Industrial Synthesis of Ethyl-3-oxalidene-1-oxa-4-azaspiro Undecane Derivatives via Novel Cyclization
Introduction to Novel Spiro-Cycle Manufacturing Technologies
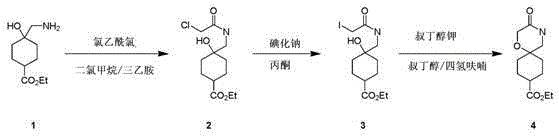
The pharmaceutical industry constantly seeks robust pathways for constructing complex heterocyclic scaffolds, particularly spiro-cyclic systems which are prevalent in modern drug discovery. Patent CN111662245A introduces a significant advancement in the synthesis of ethyl-3-oxalidene-1-oxa-4-azaspiro[5.5]undecan-9-carboxylate, a critical building block for various therapeutic agents. Historically, the construction of such fused ring systems has been plagued by harsh conditions or multi-step sequences that hinder commercial viability. This specific intellectual property outlines a streamlined three-step protocol that transforms accessible starting materials into high-value intermediates with remarkable efficiency. By leveraging standard organic transformations such as acylation and nucleophilic substitution, the methodology circumvents the need for exotic reagents, thereby positioning itself as a reliable pharmaceutical intermediate supplier solution for global supply chains.
The strategic importance of this synthesis lies in its ability to address the technical void where no suitable industrial methods previously existed. For R&D directors evaluating new routes, the predictability of the reaction outcomes is paramount. The patent details a sequence that begins with a stable amino-alcohol precursor, progressing through a chloro-acetyl intermediate and an iodo-substituted species before final ring closure. This logical progression minimizes the formation of intractable impurities often associated with direct cyclization attempts. Furthermore, the explicit documentation of reaction parameters, including specific temperatures and solvent systems, provides a clear blueprint for technology transfer teams aiming to replicate these results in a pilot plant environment without extensive trial-and-error phases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of spiro-oxa-aza cycles has relied on methodologies that suffer from significant drawbacks regarding scalability and cost-effectiveness. Many legacy routes require the use of precious metal catalysts or highly specialized ligands that drive up the cost of goods sold (COGS) prohibitively for large-scale production. Additionally, conventional approaches often involve protecting group strategies that add unnecessary steps, reducing the overall atom economy and generating excessive chemical waste. From a supply chain perspective, reliance on niche reagents creates vulnerability; any disruption in the availability of these specialized chemicals can halt production lines entirely. Moreover, older methods frequently operate under extreme conditions, such as cryogenic temperatures or high-pressure environments, which demand specialized equipment and increase energy consumption, further eroding profit margins in competitive markets.
The Novel Approach
In stark contrast, the methodology described in CN111662245A offers a paradigm shift by utilizing a direct, linear synthesis strategy that prioritizes operational simplicity. The novel approach eliminates the need for complex catalytic systems, instead relying on fundamental organic reactions that are well-understood and easily controlled. By employing a halogen exchange strategy followed by an intramolecular Williamson ether synthesis, the process achieves ring closure under relatively mild thermal conditions. This reduction in complexity translates directly to cost reduction in pharmaceutical intermediate manufacturing, as it lowers both capital expenditure on equipment and operational expenditure on utilities. The route is designed to be telescoped or run in batch mode with standard glass-lined reactors, making it an ideal candidate for facilities looking to diversify their portfolio with high-margin specialty chemicals without massive infrastructure overhauls.
Mechanistic Insights into the Three-Step Cascade Synthesis
The core of this synthetic innovation relies on a carefully orchestrated sequence of nucleophilic substitutions and cyclizations. The first stage involves the N-acylation of ethyl 4-(aminomethyl)-4-hydroxycyclohexanecarboxylate with chloroacetyl chloride. In this step, triethylamine acts as a proton scavenger, neutralizing the hydrochloric acid byproduct and driving the equilibrium towards the formation of the amide bond. This reaction is conducted in dichloromethane at room temperature, ensuring that the sensitive ester functionality remains intact while the amine is selectively acylated. The resulting chloro-amide serves as a pivotal junction point, introducing the necessary electrophilic carbon chain required for subsequent ring formation while maintaining the structural integrity of the cyclohexane core.
