Advanced Synthesis of 1-Alkyl-2-Trifluoromethyl-5-Amino-1H-Imidazole Intermediates for Commercial Scale-Up
Advanced Synthesis of 1-Alkyl-2-Trifluoromethyl-5-Amino-1H-Imidazole Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly those containing fluorine motifs which enhance metabolic stability and membrane permeability. Patent CN110903245A discloses a groundbreaking methodology for the preparation of 1-alkyl-2-trifluoromethyl-5-amino-1H-imidazole derivatives, addressing a significant technical gap in the existing literature. This innovation provides a reliable pharmaceutical intermediate supplier with a viable route to access these valuable building blocks, which are pivotal in the synthesis of potent antiviral agents such as halogenated alkyl heteroaryl benzoyl compounds. The disclosed process transforms 2-trifluoromethylbenzimidazole through a sequence of alkylation, oxidative ring opening, and Curtius rearrangement, ultimately delivering the target amino-imidazole with high structural fidelity. By leveraging this technology, manufacturers can overcome previous synthetic bottlenecks, ensuring a steady supply of high-purity materials essential for drug development pipelines targeting hepatitis viruses and other infectious diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1-alkyl-2-trifluoromethyl-5-amino-1H-imidazole cores has been fraught with challenges, primarily due to the lack of direct and efficient synthetic methodologies reported in scientific literature. Conventional approaches often struggle with the regioselective introduction of the trifluoromethyl group alongside the amino functionality on the imidazole ring, leading to complex mixtures of isomers that are difficult to separate. Furthermore, many traditional routes rely on expensive or hazardous starting materials that are not readily available on a commercial scale, thereby inflating the cost of goods and complicating supply chain logistics. The absence of a standardized protocol has forced research and development teams to invest excessive resources in route scouting, delaying the progression of promising drug candidates into clinical trials. Without a defined pathway, ensuring batch-to-batch consistency and meeting stringent regulatory purity specifications becomes an arduous task for quality control departments.
The Novel Approach
The novel approach detailed in the patent circumvents these historical hurdles by employing a strategic ring-opening strategy starting from the easily accessible 2-trifluoromethylbenzimidazole. This method elegantly constructs the target scaffold through a logical sequence that maximizes atom economy and operational simplicity. As illustrated in the comprehensive reaction scheme below, the process begins with N-alkylation, followed by a controlled oxidative cleavage of the benzene ring to generate a dicarboxylic acid intermediate. This is subsequently esterified and selectively hydrolyzed to set the stage for a key Curtius rearrangement, which installs the critical amino group with precision. 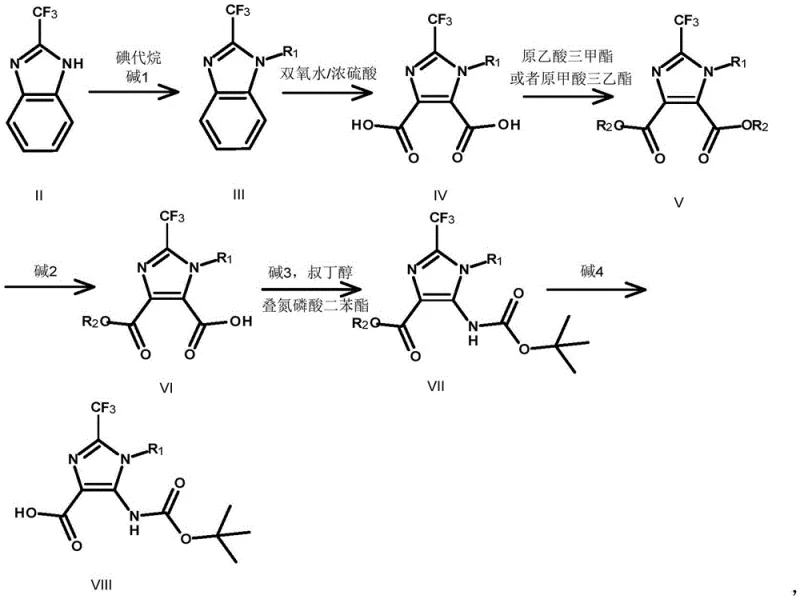 This pathway not only utilizes commodity chemicals but also operates under relatively mild conditions, significantly reducing the safety risks associated with high-pressure or high-temperature reactions. For procurement managers, this translates to cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and simplifying purification protocols, making it an attractive option for large-scale production.
This pathway not only utilizes commodity chemicals but also operates under relatively mild conditions, significantly reducing the safety risks associated with high-pressure or high-temperature reactions. For procurement managers, this translates to cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and simplifying purification protocols, making it an attractive option for large-scale production.
