Advanced Enzymatic Synthesis of Chiral 3-Hydroxy-1,2,3,6-Tetrahydropyridine for Commercial Scale-Up
Advanced Enzymatic Synthesis of Chiral 3-Hydroxy-1,2,3,6-Tetrahydropyridine for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral nitrogen-containing heterocycles, which serve as critical scaffolds for bioactive molecules targeting neurological disorders and hyperproliferative diseases. A pivotal advancement in this domain is detailed in Chinese patent CN110577974B, which discloses a highly efficient synthesis method for chiral 3-hydroxy-1,2,3,6-tetrahydropyridine. This technology addresses long-standing challenges regarding low specificity and difficult purification associated with traditional chemical routes by leveraging immobilized lipase kinetic resolution. By operating under mild conditions between 15-30°C, this process not only ensures exceptional stereochemical control but also aligns with modern green chemistry principles, making it an attractive option for reliable pharmaceutical intermediate supplier networks aiming to optimize their API manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral 3-hydroxy-1,2,3,6-tetrahydropyridine has been fraught with significant technical hurdles that impede efficient commercial production. Prior art methods, such as the reduction of boron hydrides controlled by chiral ligands, have demonstrated inadequate specificity, often resulting in isomers with chiral purity as low as 89% ee. Furthermore, these conventional chemical approaches typically necessitate complex separation and purification protocols to isolate the desired enantiomer from the reaction mixture, leading to substantial material loss and increased operational costs. The reliance on stoichiometric chiral auxiliaries or expensive transition metal catalysts further exacerbates the economic burden, while the generation of heavy metal waste poses serious environmental compliance issues for large-scale facilities.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent utilizes a biocatalytic strategy that fundamentally transforms the production landscape for this valuable intermediate. By employing immobilized lipases, specifically types PS or AK, the process achieves a kinetic resolution of the racemic alcohol precursor with remarkable precision. This enzymatic approach selectively acetylates one specific chiral configuration while leaving the other untouched, effectively converting a difficult separation problem into a straightforward physical isolation of two compounds with distinct properties. The result is a dramatic improvement in optical purity, consistently exceeding 99% ee, coupled with a simplified workflow that eliminates the need for harsh reagents and facilitates easier scale-up for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
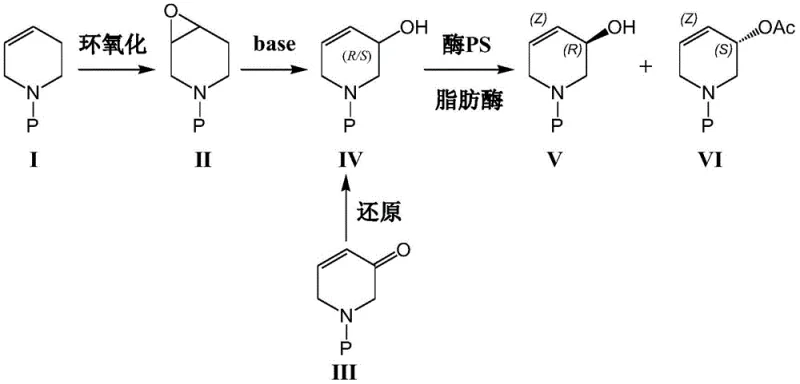
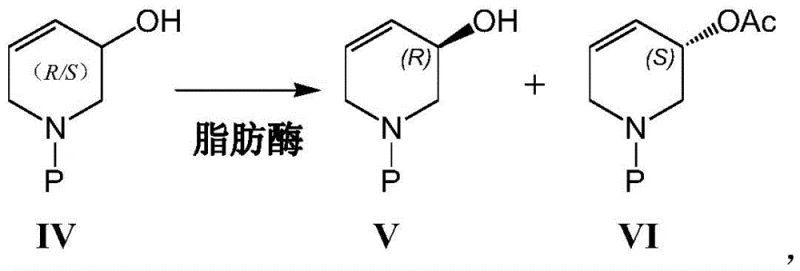
The core of this technological breakthrough lies in the exquisite stereoselectivity of the lipase enzyme towards the hydroxyl group of the tetrahydropyridine ring. The reaction mechanism involves the lipase catalyzing an esterification reaction where vinyl acetate serves as the acyl donor. The enzyme's active site is geometrically constrained to recognize and bind specifically to the alcoholic hydroxyl group of the (S)-configuration enantiomer within the racemic mixture. Consequently, the (S)-enantiomer is selectively acetylated to form the corresponding acetate ester (Formula VI), while the (R)-enantiomer (Formula V) remains unreacted as the free alcohol. This differentiation in chemical functionality allows for the facile separation of the desired chiral alcohol from the byproduct ester through standard extraction or chromatographic techniques, ensuring high purity without the need for complex chiral column chromatography.
