Scalable Green Synthesis of Diphenyldiazene Derivatives via Zirconium Hydroxide Catalysis
The chemical manufacturing landscape is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective synthetic routes, a shift vividly illustrated by the technological advancements disclosed in patent publication CN115337919A. This pivotal intellectual property introduces a groundbreaking application of zirconium hydroxide as a heterogeneous catalyst for the oxidative coupling of aniline and its derivatives to produce diphenyldiazene, commonly known as azobenzene, and its various functionalized analogues. For R&D directors and process chemists, this development represents a departure from the traditional reliance on scarce and prohibitively expensive noble metal catalysts such as gold or copper complexes, offering instead a robust system based on earth-abundant zirconium. The methodology not only simplifies the reaction workflow but also aligns perfectly with modern green chemistry principles by utilizing tert-butyl hydroperoxide (TBHP) as a clean oxidant, thereby mitigating the severe safety and environmental risks associated with older oxidation protocols that employed toxic heavy metals or unstable peracids.
Furthermore, the strategic implementation of this zirconium-based catalytic system addresses critical pain points in the supply chain for fine chemical intermediates, particularly regarding raw material availability and process scalability. By enabling the conversion of readily available industrial aniline feedstocks into high-value azo compounds under mild thermal conditions, typically around 40°C, the process drastically reduces energy consumption and operational complexity. This innovation is particularly relevant for manufacturers seeking a reliable pharmaceutical intermediate supplier who can guarantee consistent quality without the volatility of precious metal markets. The ability to achieve high conversion rates and selectivity without the need for complex ligand systems or harsh reaction environments positions this technology as a cornerstone for next-generation industrial synthesis, promising substantial improvements in both economic efficiency and environmental compliance for global chemical enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diphenyldiazene and its derivatives has been plagued by significant technical and economic hurdles that hinder efficient large-scale production. Traditional pathways often relied on the reduction of nitro compounds or the coupling of diazonium salts, processes that are notoriously multi-step, generate vast amounts of hazardous waste, and suffer from poor atom economy. More recent catalytic approaches attempted to streamline this via direct oxidative coupling but frequently necessitated the use of noble metals like gold supported on titanium dioxide or copper-bromide complexes, which introduce exorbitant raw material costs and complicate downstream purification due to metal leaching. Additionally, many of these legacy methods required aggressive oxidants such as peracetic acid or lead tetraacetate, which pose severe explosion hazards and create toxic byproduct streams that are increasingly difficult to dispose of under tightening environmental regulations. The combination of high-pressure oxygen requirements, elevated temperatures, and the need for specialized additives further exacerbated the operational risks, making these conventional routes less attractive for modern, safety-conscious manufacturing facilities.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel zirconium hydroxide-catalyzed pathway offers a streamlined, single-pot solution that dramatically simplifies the production architecture. As illustrated in the reaction scheme below, this method utilizes inexpensive zirconium hydroxide, which can be sourced commercially or prepared via simple precipitation, acting as a highly active and selective catalyst in conjunction with tert-butyl hydroperoxide. The reaction proceeds smoothly in common organic solvents like acetic acid, eliminating the need for exotic or hazardous reaction media. This approach not only bypasses the use of precious metals entirely but also operates under atmospheric pressure and mild temperatures, significantly enhancing process safety profiles. The result is a robust synthetic route that delivers high yields of the target azo compounds while minimizing the formation of over-oxidized impurities, thereby reducing the burden on purification units and improving the overall sustainability of the manufacturing process.