Mechanistic Insights into Oxidative Ring Opening and Curtius Rearrangement
The core of this synthetic innovation lies in the precise execution of the oxidative ring opening and the subsequent Curtius rearrangement, two transformations that define the efficiency of the entire process. The oxidative cleavage of the N-alkylated benzimidazole (Compound III) utilizes hydrogen peroxide in concentrated sulfuric acid to break the aromatic benzene ring, yielding the dicarboxylic acid (Compound IV). This step requires careful thermal management, as the reaction is exothermic, but the patent demonstrates that maintaining temperatures around 100-120°C ensures complete conversion while preserving the sensitive trifluoromethyl group. Following esterification to protect the carboxylic acids, a selective hydrolysis step generates the mono-acid (Compound VI), which serves as the precursor for the nitrogen insertion. The Curtius rearrangement is then triggered using diphenyl phosphorazidate (DPPA) in the presence of a base like triethylamine or DIPEA in t-butanol. 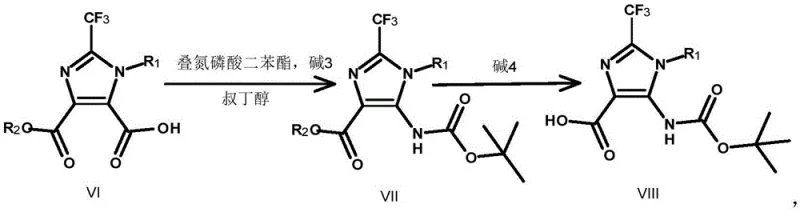 This reaction proceeds via an acyl azide intermediate which rearranges to an isocyanate, subsequently trapped by t-butanol to form the Boc-protected amine (Compound VII). This mechanistic pathway is highly advantageous for R&D directors because it avoids the use of heavy metal catalysts, thereby eliminating the need for costly and time-consuming metal scavenging steps during downstream processing.
This reaction proceeds via an acyl azide intermediate which rearranges to an isocyanate, subsequently trapped by t-butanol to form the Boc-protected amine (Compound VII). This mechanistic pathway is highly advantageous for R&D directors because it avoids the use of heavy metal catalysts, thereby eliminating the need for costly and time-consuming metal scavenging steps during downstream processing.
Furthermore, the final transformation involves the hydrolysis of the remaining ester group in Compound VII to yield the free acid (Compound VIII), followed by a simultaneous decarboxylation and deprotection step to release the final amine (Compound I). The use of acids such as p-toluenesulfonic acid or methanesulfonic acid facilitates this dual transformation efficiently. 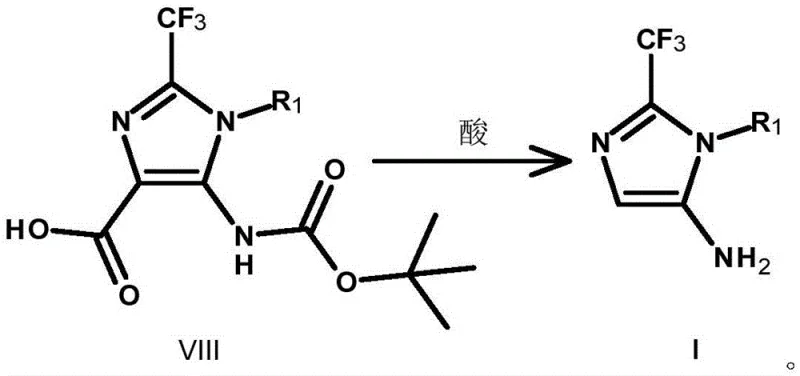 The ability to control the impurity profile during these final steps is critical, as residual protecting groups or incomplete decarboxylation products can compromise the quality of the final active pharmaceutical ingredient. The patent data indicates that optimizing the molar ratios of reagents and reaction times allows for the suppression of side reactions, ensuring that the final product meets the rigorous purity standards required for clinical applications. This level of control over the reaction mechanism provides a solid foundation for scaling the process from gram-scale laboratory experiments to multi-ton commercial batches without sacrificing quality.
The ability to control the impurity profile during these final steps is critical, as residual protecting groups or incomplete decarboxylation products can compromise the quality of the final active pharmaceutical ingredient. The patent data indicates that optimizing the molar ratios of reagents and reaction times allows for the suppression of side reactions, ensuring that the final product meets the rigorous purity standards required for clinical applications. This level of control over the reaction mechanism provides a solid foundation for scaling the process from gram-scale laboratory experiments to multi-ton commercial batches without sacrificing quality.
How to Synthesize 1-Alkyl-2-Trifluoromethyl-5-Amino-1H-Imidazole Efficiently
Implementing this synthesis requires a disciplined approach to reaction conditions and reagent stoichiometry to ensure optimal yields and purity. The process begins with the alkylation of the benzimidazole core, where the choice of base (e.g., cesium carbonate or potassium carbonate) and solvent (DMF) plays a pivotal role in driving the reaction to completion. Subsequent steps involve careful temperature control during the exothermic oxidation and the moisture-sensitive Curtius rearrangement. The detailed标准化 synthesis steps见下方的指南 provide a roadmap for executing these transformations safely and effectively. By adhering to the specified parameters, such as maintaining the hydrolysis temperature between 0-30°C and utilizing appropriate workup procedures like pH adjustment and extraction, manufacturers can consistently produce high-quality intermediates. This structured approach minimizes variability and enhances the robustness of the manufacturing process.
