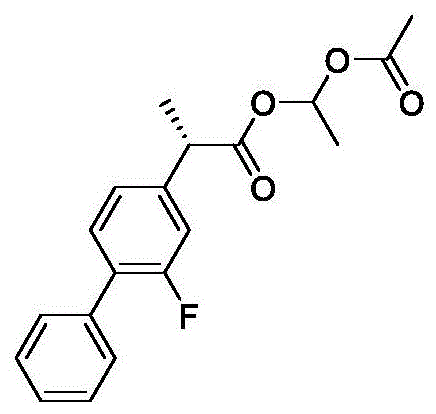
Breakthrough Synthesis of High Optical Purity 2S-(+)-Flurbiprofen Axetil for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for chiral non-steroidal anti-inflammatory drugs (NSAIDs) that minimize side effects while maximizing therapeutic efficacy. A significant advancement in this domain is detailed in patent CN112321424B, which discloses a novel preparation method for 2S-(+)-flurbiprofen axetil with exceptionally high optical purity. This prodrug is critical because the 2S-(+) enantiomer provides the desired cyclooxygenase inhibiting activity, whereas the 2R-(-) enantiomer contributes to gastrointestinal toxicity without analgesic benefit. The disclosed technology overcomes the longstanding technical difficulty of racemization at the alpha-carbon position during esterification. By employing a synergistic system of inorganic bases and phase transfer catalysts, the process achieves yields exceeding 90% and optical purity surpassing 99.9%. For global procurement teams and R&D directors, this represents a pivotal shift towards more reliable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in CN105777544B, typically relied on organic bases to drive the esterification of flurbiprofen. While organic bases offer good solubility, they introduce severe downstream processing challenges. Specifically, these methods necessitate an aqueous quenching step to remove the base, which exposes the sensitive chiral alpha-carbon to hydrolytic conditions. This exposure frequently leads to partial racemization, degrading the optical purity of the final product and necessitating expensive and yield-loss-inducing chiral resolution steps later in the pipeline. Furthermore, to mitigate this racemization, prior processes often limited the base equivalents to sub-stoichiometric levels (0.4-0.8 eq), resulting in incomplete conversion of the starting material and overall yields often struggling to remain above 50% or 80%. These inefficiencies create significant bottlenecks in cost reduction in API manufacturing and complicate supply chain reliability.
The Novel Approach
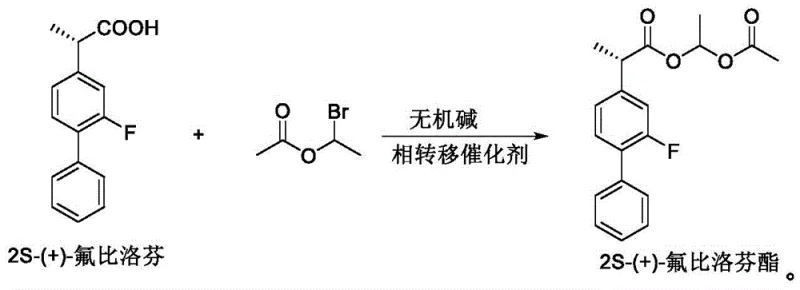
The innovative strategy presented in patent CN112321424B fundamentally rethinks the catalytic environment by utilizing inorganic bases, such as cesium carbonate or potassium carbonate, in conjunction with a phase transfer catalyst (PTC). This approach effectively solubilizes the inorganic base in the organic reaction medium, allowing for efficient deprotonation of the carboxylic acid without the need for excessive water during the reaction phase. Crucially, the post-reaction workup involves simple suction filtration to remove the inorganic salts, completely bypassing the aqueous quenching step that triggers racemization in conventional methods. This modification not only preserves the stereochemical integrity of the 2S-(+) configuration but also allows for the use of stoichiometric or near-stoichiometric base amounts, driving the reaction to completion. The result is a streamlined process that delivers superior yields and purity profiles compared to legacy organic base protocols.
Mechanistic Insights into Phase Transfer Catalyzed Esterification
The core chemical challenge in synthesizing 2S-(+)-flurbiprofen axetil lies in the lability of the chiral center located at the alpha-position relative to both the carbonyl group and the benzyl ring. This specific structural motif is highly susceptible to base-catalyzed enolization, which leads to racemization. In the patented mechanism, the phase transfer catalyst, typically a quaternary ammonium salt like benzyltriethylammonium chloride, acts as a molecular shuttle. It transports the carboxylate anion of the flurbiprofen into the organic phase where it reacts with 1-bromoethyl acetate. The use of mild inorganic bases like Cs2CO3 ensures that the basicity is sufficient to form the carboxylate but controlled enough to minimize direct attack on the chiral proton when shielded within the organic phase ion pair. This delicate balance prevents the formation of the planar enolate intermediate that would otherwise allow for the inversion of configuration, thereby locking in the desired 2S-(+) stereochemistry throughout the transformation.
