Scalable Asymmetric Synthesis of Alpha-Alkyl-Isoserine Intermediates for Pharmaceutical Applications
Scalable Asymmetric Synthesis of Alpha-Alkyl-Isoserine Intermediates for Pharmaceutical Applications
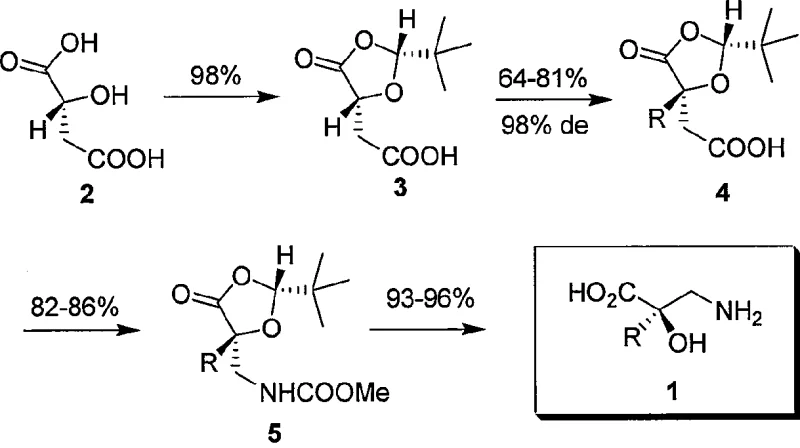
The development of efficient synthetic routes for non-proteinogenic amino acids remains a critical challenge in modern medicinal chemistry, particularly for scaffolds found in potent bioactive molecules like Taxol and Bestatin. Patent CN100473642C introduces a groundbreaking four-step methodology for the high-yield and high-selectivity synthesis of alpha-alkyl-isoserine enantiomers. This technical insight report analyzes the proprietary process which leverages chiral malic acid as a renewable, cost-effective starting material to construct complex beta-amino acid architectures. By integrating Seebach’s “Self-Regeneration of Stereocenters” (SRS) principle with an optimized Curtius rearrangement, the technology delivers optical purities exceeding 98% ee while maintaining a streamlined operational profile suitable for industrial adoption.
For procurement leaders and supply chain directors seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant opportunity for cost reduction in API manufacturing. The route eliminates the need for expensive transition metal catalysts often required in asymmetric dihydroxylation protocols, replacing them with stoichiometric reagents that are readily available in bulk quantities. Furthermore, the robustness of the 1,3-dioxolan-4-one protecting group ensures that the chiral information encoded in the starting malic acid is faithfully transferred to the final product, minimizing the risk of racemization during scale-up and reducing the burden on downstream purification processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of alpha-alkyl-substituted isoserines has been plagued by low yields, poor stereocontrol, and hazardous reaction conditions. Prior art, such as the methods reported by the Avenoza research group, typically relies on N-methoxy-N-methyl-methacrylamide as a starting substrate. This approach necessitates an initial asymmetric dihydroxylation step, which often requires costly chiral ligands and osmium catalysts, followed by a risky nucleophilic ring-opening of cyclic sulfites with sodium azide. These early-stage azide manipulations pose significant safety concerns for large-scale operations due to the potential formation of explosive intermediates. Additionally, the multi-step sequence involved in converting these acyclic precursors into the final amino acid often results in cumulative yield losses and complex impurity profiles that are difficult to purge, ultimately driving up the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN100473642C utilizes a convergent strategy rooted in the chirality pool concept. By starting with commercially available chiral malic acid, the process bypasses the need for external chiral induction entirely. The core innovation lies in the formation of a rigid 1,3-dioxolan-4-one ring system via condensation with pivalaldehyde. This cyclic acetal not only protects the sensitive hydroxyl and carboxylic acid functionalities but also locks the molecular conformation, directing subsequent electrophilic attacks with exceptional facial selectivity. The subsequent conversion of the carboxylic acid moiety into an amine via a modified Curtius rearrangement allows for the late-stage introduction of nitrogen, avoiding the handling of energetic azido-species until the very end of the synthesis where they are immediately trapped as stable carbamates. This strategic reordering of synthetic steps results in a cleaner reaction profile and significantly higher overall throughput.
Mechanistic Insights into Seebach SRS Protection and Curtius Rearrangement
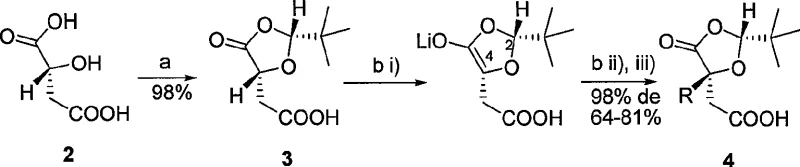
The cornerstone of this synthesis is the rigorous application of Seebach’s “Self-Regeneration of Stereocenters” (SRS) principle during the protection and alkylation phases. As illustrated in the reaction scheme below, the condensation of chiral malic acid with pivalaldehyde under acidic conditions generates a 2-tert-butyl-1,3-dioxolan-4-one derivative with high diastereoselectivity (98% de). This cyclic structure creates a steric environment that differentiates the two faces of the enolate generated in the next step. When treated with strong bases such as n-butyllithium or lithium hexamethyldisilazide (LHMDS) at cryogenic temperatures (-78°C), the resulting enolate is configurationally stable. Subsequent addition of alkyl halides (RX) proceeds via an SN2 mechanism where the bulky tert-butyl group effectively shields one face of the molecule, forcing the electrophile to attack from the less hindered side. This geometric constraint ensures that the newly formed quaternary or tertiary stereocenter is established with near-perfect fidelity, preserving the optical integrity of the starting material.