- Alkylate 2-trifluoromethylbenzimidazole (Compound II) with alkyl iodide and base to form Compound III.
- Oxidatively open the benzene ring of Compound III using hydrogen peroxide and sulfuric acid to obtain diacid Compound IV.
- Esterify Compound IV to generate diester Compound V, followed by selective hydrolysis to mono-acid Compound VI.
- Perform Curtius rearrangement on Compound VI using diphenyl phosphorazidate to yield carbamate Compound VII.
- Hydrolyze the ester group of Compound VII to form Compound VIII, then decarboxylate and deprotect to obtain the final amine Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical feasibility. The reliance on readily available starting materials like 2-trifluoromethylbenzimidazole and common alkyl halides mitigates the risk of raw material shortages, ensuring a stable and continuous supply of intermediates. Moreover, the elimination of transition metal catalysts from the synthetic sequence drastically simplifies the purification workflow, removing the need for specialized resin columns or complex filtration systems often required to meet residual metal limits. This streamlining of the downstream process directly contributes to cost reduction in API manufacturing by lowering both material costs and processing time. Additionally, the use of standard organic solvents and reagents facilitates easier sourcing and inventory management, allowing companies to maintain leaner stock levels while responding agilely to market demands.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing inexpensive and abundant reagents such as hydrogen peroxide, sulfuric acid, and alkali hydroxides, avoiding the need for precious metal catalysts or exotic ligands. The high yields reported in the patent examples across multiple steps indicate a material-efficient process that minimizes waste generation and maximizes the output per unit of input. By reducing the number of purification steps and avoiding expensive chromatographic separations in favor of crystallization or simple extractions, the overall cost of goods sold is substantially lowered. This economic advantage allows pharmaceutical companies to allocate resources more effectively towards other critical areas of drug development.
- Enhanced Supply Chain Reliability: The synthetic route is designed with supply chain resilience in mind, utilizing commodity chemicals that are widely produced and distributed globally. This reduces dependency on single-source suppliers for niche reagents, thereby mitigating the risk of disruptions due to geopolitical issues or logistical bottlenecks. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without compromising product quality, further enhances reliability by reducing the likelihood of batch failures. Consequently, manufacturers can offer more predictable lead times and maintain consistent inventory levels, fostering stronger partnerships with their downstream clients.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is well-suited for commercial scale-up due to its moderate operating conditions and the absence of highly toxic or explosive reagents. The oxidative ring opening, while exothermic, is manageable with standard cooling systems, and the Curtius rearrangement is conducted in solution, minimizing the accumulation of potentially unstable azide intermediates. The waste streams generated are primarily aqueous and organic solvents that can be treated using conventional wastewater treatment facilities, aligning with modern green chemistry principles. This compliance with environmental regulations simplifies the permitting process for new production lines and supports the company's sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 1-alkyl-2-trifluoromethyl-5-amino-1H-imidazole intermediates. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect a commitment to transparency and technical excellence, ensuring that all partners have a clear understanding of the value proposition.
Q: What are the key advantages of this synthetic route for 1-alkyl-2-trifluoromethyl-5-amino-1H-imidazole?
A: The process utilizes readily available raw materials like 2-trifluoromethylbenzimidazole and avoids harsh conditions where possible. The oxidative ring opening and Curtius rearrangement steps are well-controlled, ensuring high purity and suitable overall yields for industrial application.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the patent describes reactions using common solvents like DMF, methanol, and t-butanol, and standard reagents such as alkali hydroxides and diphenyl phosphorazidate. The operational simplicity and mild reaction temperatures (e.g., 0-30°C for hydrolysis) support safe scale-up from laboratory to multi-ton production.
Q: How is the trifluoromethyl group preserved during the oxidative ring opening?
A: The oxidative cleavage specifically targets the benzene ring fused to the imidazole core while leaving the trifluoromethyl substituent at the 2-position intact. This chemoselectivity is crucial for maintaining the biological activity profile required for downstream antiviral applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Alkyl-2-Trifluoromethyl-5-Amino-1H-Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and timeliness. We are equipped with state-of-the-art facilities and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-alkyl-2-trifluoromethyl-5-amino-1H-imidazole delivered meets the highest industry standards. Our commitment to quality and consistency makes us the preferred choice for global pharmaceutical companies seeking to secure their supply chains for next-generation antiviral therapies.
We invite you to collaborate with us to explore how this innovative synthetic route can benefit your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating the economic potential of this technology. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
