Advanced Enzymatic Synthesis of D-Heterocyclic Amino Acids for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for producing chiral building blocks, and patent CN108300744B presents a significant breakthrough in this domain. This intellectual property details a novel synthetic method for D-heterocyclic amino acids, utilizing a specific diaminopimelate dehydrogenase derived from Novibacillus thermophilus. Unlike traditional chemical approaches that often rely on harsh conditions and toxic catalysts, this biocatalytic route operates under mild physiological conditions, offering a cleaner and more selective alternative. The technology addresses the critical need for high-purity non-natural amino acids, which serve as essential intermediates in the development of advanced peptide therapeutics and agrochemicals. By leveraging the high stereoselectivity of the enzyme designated as SEQ ID NO:1, manufacturers can achieve superior optical purity without the burden of complex chiral resolution steps. This report analyzes the technical merits and commercial implications of this enzymatic process for global supply chain decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral non-natural heterocyclic amino acids has been dominated by chemical methodologies that present substantial operational and environmental challenges. Traditional routes frequently employ asymmetric catalytic hydrogenation using precious metals such as rhodium or ruthenium, which are not only prohibitively expensive but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient. Furthermore, these chemical processes often require large volumes of organic solvents, leading to significant environmental pollution and increased waste disposal costs. Another common strategy involves chiral resolution of racemates, which inherently limits the maximum theoretical yield to fifty percent, resulting in the wastage of valuable starting materials. Additionally, the presence of heterocyclic moieties in the substrate can interfere with metal-ligand coordination, reducing catalytic efficiency and complicating the purification process. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for scalable manufacturing.
The Novel Approach
In contrast, the method disclosed in patent CN108300744B utilizes a biocatalytic strategy that circumvents the drawbacks of metal-catalyzed synthesis. By employing diaminopimelate dehydrogenase, the process achieves high conversion rates and exceptional enantiomeric excess under aqueous conditions. The enzymatic reaction is highly specific, targeting the D-configuration of the heterocyclic amino acid directly from the corresponding keto acid precursor. This specificity eliminates the need for downstream chiral separation, thereby streamlining the production workflow. The reaction conditions are mild, typically operating at temperatures between 30°C and 40°C and a pH range of 7.8 to 9.0, which reduces energy consumption and equipment stress. Moreover, the use of biological catalysts aligns with green chemistry principles, minimizing the use of hazardous organic solvents and reducing the overall environmental footprint of the manufacturing process. This shift from chemical to enzymatic synthesis represents a paradigm change in how complex chiral intermediates are produced commercially.
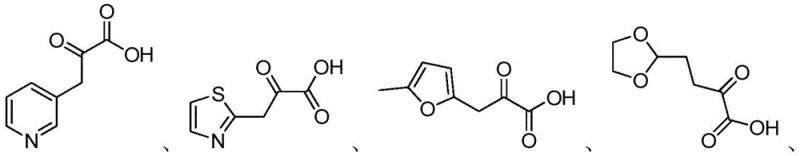
Mechanistic Insights into Diaminopimelate Dehydrogenase Catalysis
The core of this technological advancement lies in the specific activity of the diaminopimelate dehydrogenase enzyme, which facilitates the reductive amination of D-heterocyclic keto acids. The enzyme functions in conjunction with a coenzyme, typically β-NADP+ or β-NAD+, which acts as an electron carrier to drive the reduction of the ketone group to an amine. To maintain catalytic efficiency, the system often incorporates a co-substrate like ammonium formate or ammonium chloride as the amino donor, ensuring a continuous supply of nitrogen for the transformation. The enzyme's active site is structured to accommodate various heterocyclic rings, including pyridine, thiazole, and furan derivatives, demonstrating broad substrate tolerance while maintaining strict stereocontrol. This mechanistic precision ensures that the resulting amino acids possess the desired D-configuration with minimal formation of the L-enantiomer impurity. The stability of the enzyme under the specified reaction conditions allows for prolonged catalytic cycles, enhancing the overall space-time yield of the process.
Impurity control is another critical aspect where this enzymatic method excels over chemical alternatives. In chemical synthesis, side reactions such as over-reduction or non-specific binding of catalysts can generate difficult-to-remove by-products. However, the enzymatic pathway is highly selective, significantly reducing the formation of structural impurities. The patent data indicates that the chiral purity of the products consistently exceeds 98%, as verified by liquid chromatography analysis. This high level of purity simplifies the downstream purification steps, often requiring only basic filtration and pH adjustment followed by ion exchange or crystallization. The use of diatomaceous earth for filtration and hydrochloric acid for reaction termination provides a robust and scalable workup procedure. By minimizing the impurity profile at the source, the process reduces the burden on quality control laboratories and ensures that the final material meets stringent pharmaceutical specifications without extensive reprocessing.