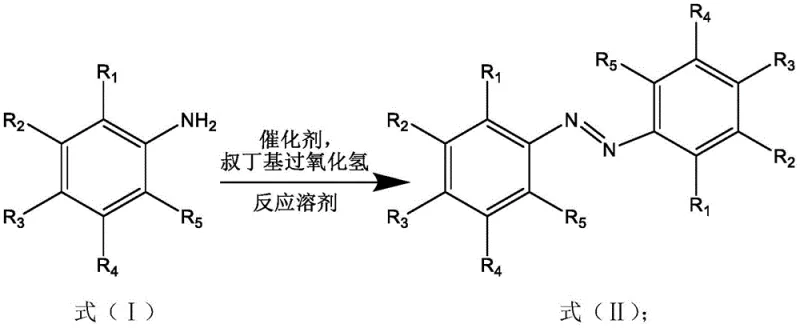
Mechanistic Insights into Zirconium Hydroxide-Catalyzed Oxidative Coupling
From a mechanistic perspective, the efficacy of zirconium hydroxide in this transformation lies in its unique surface properties and Lewis acidity, which facilitate the activation of the amine substrate towards oxidative dimerization. The zirconium centers likely coordinate with the nitrogen lone pair of the aniline, lowering the oxidation potential and enabling the tert-butyl hydroperoxide to efficiently abstract hydrogen atoms to form the reactive nitroso or radical intermediates necessary for N-N bond formation. This specific interaction ensures high chemoselectivity, preventing the common side reaction where anilines are over-oxidized to nitrobenzenes or azoxybenzenes, a frequent issue with less selective oxidants. The heterogeneous nature of the catalyst also suggests that the reaction occurs at the solid-liquid interface, which can simplify catalyst recovery and potentially allow for recycling, although the patent emphasizes the low cost of the catalyst making single-use economically viable. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters, as the balance between catalyst loading, oxidant stoichiometry, and solvent polarity directly influences the rate of radical generation and coupling efficiency.
Moreover, the versatility of this catalytic system is evidenced by its remarkable tolerance to various electronic and steric environments on the aromatic ring, allowing for the synthesis of a diverse library of functionalized azobenzenes. Whether the substrate bears electron-donating groups like methyl or methoxy, or electron-withdrawing substituents such as chloro and bromo, the zirconium catalyst maintains high activity, delivering yields that consistently exceed 80% and often reach upwards of 95% under optimized conditions. For instance, the synthesis of 2,2'-dimethyl-diphenyldiazene from o-toluidine proceeds with excellent efficiency, demonstrating that steric hindrance at the ortho position does not significantly impede the catalytic cycle. This broad substrate scope is visually represented by the successful formation of various derivatives, confirming that the electronic nature of the substituents does not deactivate the catalyst, making this method universally applicable for producing a wide range of high-purity pharmaceutical intermediates and fine chemicals required for complex molecular architectures.
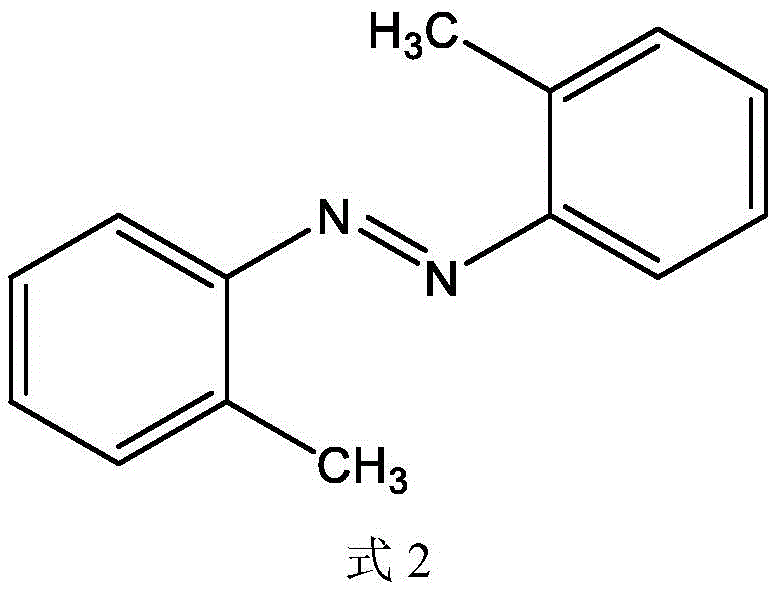
How to Synthesize Diphenyldiazene Efficiently
Implementing this advanced synthetic route requires careful attention to reaction parameters to maximize yield and purity, though the overall procedure is designed for operational simplicity. The process begins with the suspension of the zirconium hydroxide catalyst in the chosen solvent, typically acetic acid, followed by the addition of the aniline substrate. The oxidant, tert-butyl hydroperoxide, is then introduced gradually to control the exotherm and ensure steady conversion. Maintaining the reaction temperature within the optimal window of 30°C to 50°C is critical; temperatures that are too low may result in incomplete conversion, while excessive heat could promote side reactions or decomposition of the oxidant. Following the reaction period, usually around 12 hours, the mixture is subjected to standard workup procedures including filtration to remove the solid catalyst, distillation to recover the solvent, and recrystallization to obtain the final product in high purity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding zirconium hydroxide catalyst and aniline derivative to an organic solvent such as acetic acid.
