Advanced Copper-Catalyzed Synthesis of Chiral 2,2'-Dihalogenated Biaryl Intermediates for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of Chiral 2,2'-Dihalogenated Biaryl Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex biaryl frameworks, which serve as critical scaffolds in numerous active pharmaceutical ingredients (APIs) and functional materials. A significant breakthrough in this domain is detailed in patent CN112194559A, which discloses a robust and versatile method for synthesizing chiral and achiral 2,2'-dihalogenated biaryl compounds. This technology leverages a copper-catalyzed ring-opening reaction of diaryl cyclic iodonium salts, offering a streamlined alternative to traditional multi-step syntheses. By utilizing inexpensive halide salts as nucleophiles, this approach not only simplifies the operational procedure but also dramatically enhances the structural diversity of the resulting products. For R&D directors and procurement specialists, this represents a pivotal shift towards more cost-effective and sustainable manufacturing of high-value biaryl intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,2'-dihalogenated biaryl compounds has relied heavily on the Sandmeyer reaction, a process that involves converting arylamines into aryl halides via diazonium salts. This conventional pathway is fraught with significant operational challenges, including the requirement for harsh acidic conditions, low temperatures, and the generation of substantial hazardous waste. Furthermore, accessing axially chiral biaryl halides through these traditional means often necessitates the use of expensive chiral starting materials or cumbersome resolution processes, which severely limits the economic viability for large-scale production. The structural rigidity of these methods also restricts the ability to introduce diverse functional groups, thereby constraining the chemical space available for drug discovery and material science applications.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes diaryl cyclic iodonium salts as electrophilic precursors, which undergo a facile ring-opening reaction in the presence of a copper catalyst and a cheap halide salt. This one-step transformation proceeds under remarkably mild conditions, typically at temperatures between 20°C and 30°C, and can even be conducted in air, eliminating the need for stringent inert atmosphere protocols. The reaction exhibits exceptional functional group tolerance, allowing for the incorporation of various substituents such as methyl, chloro, fluoro, and trifluoromethyl groups without compromising yield. This flexibility enables the rapid generation of a wide library of biaryl derivatives, accelerating the lead optimization phase in drug development while simultaneously reducing the environmental footprint of the synthesis.
Mechanistic Insights into Copper-Catalyzed Ring-Opening
The core of this synthetic innovation lies in the copper-catalyzed activation of the hypervalent iodine species within the diaryl cyclic iodonium salt. The copper catalyst, which can be selected from a range of salts including cuprous iodide, cuprous bromide, or copper trifluoromethanesulfonate, facilitates the nucleophilic attack of the halide ion on the aromatic ring. This interaction triggers the cleavage of the carbon-iodine bond in the cyclic structure, effectively opening the ring to form the desired 2,2'-dihalogenated biaryl product. The mechanism is highly efficient, with catalyst loadings as low as 0.5 mol% to 5 mol% proving sufficient to drive the reaction to completion. This high catalytic efficiency minimizes metal contamination in the final product, a critical factor for pharmaceutical applications where residual heavy metals must be strictly controlled.
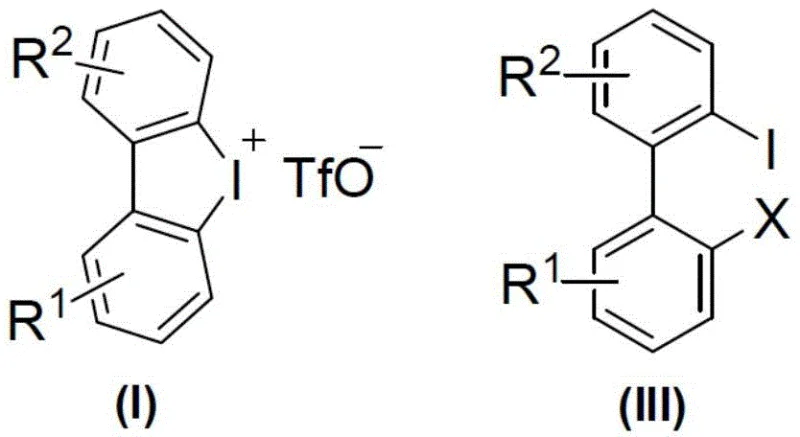
For the synthesis of optically active compounds, the system incorporates chiral ligands, specifically chiral oxazoline derivatives like (R,R)-2,2'-isopropylidenebis(4-phenyl-2-oxazoline), alongside an additive such as calcium oxide. These chiral modifiers create a stereoselective environment around the copper center, guiding the ring-opening process to favor one enantiomer over the other with high precision. The result is the formation of axially chiral biaryl halides with enantiomeric excess (ee) values reaching up to 98%, as demonstrated in the synthesis of compounds like (R)-IIIa and (R)-IIIc. This level of stereocontrol is achieved without the need for complex chiral pool starting materials, representing a significant advancement in asymmetric synthesis technology.