Furthermore, the impurity control mechanism is intrinsically linked to the workup procedure. In traditional aqueous workups, the interface between the organic product and the alkaline aqueous layer is a hotspot for epimerization. By replacing this with a solid-liquid separation (filtration), the patent eliminates the primary vector for impurity generation. The inorganic salt byproducts are removed as solids, leaving the product in the organic solution largely untouched by hydrolytic agents. Subsequent purification via column chromatography using acidic or neutral silica gel further ensures that any trace acidic or basic impurities are neutralized or retained, resulting in a final product with a Dr value (diastereomeric ratio) consistently above 99.9%. This rigorous control over the reaction environment is essential for meeting the stringent purity specifications required for injectable NSAID formulations.
How to Synthesize 2S-(+)-Flurbiprofen Axetil Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for laboratory and pilot-scale production, emphasizing temperature control and reagent stoichiometry. The process begins with the dissolution of the chiral acid in a solvent like dichloromethane, followed by cooling to suppress thermal racemization. The sequential addition of the alkylating agent and the catalyst system is critical to maintaining reaction homogeneity and rate. While the patent details specific experimental examples, the generalized workflow below captures the essential operational parameters for achieving the reported high yields and optical purity. For detailed standard operating procedures and safety data, please refer to the technical guidelines below.
- Dissolve 2S-(+)-flurbiprofen in an organic solvent such as dichloromethane or acetone and cool the mixture to between 0°C and 10°C.
- Add 1-bromoethyl acetate and a quaternary ammonium phase transfer catalyst, followed by the slow addition of an inorganic base like cesium carbonate.
- Stir the reaction at controlled temperatures (10-20°C), filter off inorganic salts, wash the organic phase, and purify the crude oil via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this inorganic base-mediated process offers tangible strategic benefits beyond mere chemical elegance. The shift from specialized organic bases to commodity inorganic salts like cesium or potassium carbonate represents a direct material cost optimization. Additionally, the simplification of the workup procedure from a multi-stage aqueous extraction to a single filtration step drastically reduces solvent consumption and processing time. This efficiency gain translates into higher throughput capacity for existing manufacturing assets, effectively increasing production volume without the need for capital-intensive equipment upgrades. The robustness of the method also implies a more stable supply of high-purity intermediates, reducing the risk of batch failures due to optical purity deviations.
- Cost Reduction in Manufacturing: The elimination of expensive organic bases and the reduction in solvent usage for extractions significantly lowers the variable cost per kilogram of the produced intermediate. By avoiding the need for chiral resolution of the final ester—a step often required to fix racemization issues in older methods—the process saves substantial resources and time. The high conversion rate ensures that the valuable chiral starting material, 2S-(+)-flurbiprofen, is utilized with maximum efficiency, minimizing waste and improving the overall economic viability of the synthesis route.
- Enhanced Supply Chain Reliability: The reliance on widely available inorganic reagents and standard solvents mitigates the risk of supply disruptions associated with niche specialty chemicals. The simplified process flow reduces the complexity of the manufacturing schedule, allowing for faster turnaround times between batches. This agility is crucial for responding to fluctuating market demands for analgesic drugs. Furthermore, the high consistency of the product quality reduces the need for extensive re-testing or re-processing, ensuring a steady flow of certified material to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful amplification experiments in the patent data. The avoidance of large volumes of aqueous waste streams containing organic bases simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The solid waste generated (inorganic salts) is generally easier to handle and dispose of compared to complex organic aqueous mixtures. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations in major pharmaceutical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for evaluating the process feasibility. Understanding these nuances is vital for technical teams assessing the integration of this method into their existing production portfolios.
Q: Why is inorganic base preferred over organic base for this specific esterification?
A: Traditional methods using organic bases often require aqueous quenching which promotes racemization at the sensitive alpha-chiral center. The patented inorganic base method allows for solid-liquid separation via filtration, avoiding water contact and preserving >99.9% optical purity.
Q: What is the expected yield and purity profile of this new process?
A: Experimental data from the patent indicates that optimizing the phase transfer catalyst and inorganic base ratio can achieve reaction yields exceeding 90% with a diastereomeric ratio (Dr) greater than 99.9%, significantly outperforming prior art methods.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commodity inorganic salts and standard solvents like dichloromethane or acetone. The workup involves simple filtration rather than complex extractions, making it highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2S-(+)-Flurbiprofen Axetil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of optical purity and process robustness in the production of advanced pharmaceutical intermediates. Our team of expert chemists has extensively analyzed the methodology described in CN112321424B and possesses the technical capability to implement this phase transfer catalyzed route at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the >99.9% optical purity required for this sensitive molecule.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By leveraging our manufacturing expertise, you can secure a consistent supply of high-quality 2S-(+)-flurbiprofen axetil while achieving significant operational efficiencies. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project milestones and quality standards.
