Scalable Chemical Resolution of 1-R-3-Fluoropiperidine-4-Carboxylic Acid for High-Purity API Synthesis
Scalable Chemical Resolution of 1-R-3-Fluoropiperidine-4-Carboxylic Acid for High-Purity API Synthesis
The introduction of fluorine atoms into organic molecules has revolutionized modern medicinal chemistry, particularly in the development of antimetabolite agents and nuclear receptor modulators, as evidenced by extensive pharmacological literature. However, the synthesis of optically pure fluorinated building blocks remains a significant bottleneck, often relying on prohibitively expensive chiral separation technologies. Patent CN110922354A discloses a groundbreaking chemical resolution preparation method for 1-R-3-fluoropiperidine-4-carboxylic acid, a critical intermediate for various bioactive compounds. This innovation shifts the paradigm from resource-intensive chiral chromatography to a robust, two-step chemical resolution process involving Mitsunobu esterification and catalytic hydrogenolysis. By leveraging the distinct polarity differences of diastereomeric esters, this method enables the isolation of high-purity enantiomers under mild, neutral conditions that preserve the integrity of sensitive chiral centers. For R&D directors and procurement specialists, this represents a pivotal advancement in securing reliable supply chains for complex fluorinated pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the structural resolution of optically pure cis-1-R-3-fluoropiperidine-4-carboxylic acid enantiomers has relied heavily on chiral column chromatography. While effective on a laboratory scale, this technique presents severe economic and logistical barriers for industrial application. The process consumes vast quantities of specialized mobile phases, driving up operational expenditures significantly. Furthermore, chiral columns have limited loading capacities, meaning that producing kilogram or ton quantities requires excessive time and repetitive cycling, leading to long lead times and high product costs. This inefficiency creates a supply chain bottleneck, restricting the availability of these crucial fluorinated scaffolds for drug development programs. Additionally, the harsh conditions sometimes required for elution can risk racemization or degradation of the fluorine-containing stereocenters, compromising the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a clever chemical resolution strategy that bypasses the need for chiral stationary phases entirely. The method involves derivatizing the racemic mixture with a chiral auxiliary, specifically chiral phenethyl alcohol, to form a pair of diastereomers. Unlike enantiomers, these diastereomers possess different physical properties, such as polarity, allowing them to be separated using conventional, low-cost silica gel chromatography. Following separation, the chiral auxiliary is cleanly removed via catalytic hydrogenation, regenerating the free carboxylic acid without disturbing the stereochemistry. 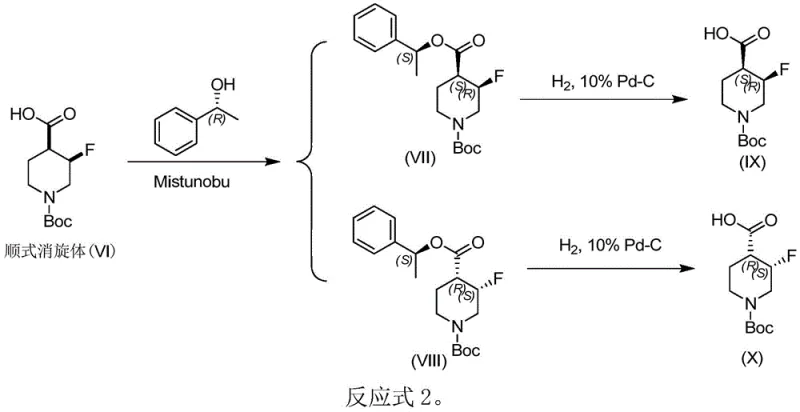 This streamlined workflow not only drastically reduces solvent consumption and waste but also simplifies the purification process, making it highly amenable to commercial scale-up. The ability to obtain both enantiomers simultaneously further enhances the economic viability of the process for diverse synthetic applications.
This streamlined workflow not only drastically reduces solvent consumption and waste but also simplifies the purification process, making it highly amenable to commercial scale-up. The ability to obtain both enantiomers simultaneously further enhances the economic viability of the process for diverse synthetic applications.
Mechanistic Insights into Mitsunobu Esterification and Hydrogenolysis
The core of this resolution technology lies in the precise execution of the Mitsunobu reaction, followed by a selective hydrogenolysis step. In the first stage, the cis-racemic carboxylic acid reacts with a chiral alcohol, such as (R)- or (S)-1-phenethyl alcohol, in the presence of triphenylphosphine and an azodicarboxylate reagent like DEAD or DIAD. This reaction proceeds through the formation of a betaine intermediate, facilitating the nucleophilic attack of the carboxylate on the activated alcohol. Crucially, this esterification creates diastereomeric pairs—specifically the (3R,4S)-ester and the (3S,4R)-ester when using a specific chiral alcohol. The steric bulk of the phenethyl group combined with the fluorine substituent induces sufficient conformational differences, resulting in distinct retention times on silica gel. 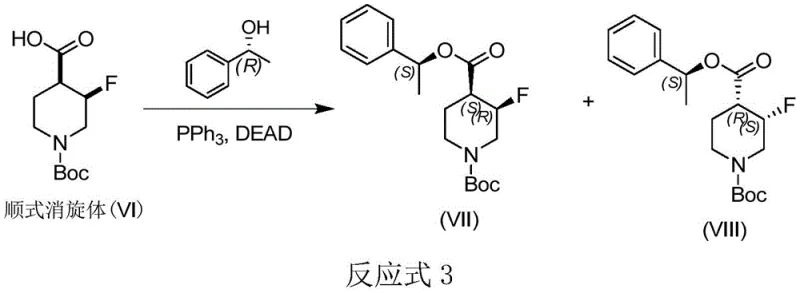 This polarity divergence is the key to successful separation, allowing for the isolation of single isomers with high diastereomeric excess using standard flash chromatography techniques.