- Slowly add tert-butyl hydroperoxide (TBHP) as the oxidant while maintaining the reaction temperature between 30°C and 50°C.
- Stir the mixture for approximately 12 hours, then filter, distill, and recrystallize to isolate the high-purity diphenyldiazene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this zirconium-catalyzed technology translates into tangible strategic benefits that extend far beyond simple chemical yield metrics. The primary advantage lies in the drastic reduction of raw material costs, achieved by replacing expensive noble metal catalysts with commodity-grade zirconium hydroxide, which is abundant and inexpensive. This substitution eliminates the financial volatility associated with precious metal markets and removes the need for costly metal scavenging resins or complex purification steps to meet strict residual metal specifications required in pharmaceutical applications. Furthermore, the use of readily available aniline derivatives as starting materials ensures a stable and secure supply chain, reducing the risk of production delays caused by sourcing specialized or regulated precursors. The mild reaction conditions also contribute to lower utility costs, as the process does not require high-pressure reactors or extensive heating and cooling infrastructure, thereby lowering the total cost of ownership for the manufacturing asset.
- Cost Reduction in Manufacturing: The elimination of noble metals such as gold or copper from the catalyst formulation results in a significant decrease in direct material expenses, while the simplified workup process reduces labor and waste disposal costs. By avoiding the use of hazardous oxidants like peracetic acid, the facility also saves on safety compliance and environmental treatment expenditures, leading to a leaner and more profitable production model. The high selectivity of the reaction minimizes the formation of byproducts, which maximizes the utilization of raw materials and reduces the volume of waste requiring treatment, further enhancing the economic viability of the process on a commercial scale.
- Enhanced Supply Chain Reliability: Utilizing common industrial chemicals like acetic acid and tert-butyl hydroperoxide ensures that the supply chain is resilient against disruptions, as these reagents are produced globally in massive quantities. The robustness of the catalyst means that production schedules are less likely to be impacted by catalyst deactivation or the need for frequent replenishment of specialized reagents. This reliability allows for more accurate forecasting and inventory management, ensuring that downstream customers receive their orders on time without the variability often associated with more finicky catalytic systems that require precise handling or storage conditions.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at atmospheric pressure and moderate temperatures, removing the engineering barriers often faced when transitioning from benchtop to pilot or commercial plant scales. The green nature of the chemistry, characterized by the absence of heavy metal contamination and the use of safer oxidants, facilitates easier regulatory approval and permits for new production lines. This environmental compatibility not only future-proofs the manufacturing site against tightening emissions regulations but also enhances the brand reputation of the company as a sustainable manufacturer, which is increasingly a key differentiator in global B2B markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zirconium hydroxide catalytic system, derived from the specific data and advantages outlined in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for the production of azo compounds. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios to achieve better efficiency and sustainability goals.
Q: What are the advantages of using zirconium hydroxide over traditional noble metal catalysts?
A: Zirconium hydroxide is significantly cheaper and more environmentally friendly than gold or copper-based catalysts. It eliminates the need for expensive heavy metal removal steps, reducing overall production costs and environmental impact while maintaining high catalytic activity and selectivity.
Q: Can this method be applied to substituted aniline derivatives?
A: Yes, the process demonstrates excellent substrate scope. It effectively converts various substituted anilines, including those with methyl, chloro, bromo, and methoxy groups, into their corresponding diphenyldiazene derivatives with high yields, often exceeding 90%.
Q: Is the reaction condition safe for large-scale industrial production?
A: The reaction operates under mild conditions, typically around 40°C and atmospheric pressure, using tert-butyl hydroperoxide as a manageable oxidant. This avoids the safety hazards associated with high-pressure oxygen systems or explosive peracids used in conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyldiazene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this zirconium-catalyzed synthesis route and have integrated such advanced methodologies into our core capabilities to serve the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of diphenyldiazene or its derivatives meets the highest international standards, providing our partners with the confidence they need for their own downstream applications. Our commitment to technical excellence means we can adapt this green chemistry protocol to meet specific customer requirements, delivering high-quality intermediates that drive innovation in the pharmaceutical and fine chemical sectors.
We invite you to engage with our technical procurement team to discuss how this cost-effective and sustainable synthesis method can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in catalytic oxidative coupling can become a strategic asset for your organization, ensuring a reliable supply of high-performance chemical intermediates for years to come.
