Scalable Enzymatic Synthesis of Chiral GNE-140 Intermediates for Industrial Pharmaceutical Manufacturing
Scalable Enzymatic Synthesis of Chiral GNE-140 Intermediates for Industrial Pharmaceutical Manufacturing
The development of Lactate Dehydrogenase A (LDHA) inhibitors represents a significant frontier in oncology therapeutics, specifically targeting the glycolytic metabolism of hypoxic tumor cells. Patent CN109369631B discloses a groundbreaking synthesis method for a key intermediate, 6,6-aromatic ring substituted-2,4-piperidinedione, which serves as the core scaffold for the potent inhibitor GNE-140. This technology addresses critical bottlenecks in the production of high-purity chiral intermediates by replacing traditional resolution methods with a highly selective enzymatic cyclization strategy. For R&D directors and procurement specialists, this patent offers a pathway to significantly reduce manufacturing complexity while ensuring the stringent stereochemical purity required for clinical efficacy. The structural integrity of the final API, GNE-140, relies heavily on the precise construction of this piperidinedione core, as illustrated in the molecular architecture below.
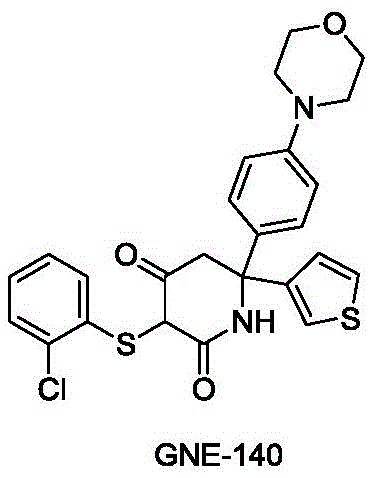
The strategic implementation of this synthesis route allows for the direct generation of single-configuration isomers with an enantiomeric excess (ee) value exceeding 99%, effectively bypassing the costly and material-intensive chiral resolution steps found in earlier literature such as WO2015140133A1. By integrating chiral auxiliaries early in the sequence and leveraging biocatalysis for the ring-closing step, the process achieves a level of operational simplicity and environmental compatibility that is essential for modern green chemistry initiatives in the pharmaceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
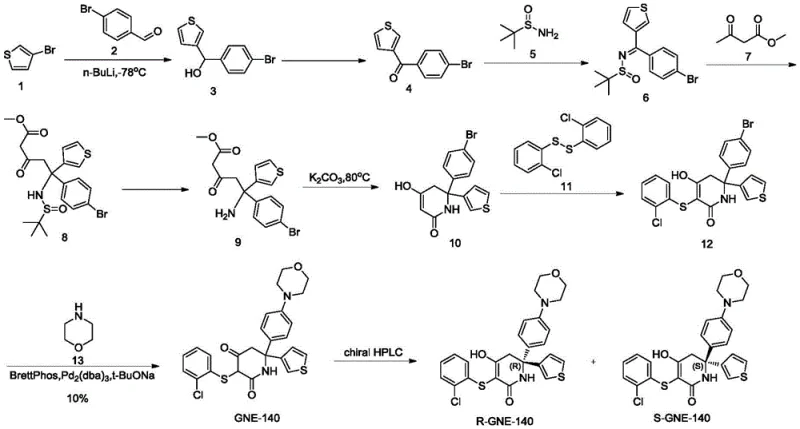
Prior art methodologies for synthesizing the GNE-140 scaffold, as detailed in documents like WO2015140133A1 and ACS Medicinal Chemistry Letters, suffer from severe scalability and efficiency drawbacks that hinder commercial viability. The traditional route typically involves the preparation of a racemic mixture followed by chiral column resolution, a process characterized by a maximum theoretical yield of 50% for the desired isomer and substantial waste generation. Furthermore, the chemical transformations often demand harsh reaction conditions, such as the use of n-butyllithium at cryogenic temperatures of -78°C, which imposes rigorous demands on reactor equipment and energy consumption. When attempting to amplify these reactions to hectogram or kilogram scales, the yield of the critical cyclization step drops precipitously to below 40%, accompanied by the formation of up to 50% decomposition byproducts that are notoriously difficult to separate. These factors collectively result in high production costs, extended lead times, and significant safety risks associated with handling pyrophoric reagents on a large scale.