Following the establishment of the carbon skeleton, the transformation of the carboxylic acid into the amino functionality is achieved through a highly optimized Curtius rearrangement. Traditional Curtius protocols often suffer from the isolation of unstable acyl azides, which can be hazardous and lead to variable yields. The patented process circumvents this by generating the acyl azide in situ using diphenylphosphoryl azide (DPPA) and triethylamine in anhydrous toluene. Upon heating, the acyl azide undergoes rearrangement to an isocyanate intermediate, which is immediately trapped by an alcohol nucleophile (such as methanol or benzyl alcohol) to form a stable carbamate. Experimental data indicates that performing the rearrangement in toluene prior to the addition of the alcohol trap significantly improves yields compared to direct reflux in alcohol. This modification prevents competitive side reactions and ensures that the reactive isocyanate species is efficiently converted to the desired protected amino acid derivative, setting the stage for the final global deprotection.

How to Synthesize Alpha-Alkyl-Isoserine Efficiently
The execution of this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to maintain the high stereochemical standards defined in the patent. The process begins with the activation of the malic acid derivative, followed by the critical alkylation step which defines the side-chain identity (benzyl, ethyl, allyl, or methyl). Following the carbon-carbon bond formation, the Curtius rearrangement serves as the pivotal functional group interconversion. For R&D teams looking to implement this technology, it is crucial to note that the choice of alcohol trap in the rearrangement step influences the ease of final deprotection; methyl carbamates are generally preferred for their straightforward hydrolysis profiles compared to benzyl analogues which require hydrogenolysis. The detailed standardized synthesis steps see the guide below.
- Diastereoselective protection of chiral malic acid with pivalaldehyde to form a 1,3-dioxolan-4-one derivative.
- Stereoselective alkylation of the protected intermediate using strong bases like n-BuLi or LHMDS at low temperatures.
- Conversion of the carboxylic acid to a carbamate via Curtius rearrangement using DPPA and an alcohol trap.
- Final deprotection of the acetal and carbamate groups using acidic hydrolysis to yield the free alpha-alkyl-isoserine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The reliance on chiral malic acid, a fermentation-derived commodity chemical, decouples the supply chain from the volatility associated with specialized chiral catalysts or resolution agents. This shift to a chirality-pool strategy inherently stabilizes raw material costs and ensures long-term supply continuity, as malic acid is produced on a massive global scale for the food and beverage industry. Furthermore, the elimination of heavy metal catalysts (such as osmium or palladium in early steps) simplifies the regulatory dossier for the final drug substance, as residual metal testing becomes less burdensome. The process operates in common organic solvents like toluene, THF, and ethyl acetate, which are easily recovered and recycled, contributing to a greener manufacturing footprint and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive chiral ligands and transition metal catalysts with stoichiometric reagents derived from bulk chemicals. The high overall yield (52-65%) across four steps minimizes material loss, while the use of crystalline intermediates facilitates purification via simple recrystallization rather than expensive preparative chromatography. Additionally, the avoidance of cryogenic conditions for extended periods (only required briefly for enolate formation) reduces energy consumption compared to prolonged low-temperature protocols found in alternative methods.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are available from multiple global suppliers, the risk of single-source bottlenecks is drastically mitigated. The robustness of the 1,3-dioxolan-4-one intermediate allows for potential stockpiling or telescoping of steps, providing flexibility in production scheduling. The chemistry is insensitive to minor fluctuations in moisture or oxygen when standard inert atmosphere techniques are applied, ensuring consistent batch-to-batch quality and reducing the rate of failed batches due to environmental factors.
- Scalability and Environmental Compliance: The reaction conditions are amenable to large-scale reactor operations, with exotherms that are manageable through standard cooling jackets. The waste stream primarily consists of benign salts and recoverable organic solvents, avoiding the generation of toxic heavy metal waste or persistent organic pollutants. This alignment with green chemistry principles simplifies environmental permitting and reduces the liability associated with hazardous waste treatment, making the process highly attractive for contract development and manufacturing organizations (CDMOs) aiming to expand their capacity for complex amino acid derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-alkyl-isoserine synthesis platform. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on scalability, purity specifications, and operational safety. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into existing production lines.
Q: What is the stereochemical purity of the synthesized alpha-alkyl-isoserine?
A: The process described in patent CN100473642C achieves exceptional stereocontrol, consistently delivering products with 98% enantiomeric excess (ee) and 98% diastereomeric excess (de) through the application of Seebach's Self-Regeneration of Stereocenters (SRS) principle.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the route utilizes robust, commodity-grade starting materials like chiral malic acid and standard solvents such as toluene and THF. The intermediates are crystalline and stable, facilitating safe scale-up from laboratory to multi-ton commercial manufacturing without requiring exotic catalysts.
Q: What are the key advantages over conventional methacrylamide-based routes?
A: Unlike traditional methods that rely on complex asymmetric dihydroxylation and hazardous azide openings early in the sequence, this malic acid-derived route postpones nitrogen introduction until the Curtius rearrangement step. This simplifies purification, improves overall yield (52-65%), and enhances operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Alkyl-Isoserine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of robust synthetic pathways like the one described in CN100473642C for the production of high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our facility is equipped to handle the specific solvent systems and thermal requirements of the Curtius rearrangement and alkylation steps safely and effectively.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to validate the route feasibility for a new drug candidate, we are ready to provide specific COA data and route feasibility assessments. Contact us today to secure a reliable supply of high-purity alpha-alkyl-isoserine derivatives and accelerate your drug development timeline with a partner dedicated to quality and innovation.