Furthermore, the versatility of the precursor synthesis adds another layer of robustness to the overall process, offering two distinct pathways to generate the requisite racemic substrate (Formula IV). Route A involves the reduction of an alpha,beta-unsaturated ketone (Formula III) using sodium borohydride in the presence of cerium chloride, a Luche-type reduction that proceeds efficiently to yield the allylic alcohol. Alternatively, Route B employs an epoxidation-elimination sequence starting from a protected olefin (Formula I). In this pathway, the double bond is first epoxidized to form the epoxy compound (Formula II), which subsequently undergoes hydrolytic ring opening and elimination mediated by a strong base such as lithium diisopropylamide (LDA) or LiHMDS. This dual-pathway capability ensures supply chain resilience, allowing manufacturers to select the most cost-effective starting material based on market availability.
How to Synthesize Chiral 3-Hydroxy-1,2,3,6-Tetrahydropyridine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and enantiomeric excess. The process begins with the preparation of the racemic alcohol, followed by the critical enzymatic resolution step conducted in an organic solvent like toluene. The choice of immobilized enzyme and the ratio of vinyl acetate are key variables; the patent specifies a mass ratio of lipase to substrate of approximately 1:(0.5-0.6) and a vinyl acetate equivalent of 3.0-5.0. Detailed standardized operating procedures regarding temperature control, quenching methods, and workup protocols are essential for reproducibility. For the complete step-by-step technical guide including specific reagent quantities and safety precautions, please refer to the structured synthesis protocol below.
- Prepare the racemic alcohol precursor (Formula IV) either by reducing the alpha,beta-unsaturated ketone (Formula III) with NaBH4/CeCl3 or via epoxidation of the olefin (Formula I) followed by base-mediated elimination.
- Dissolve the racemic compound (Formula IV) in toluene and introduce vinyl acetate along with an immobilized lipase catalyst such as Lipase PS or AK.
- Maintain the reaction mixture at a mild temperature of 15-30°C to facilitate selective acetylation, yielding the desired chiral alcohol (Formula V) and the separable acetate byproduct (Formula VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology presents a compelling value proposition centered on sustainability and operational efficiency. The shift from traditional chemical resolution to biocatalysis eliminates the dependency on scarce chiral ligands and toxic heavy metals, thereby mitigating regulatory risks associated with residual metal limits in final drug substances. Moreover, the ability to recycle the immobilized enzyme and recover solvents creates a closed-loop system that drastically reduces raw material consumption and waste disposal costs. This streamlined approach not only enhances the economic viability of the project but also strengthens the reliability of the supply chain by simplifying the manufacturing process and reducing the potential for batch failures due to complex purification steps.
- Cost Reduction in Manufacturing: The implementation of immobilized lipase catalysis significantly lowers production costs by removing the need for expensive chiral resolving agents and reducing the number of purification cycles. Since the enzyme can be recovered and reused, the recurring cost of the biocatalyst is minimized, while the use of commodity chemicals like vinyl acetate and toluene keeps reagent expenses low. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove impurities, leading to higher overall yields and reduced loss of valuable intermediates during isolation.
- Enhanced Supply Chain Reliability: The synthetic route relies on readily available starting materials such as protected olefins or unsaturated ketones, which are stable and easy to source from multiple global suppliers. This raw material accessibility ensures continuity of supply and protects against market volatility. Furthermore, the mild reaction conditions (15-30°C) reduce the energy demand for heating or cryogenic cooling, making the process less susceptible to utility fluctuations and equipment limitations in diverse manufacturing environments.
- Scalability and Environmental Compliance: Designed with industrial application in mind, this method avoids the use of hazardous reagents and generates minimal waste, aligning perfectly with strict environmental regulations. The simplicity of the workup, involving basic filtration and extraction, facilitates seamless scale-up from laboratory benchtop to multi-ton commercial production. The absence of heavy metal catalysts simplifies the validation process for pharmaceutical applications, accelerating the timeline from process development to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of chiral 3-hydroxy-1,2,3,6-tetrahydropyridine. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the feasibility and advantages of this enzymatic route. Understanding these details is crucial for R&D teams evaluating process transfer and for procurement specialists assessing vendor capabilities.
Q: What advantages does the enzymatic resolution method offer over traditional chiral ligand reduction?
A: Traditional methods using chiral ligands often suffer from low specificity, typically achieving only around 89% ee, and require complex post-reaction purification. In contrast, the enzymatic kinetic resolution described in patent CN110577974B consistently achieves optical purity exceeding 99% ee while utilizing environmentally benign conditions that simplify downstream processing.
Q: Is the lipase catalyst reusable in this industrial process?
A: Yes, the process utilizes immobilized lipases which can be easily separated from the reaction mixture by filtration. This immobilization allows the biocatalyst to be recycled and reused multiple times, significantly reducing catalyst consumption costs and minimizing solid waste generation compared to homogeneous catalytic systems.
Q: What are the primary starting materials for this synthesis route?
A: The synthesis offers flexibility with two viable pathways for generating the racemic precursor. Route A utilizes an alpha,beta-unsaturated ketone (Formula III), while Route B starts from a readily available olefin compound (Formula I). Both starting materials are simple to obtain, ensuring a robust and cost-effective supply chain for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-Hydroxy-1,2,3,6-Tetrahydropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral 3-hydroxy-1,2,3,6-tetrahydropyridine meets the highest standards for enantiomeric excess and chemical purity required by global regulatory agencies.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while optimizing your manufacturing budget.