The Novel Approach
In stark contrast, the methodology disclosed in CN109369631B introduces a robust and scalable alternative that fundamentally reengineers the synthetic logic. The novel approach utilizes a chiral sulfinamide auxiliary to establish stereochemistry early in the linear sequence, thereby avoiding the need for late-stage resolution of the final bicyclic system. The cornerstone of this innovation is the replacement of thermal or chemical cyclization with an enzyme-catalyzed ring closure, utilizing immobilized lipases such as Novozym 435 or Lipozyme TL IM. This biocatalytic step proceeds under mild conditions, typically between 25°C and 60°C, eliminating the thermal degradation issues plaguing the prior art. Additionally, the strategy incorporates the morpholine moiety at an earlier stage of the synthesis, which circumvents the low-yielding coupling reactions observed in previous attempts. The overall result is a streamlined process with high atom economy, simplified purification protocols, and a total yield that supports industrial manufacturing requirements.
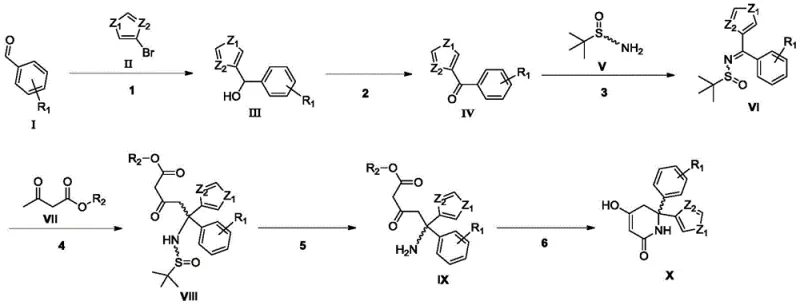
Mechanistic Insights into Enzymatic Kinetic Cyclization
The mechanistic elegance of this synthesis lies in the precise orchestration of chemocatalysis and biocatalysis to achieve absolute stereocontrol. The process begins with the formation of a chiral sulfinimine intermediate, which directs the subsequent Michael addition of a beta-keto ester to generate a quaternary carbon center with high diastereoselectivity. Following the removal of the chiral auxiliary via acidolysis, the resulting amino-dicarbonyl precursor undergoes an intramolecular condensation. Unlike traditional base-mediated cyclizations that produce racemates, the patented method employs a lipase to catalyze this ring closure. The enzyme's active site provides a chiral environment that selectively facilitates the formation of the desired (R) or (S) configured piperidinedione ring while suppressing the formation of the opposite enantiomer. This enzymatic kinetic resolution or dynamic kinetic process ensures that the final product emerges with an ee value greater than 99%, as confirmed by chiral HPLC analysis in the patent examples.
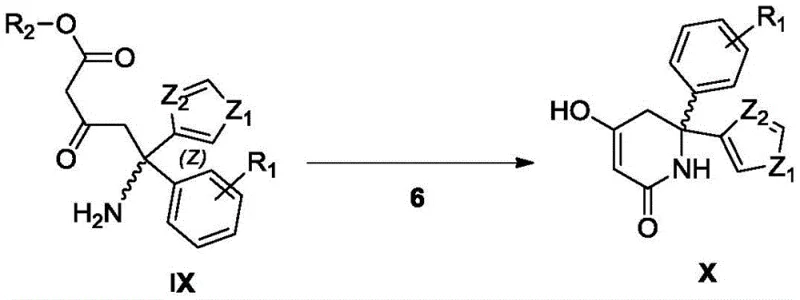
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions such as polymerization or elimination that are common under strong basic conditions. The specificity of the enzyme reduces the complexity of the crude reaction mixture, allowing for simpler work-up procedures such as filtration and crystallization rather than extensive chromatographic purification. This reduction in impurity burden is critical for meeting the rigorous quality standards demanded by regulatory bodies for oncology drug substances, ensuring that the supply chain remains robust and compliant with Good Manufacturing Practices (GMP).