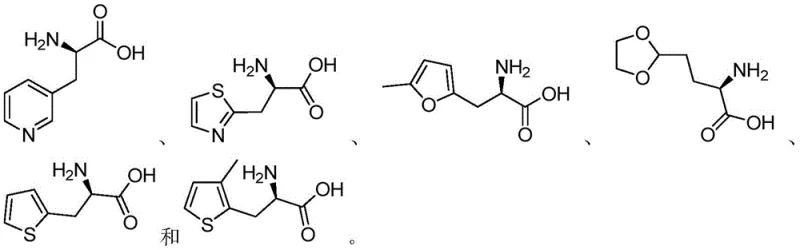
How to Synthesize D-Heterocyclic Amino Acid Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of the reaction mixture, where the D-heterocyclic keto acid is dispersed in a suitable buffer system. The choice of buffer, whether Tris-HCl, triethanolamine, or phosphate, can influence the reaction kinetics and should be optimized based on the specific substrate being used. Once the pH is adjusted to the optimal range, the enzyme and coenzyme are introduced to initiate the biotransformation. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by mixing D-heterocyclic keto acid compounds with an amino donor such as ammonium formate or ammonium chloride in a suitable buffer.
- Add diaminopimelate dehydrogenase (SEQ ID NO: 1) and a coenzyme like β-NADP+ or β-NAD+ to the mixture, maintaining pH between 7.8 and 9.0.
- Incubate the reaction at 30-40°C, then terminate with hydrochloric acid and purify the product through filtration and ion exchange resin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers tangible benefits in terms of cost structure and supply reliability. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the risk associated with the price volatility of rare earth metals. Furthermore, the simplified downstream processing reduces the consumption of solvents and purification media, leading to substantial cost savings in material procurement. The mild reaction conditions also translate to lower energy requirements for heating and cooling, contributing to a more sustainable and cost-effective manufacturing profile. These efficiencies allow for more competitive pricing strategies while maintaining healthy margins, making the supply of these critical intermediates more resilient to market fluctuations.
- Cost Reduction in Manufacturing: The transition to a biocatalytic process fundamentally alters the cost equation by removing the need for expensive chiral ligands and heavy metal catalysts. This reduction in raw material costs is compounded by the higher atom economy of the enzymatic reaction, which minimizes waste generation. Additionally, the simplified purification workflow reduces the consumption of chromatography resins and organic solvents, further lowering operational expenditures. The overall effect is a leaner manufacturing process that delivers high-value products at a reduced cost basis, enhancing the competitiveness of the supply chain.
- Enhanced Supply Chain Reliability: Reliance on complex chemical synthesis often introduces supply chain vulnerabilities due to the sourcing of specialized reagents and catalysts. The enzymatic approach utilizes readily available amino donors and cofactors, which are easier to source in bulk quantities. The robustness of the enzyme under mild conditions also reduces the risk of batch failures due to equipment malfunction or parameter deviation. This stability ensures consistent production output and shorter lead times, allowing supply chain managers to maintain optimal inventory levels and respond more agilely to demand changes.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more straightforward than scaling complex chemical reactions involving hazardous reagents. The aqueous nature of the reaction reduces the risk of fire and explosion, simplifying safety compliance and facility requirements. Moreover, the reduced environmental impact aligns with increasingly stringent global regulations on waste disposal and emissions. This compliance advantage minimizes the risk of regulatory delays and facilitates smoother market access for the final pharmaceutical products, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable reference for technical assessment. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines.
Q: What are the advantages of using diaminopimelate dehydrogenase over chemical synthesis?
A: The enzymatic method avoids expensive precious metal catalysts, eliminates heavy metal residues, and achieves higher chiral purity (>98% ee) under mild aqueous conditions compared to traditional asymmetric hydrogenation.
Q: Which buffers are optimal for this enzymatic conversion?
A: Tris-HCl, triethanolamine, phosphate, or boric acid-sodium hydroxide buffers are effective, with specific selection depending on the substrate to maximize conversion rates and chiral purity.
Q: How is the product purified after the enzymatic reaction?
A: The reaction is terminated with hydrochloric acid to pH ≤ 1, filtered through diatomaceous earth, and the filtrate is purified using strong acid cation exchange resin or extraction methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Heterocyclic Amino Acid Supplier
The technological potential of this enzymatic synthesis route is immense, offering a pathway to high-quality intermediates that are essential for modern drug development. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the market. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the highest industry standards. We are committed to leveraging advanced biocatalytic technologies to deliver superior value to our partners.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your projects. Let us help you secure a reliable and cost-effective supply of high-purity D-heterocyclic amino acids.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