This polarity divergence is the key to successful separation, allowing for the isolation of single isomers with high diastereomeric excess using standard flash chromatography techniques.
Following the separation of the diastereomeric esters, the second critical mechanistic step is the removal of the chiral auxiliary. This is achieved through catalytic hydrogenolysis using palladium on carbon (Pd/C) under mild hydrogen pressure, typically between 1 to 5 atmospheres. The benzyl-like C-O bond in the phenethyl ester is susceptible to hydrogenolytic cleavage, while the robust C-F bond and the carbamate protecting group (if present, such as Boc) remain intact under these neutral conditions. This selectivity is paramount; harsh acidic or basic hydrolysis conditions often used for ester cleavage could induce epimerization at the alpha-carbon or defluorination. By employing hydrogenolysis, the process ensures that the absolute configuration established during the resolution is preserved, yielding the target 1-R-3-fluoropiperidine-4-carboxylic acid with optical purity exceeding 95% ee, as confirmed by single-crystal X-ray diffraction of derivatives.
How to Synthesize 1-Boc-3-fluoropiperidine-4-carboxylic acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-purity fluorinated piperidine intermediates suitable for GMP manufacturing. The process begins with the activation of the racemic acid using standard Mitsunobu reagents in anhydrous tetrahydrofuran, followed by careful temperature control to manage exotherms. After the formation of the diastereomeric esters, the mixture is subjected to silica gel column chromatography using a hexane/ethyl acetate gradient to isolate the individual isomers. The final deprotection step utilizes wet Pd/C catalyst under a hydrogen atmosphere, a operation that is straightforward to perform in standard hydrogenation reactors. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform Mitsunobu esterification on cis-racemic 1-Boc-3-fluoropiperidine-4-carboxylic acid using chiral phenethyl alcohol, triphenylphosphine, and DEAD.
- Separate the resulting diastereomeric esters via standard silica gel column chromatography to isolate single isomers.
- Conduct catalytic hydrogenolysis using Pd/C under mild hydrogen pressure to remove the chiral auxiliary and yield the pure acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chiral chromatography to this chemical resolution method offers profound strategic benefits. The primary advantage is the drastic reduction in manufacturing costs associated with the elimination of expensive chiral columns and the massive volumes of HPLC-grade solvents they require. By switching to standard silica gel purification, the variable costs per kilogram of product are significantly lowered, improving margin potential for downstream API production. Furthermore, the reagents utilized—triphenylphosphine, azodicarboxylates, and chiral phenethyl alcohols—are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base ensures greater resilience against market fluctuations and raw material shortages, securing a stable flow of critical intermediates for long-term drug development projects.
- Cost Reduction in Manufacturing: The replacement of preparative chiral HPLC with standard flash chromatography represents a fundamental shift in cost structure. Chiral columns are capital-intensive assets with limited lifespans, whereas silica gel is inexpensive and disposable. Additionally, the solvent recovery rates for normal phase chromatography are generally higher and less energy-intensive than the complex solvent systems needed for chiral separations. This operational efficiency translates directly into lower cost of goods sold (COGS), allowing pharmaceutical companies to allocate resources more effectively across their R&D pipelines without compromising on the quality of the fluorinated building blocks.
- Enhanced Supply Chain Reliability: The robustness of this chemical resolution process enhances supply chain reliability by simplifying the production workflow. The reaction conditions are mild, operating between 0°C and 50°C, which reduces the need for specialized cryogenic or high-temperature equipment. This flexibility allows the process to be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without extensive re-validation. Moreover, the ability to produce both enantiomers from the same racemic starting material maximizes atom economy and ensures that demand for either the (3R,4S) or (3S,4R) isomer can be met flexibly, preventing inventory bottlenecks.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles. The hydrogenolysis step produces benign byproducts (primarily the chiral alcohol which can potentially be recycled), and the overall process avoids the generation of heavy metal waste often associated with asymmetric catalysis using transition metals. The simplicity of the workup procedures—filtration and concentration—facilitates scaling from pilot plant batches to multi-ton commercial production. This scalability ensures that as a drug candidate progresses from clinical trials to market launch, the supply of the fluorinated intermediate can be ramped up seamlessly to meet increasing commercial demand without requiring a complete process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this synthetic route for your specific project needs.
Q: Why is this chemical resolution method superior to chiral column chromatography?
A: Chiral column chromatography consumes large amounts of expensive mobile phases and has limited loading capacity, making it cost-prohibitive for mass production. This chemical resolution method uses standard silica gel chromatography on diastereomers, which is significantly cheaper and easier to scale industrially.
Q: What represents the key technical breakthrough in this patent (CN110922354A)?
A: The breakthrough lies in the selection of chiral phenethyl alcohol as a derivatization fragment. It creates diastereomers with distinct polarity differences for easy separation and can be cleanly removed via simple hydrogenation without affecting the sensitive fluorine-containing chiral centers.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates under mild conditions (0-50°C, 1-5 atm hydrogen pressure) using readily available reagents like triphenylphosphine and Pd/C. The elimination of complex chiral HPLC steps drastically reduces production time and equipment costs, facilitating ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-R-3-fluoropiperidine-4-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from early-stage discovery through to full-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 1-R-3-fluoropiperidine-4-carboxylic acid meets the highest standards of optical purity and chemical integrity. Our commitment to process excellence means we can leverage innovations like the chemical resolution method described in CN110922354A to deliver cost-effective solutions tailored to your specific timeline and budget requirements.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for fluorinated piperidine derivatives. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this resolution method for your specific API. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering complex pharmaceutical intermediates with speed, precision, and reliability.