How to Synthesize 6,6-Aromatic Ring Substituted-2,4-Piperidinedione Efficiently
The synthesis of this critical LDHA inhibitor intermediate is achieved through a six-step sequence that balances chemical efficiency with stereochemical precision. The route initiates with a Grignard addition to a functionalized benzaldehyde, followed by oxidation to the corresponding ketone. Subsequent condensation with tert-butylsulfinamide establishes the chiral framework, which is then elaborated via a Michael addition with a beta-keto ester. The final stages involve acidic hydrolysis to unmask the amine and the pivotal enzymatic cyclization to form the piperidinedione core. This logical progression allows for the introduction of diverse substituents, providing flexibility for analog synthesis while maintaining high overall yields. For detailed laboratory protocols and specific reaction parameters, please refer to the standardized guide below.
- Perform Grignard addition of 3-bromothiophene to a substituted benzaldehyde, followed by oxidation to the corresponding ketone.
- Condense the ketone with tert-butylsulfinamide to form a chiral sulfinimine, then react with a beta-keto ester under basic conditions.
- Execute acid hydrolysis to remove the sulfinyl group, followed by enzymatic ring closure using immobilized lipase to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic synthesis route translates into tangible strategic benefits regarding cost stability and supply security. The elimination of chiral preparative HPLC, which is a major cost driver in the production of single-enantiomer drugs, drastically simplifies the downstream processing workflow. By avoiding the need for cryogenic reactors and hazardous organolithium reagents in the final steps, the facility requirements are lowered, reducing both capital expenditure and operational risk. The use of commercially available immobilized enzymes further enhances supply chain reliability, as these biocatalysts are robust, reusable, and sourced from established global suppliers, mitigating the risk of raw material shortages.
- Cost Reduction in Manufacturing: The transition from a resolution-based process to an asymmetric synthesis driven by enzymatic catalysis fundamentally alters the cost structure of the intermediate. By achieving high enantiomeric excess directly, the process avoids the inherent 50% yield loss associated with resolving racemates, effectively doubling the material throughput from the same amount of starting inputs. Furthermore, the mild reaction conditions reduce energy consumption associated with cooling and heating, while the simplified purification steps lower solvent usage and waste disposal costs. These cumulative efficiencies result in a significantly more economical manufacturing process that enhances margin potential for the final API.
- Enhanced Supply Chain Reliability: The reliance on standard organic transformations and widely available biocatalysts ensures a resilient supply chain that is less susceptible to disruptions. Unlike processes dependent on custom-synthesized chiral catalysts or specialized low-temperature infrastructure, this method can be implemented in standard multipurpose pharmaceutical plants. The high yield and reproducibility of the enzymatic step ensure consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed batches or out-of-specification results. This reliability is crucial for maintaining continuous supply to clinical and commercial markets.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, having demonstrated success in moving from gram to multi-gram scales without the yield collapse seen in prior art. The replacement of toxic heavy metal catalysts or harsh bases with biodegradable enzymes aligns with increasingly stringent environmental regulations and corporate sustainability goals. The reduction in hazardous waste generation and the use of greener solvents contribute to a lower environmental footprint, facilitating smoother regulatory approvals and enhancing the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of GNE-140 intermediates, derived directly from the technical disclosures and experimental data within the patent documentation. These insights are intended to clarify the operational advantages and chemical feasibility of the described methodology for potential partners and licensees.
Q: What are the primary limitations of the conventional synthesis route for GNE-140 intermediates?
A: Conventional methods rely on harsh cryogenic conditions (-78°C) using n-butyllithium and require chiral column resolution of racemates, leading to low atom economy, difficult byproduct separation, and yields dropping below 40% upon scale-up.
Q: How does the enzymatic cyclization step improve the process efficiency?
A: The use of immobilized lipases such as Novozym 435 allows for ring closure under mild conditions (25-60°C) with exceptional stereoselectivity, achieving ee values greater than 99% without the need for expensive chiral chromatography.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method avoids extreme low temperatures in the final steps, utilizes commercially available enzymes, and demonstrates high total yields with simple post-treatment, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GNE-140 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in enzymatic processes and asymmetric synthesis positions us as an ideal partner for bringing complex LDHA inhibitors to market.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this enzymatic platform. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring your supply chain is optimized for speed, quality, and cost-effectiveness.