How to Synthesize 2,2'-Dihalogenated Biaryl Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of execution and scalability. The process begins with the uniform mixing of the diaryl cyclic iodonium salt substrate, a stoichiometric amount of a halide salt (such as sodium iodide or tetrabutylammonium bromide), and the copper catalyst in a solvent like dichloromethane. For chiral targets, the chiral ligand and calcium oxide are added at this stage. The reaction mixture is then stirred at room temperature for a period ranging from 6 to 20 hours, depending on the specific substrate reactivity. Post-reaction processing is straightforward, involving concentration of the solution followed by purification via silica gel column chromatography using a petroleum ether-ethyl acetate gradient. Detailed standardized synthesis steps for specific derivatives are outlined below.
- Mix diaryl cyclic iodonium salt, halide salt (e.g., NaI, LiBr), copper catalyst (e.g., CuI), and solvent (dichloromethane) in a reaction vessel.
- For chiral products, add a chiral oxazoline ligand and calcium oxide to the mixture before stirring.
- Stir the reaction at 20-30°C for 6-20 hours, then concentrate and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly address the pain points of modern supply chain management and cost control. The reliance on cheap, commodity-grade halide salts instead of specialized organometallic reagents drastically reduces the raw material costs associated with production. Additionally, the elimination of multi-step substrate preparation and the ability to run reactions in air simplify the manufacturing infrastructure requirements, leading to significant operational expenditure savings. The high yields reported, often exceeding 95% for both achiral and chiral variants, ensure maximum material throughput and minimize waste disposal costs, aligning perfectly with green chemistry principles and regulatory compliance standards.
- Cost Reduction in Manufacturing: The substitution of expensive chiral auxiliaries or complex diazonium precursors with inexpensive halide salts and readily available iodonium salts creates a leaner cost structure. By removing the need for cryogenic conditions and inert gas protection, energy consumption and equipment maintenance costs are substantially lowered. The high atom economy of the ring-opening reaction ensures that a greater proportion of the input mass is converted into the final product, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate ambient air and moisture to a significant degree, reduces the risk of batch failures due to environmental fluctuations. The use of common solvents like dichloromethane and widely sourced copper salts mitigates the risk of supply disruptions for specialized reagents. This reliability translates into more predictable lead times and a steadier flow of high-purity intermediates to downstream customers, ensuring continuity in their own production schedules.
- Scalability and Environmental Compliance: The mild thermal profile of the reaction (20-30°C) makes it inherently safer and easier to scale from laboratory benchtop to industrial reactors without the need for complex heat exchange systems. The simplified work-up procedure, which avoids aqueous quenching of reactive intermediates, reduces the volume of wastewater generated. These factors collectively facilitate a smoother transition to commercial scale production while maintaining strict adherence to environmental regulations regarding waste discharge and solvent usage.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and scope of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction parameters and product quality.
Q: What are the advantages of using diaryl cyclic iodonium salts over traditional Sandmeyer reactions?
A: The use of diaryl cyclic iodonium salts allows for a direct one-step ring-opening reaction under mild conditions (20-30°C), avoiding the harsh acidic conditions and multi-step preparation required for diazonium salts in traditional Sandmeyer reactions. This results in higher atom economy and simpler post-treatment.
Q: Can this method produce optically pure compounds with high enantiomeric excess?
A: Yes, by incorporating chiral oxazoline ligands such as (R,R)-2,2'-isopropylidenebis(4-phenyl-2-oxazoline) and calcium oxide, the method achieves enantiomeric excess (ee) values up to 98%, making it suitable for synthesizing chiral drug intermediates.
Q: Is the process scalable for industrial manufacturing?
A: The process is highly scalable due to its operation in air, mild temperature requirements, and the use of inexpensive, commercially available halide salts. The simple work-up involving concentration and column chromatography facilitates easy adaptation to larger production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dihalogenated Biaryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in advancing the synthesis of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise determination of enantiomeric excess for chiral batches, guaranteeing that every shipment meets the highest industry standards.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data for our catalog of biaryl halides or to discuss route feasibility assessments for your proprietary targets, ensuring a partnership built on technical excellence and supply chain security.