Following acylation, the process employs a classic Finkelstein reaction to activate the side chain for cyclization. Treating the chloro-intermediate with sodium iodide in refluxing acetone facilitates a halogen exchange, converting the less reactive chloride into a highly reactive iodide. This transformation is crucial because the iodide is a superior leaving group, significantly lowering the activation energy for the final ring-closing step. The final stage utilizes potassium tert-butoxide as a strong, non-nucleophilic base to deprotonate the pendant hydroxyl group. The resulting alkoxide attacks the adjacent carbon bearing the iodine atom, displacing the iodide and forming the thermodynamically stable six-membered oxazine ring fused to the cyclohexane system. This intramolecular SN2 mechanism ensures high regioselectivity, minimizing the risk of oligomerization or polymerization side reactions.
How to Synthesize Ethyl-3-oxalidene-1-oxa-4-azaspiro Undecane Efficiently
Executing this synthesis requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent data to ensure optimal conversion and purity. The process begins with the dissolution of the starting amino-alcohol in anhydrous dichloromethane, followed by the controlled addition of the acid chloride to manage exotherms. Subsequent steps involve solvent swaps and precise temperature controls, specifically maintaining the iodination step at reflux to drive the equilibrium forward. The final cyclization demands careful handling of the strong base and anhydrous conditions to prevent hydrolysis of the ester moiety. For a comprehensive breakdown of the specific masses, molar equivalents, and workup procedures required to replicate this chemistry, please refer to the standardized protocol below.
- Perform N-acylation of ethyl 4-(aminomethyl)-4-hydroxycyclohexanecarboxylate with chloroacetyl chloride in dichloromethane using triethylamine as a base.
- Execute a Finkelstein reaction by treating the chloro-intermediate with sodium iodide in refluxing acetone to generate the iodo-derivative.
- Conduct intramolecular cyclization using potassium tert-butoxide in a mixture of tert-butanol and tetrahydrofuran under reflux conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the sourcing strategy; all key reagents, including chloroacetyl chloride, sodium iodide, and potassium tert-butoxide, are commodity chemicals available from multiple global vendors. This diversity of supply sources mitigates the risk of single-supplier dependency, ensuring business continuity even during market fluctuations. Furthermore, the elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which are often a bottleneck in API manufacturing. This simplification not only reduces the cost of raw materials but also shortens the overall cycle time from reactor charging to final isolation.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and the avoidance of costly proprietary catalysts. By utilizing inexpensive inorganic salts and common organic solvents, the direct material costs are significantly minimized compared to alternative routes. Additionally, the workup procedures involve standard aqueous washes and crystallization or filtration steps, which are far less capital-intensive than chromatographic purifications. This lean manufacturing approach allows for substantial cost savings that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable production schedule. Since the reactions proceed at atmospheric pressure and moderate temperatures, they are less prone to safety incidents or equipment failures that could cause unplanned downtime. The use of stable intermediates also allows for potential storage between steps if necessary, providing flexibility in production planning. This reliability is critical for meeting the just-in-time delivery requirements of downstream pharmaceutical clients who depend on consistent intermediate flows.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is favorable due to the generation of manageable waste streams. The solvents used, such as acetone and ethyl acetate, are readily recyclable, supporting green chemistry initiatives. The absence of toxic heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with effluent discharge. These factors collectively enhance the scalability of the process, allowing manufacturers to ramp up production from kilogram to tonne scales with confidence in meeting environmental standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its integration into their supply networks. The following questions address common concerns regarding the practical implementation and quality attributes of the process. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of this synthesis route for industrial scaling?
A: The process utilizes readily available commodity chemicals such as chloroacetyl chloride and sodium iodide, avoiding expensive transition metal catalysts. The operational simplicity, involving standard solvent systems like DCM and acetone, facilitates easy scale-up and waste management.
Q: How is purity controlled during the synthesis of the spiro-intermediate?
A: The method employs distinct purification stages, including aqueous quenching and brine washing, which effectively remove inorganic salts and byproducts. The intermediate steps allow for monitoring via TLC or HPLC, ensuring high purity profiles before the final cyclization step.
Q: Is this method suitable for producing large quantities of API intermediates?
A: Yes, the patent explicitly addresses the lack of industrial methods by designing a route with easily controllable reaction conditions. The use of common solvents and standard temperature ranges (room temperature to 120°C) supports commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl-3-oxalidene-1-oxa-4-azaspiro Undecane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis or established catalog items, our infrastructure is designed to support the demanding timelines of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our efficient processes can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence.
