications of this new method. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of why this route represents a significant advancement over the state of the art.
Q: Why is the new synthesis route for Compound I superior to conventional nitration methods?
A: Conventional routes rely on nitration reactions which generate difficult-to-separate isomers and require multiple column chromatography steps, resulting in low total yields (15.6%-33.2%). The new method utilizes Friedel-Crafts cyclization of protected aminobenzenebutyric acid, completely avoiding nitration and oxidation steps, thereby significantly improving purity and scalability for industrial production.
Q: What are the key intermediates in this novel synthetic pathway?
A: The process relies on novel intermediates such as Compound D (di-acetylated amino ester) and Compound E (di-acetylated amino acid). These stable intermediates allow for precise control over the regioselectivity of the subsequent cyclization step, ensuring the formation of the correct 5,8-diamino substitution pattern required for camptothecin derivatives.
Q: How does this process impact the supply chain for Antibody-Drug Conjugates (ADCs)?
A: By eliminating hazardous nitration steps and complex purification procedures, the new route enhances supply chain reliability and safety. It offers a more direct path to Compound I, the critical precursor for CPT-1 and other topoisomerase I inhibitors used in ADCs like Enhertu and Trodelvy, reducing lead times and manufacturing risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,8-Diamino-3,4-dihydro-2H-1-naphthalenone Supplier
As the demand for next-generation ADC therapies continues to surge, the ability to manufacture key intermediates like 5,8-diamino-3,4-dihydro-2H-1-naphthalenone with high efficiency and purity becomes a strategic imperative for pharmaceutical companies. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals by leveraging this cutting-edge synthetic technology. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to navigate the complexities of Friedel-Crafts cyclizations and palladium-catalyzed couplings. Our rigorous QC labs and commitment to stringent purity specifications ensure that every batch meets the exacting standards required for clinical and commercial API manufacturing, providing you with a secure and compliant supply chain partner.
We invite you to engage with our technical team to explore how this optimized route can enhance your project economics and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential reductions in COGS and lead times specific to your volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your ADC pipeline remains robust and competitive in the global market.
