Advanced Rhodium-Catalyzed Synthesis of Axial Chiral Halogenated Biaryl Compounds for Commercial Scale-up
Advanced Rhodium-Catalyzed Synthesis of Axial Chiral Halogenated Biaryl Compounds for Commercial Scale-up
The landscape of asymmetric synthesis is undergoing a significant transformation with the introduction of novel catalytic methodologies that address long-standing inefficiencies in producing chiral building blocks. Patent CN114957103A discloses a groundbreaking preparation method for axial chiral halogenated biaryl compounds, utilizing a highly efficient rhodium-catalyzed asymmetric C-H activation strategy. This technology represents a pivotal advancement for the fine chemical industry, particularly for the production of complex pharmaceutical intermediates and agrochemical active ingredients where stereochemical purity is paramount. By leveraging a specialized chiral cyclopentadienyl rhodium catalyst system in conjunction with silver and copper salts, this process achieves exceptional enantioselectivity and reaction yields that far surpass conventional approaches. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic robustness and scalability of this Rh-catalyzed pathway is essential for securing a competitive edge in the supply of high-value chiral molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral halogenated biaryl scaffolds has relied heavily on palladium-catalyzed kinetic resolution strategies or stoichiometric chiral auxiliaries, both of which present substantial bottlenecks for industrial application. As noted in the background art of the patent, previous methods, such as those reported by Gao et al. in 2014, utilized chiral amino acid ligands with palladium to achieve asymmetric C-H iodination. However, these legacy processes are fundamentally constrained by the nature of kinetic resolution, which theoretically limits the maximum yield to 50% unless dynamic kinetic resolution is employed, often resulting in poor atom economy and increased waste generation. Furthermore, these older protocols frequently exhibit modest enantioselective control, with ee values hovering around 94% or lower, necessitating costly and time-consuming recrystallization steps to meet the stringent purity specifications required for API manufacturing. The reliance on specific substrate pre-functionalization and the limited scope of tolerated functional groups further restrict the versatility of these conventional methods, making them less ideal for the diverse library synthesis needed in modern drug discovery.
The Novel Approach
In stark contrast, the methodology described in CN114957103A introduces a direct asymmetric C-H activation pathway that circumvents the inherent yield limitations of kinetic resolution. By employing a chiral rhodium catalyst, specifically trivalent rhodium complexes bearing planar chiral cyclopentadienyl ligands, the process enables the direct installation of halogen atoms onto the biaryl backbone with remarkable precision. This novel approach not only boosts reaction yields to near-quantitative levels (up to 99% in optimized examples) but also consistently delivers enantiomeric excess values exceeding 94%, with many substrates achieving greater than 97% ee. The reaction conditions are notably mild, typically proceeding at temperatures between 40°C and 80°C in common organic solvents like acetonitrile, which significantly reduces energy consumption compared to high-temperature alternatives. This shift from resolution-based strategies to direct asymmetric synthesis represents a paradigm shift in cost reduction in fine chemical manufacturing, offering a streamlined route that minimizes raw material usage and maximizes throughput for high-purity axial chiral compounds.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric C-H Halogenation
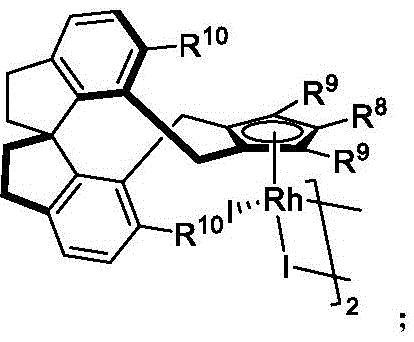
The core of this technological breakthrough lies in the sophisticated design of the chiral rhodium catalyst system, which orchestrates the stereochemical outcome of the C-H bond cleavage and subsequent halogenation. The catalytic cycle initiates with the coordination of the biaryl substrate to the cationic rhodium(III) center, facilitated by the directing group inherent in the substrate structure, such as the isoquinoline or pyridine moiety. The chiral environment provided by the pentamethylcyclopentadienyl (Cp*) ligand, modified with specific chiral linkers (as seen in catalysts C1 through C32), creates a sterically demanding pocket that differentiates between the two prochiral faces of the biaryl axis. This differentiation is critical during the C-H metallation step, where the rhodium center selectively activates one specific C-H bond over the other, thereby establishing the axial chirality at the earliest stage of the reaction. The presence of silver salts, such as AgNO3 or AgSbF6, serves a dual purpose: acting as a halide scavenger to generate the active cationic rhodium species and potentially participating in the oxidation state management of the metal center.
Following the C-H activation, the halogenating agent, typically an N-halosuccinimide (like NIS or NBS), interacts with the organorhodium intermediate to effect the transfer of the halogen atom. The copper salt, preferably Cu(OAc)2, acts as a crucial co-catalyst or oxidant, facilitating the turnover of the catalytic cycle and ensuring the regeneration of the active Rh(III) species. This synergistic interaction between the rhodium catalyst, silver additive, and copper salt is what enables the high turnover numbers and sustained enantioselectivity observed across a broad range of substrates. The mechanism effectively suppresses racemic background reactions, ensuring that the final product retains the high optical purity dictated by the chiral ligand. For technical teams evaluating process feasibility, this mechanistic robustness implies a high tolerance for variation in raw material quality and a reduced risk of forming difficult-to-remove diastereomeric impurities, thereby simplifying downstream purification and enhancing the overall reliability of the synthetic route for commercial scale-up of complex polymer additives or pharmaceutical intermediates.
How to Synthesize Axial Chiral Halobiaryl Compound Efficiently
The practical implementation of this synthesis protocol is designed for operational simplicity while maintaining rigorous control over stereochemical outcomes. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically argon, to prevent catalyst deactivation by oxygen or moisture. The substrate, halogenating agent, and the specific chiral rhodium catalyst are combined in a nitrile solvent, with the concentration carefully adjusted to optimize reaction kinetics and heat transfer. Detailed standardized operating procedures for the precise addition of reagents, temperature ramping, and workup protocols are critical for reproducing the high yields and ee values reported in the patent data. The following guide outlines the essential steps for executing this transformation effectively in a laboratory or pilot plant setting.
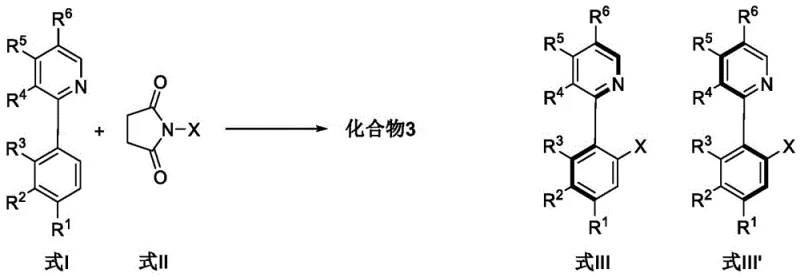
- Prepare the reaction mixture by combining the biaryl substrate (Formula I) and the halogenating agent (Formula II) in an organic solvent such as acetonitrile under an inert atmosphere.
- Add the chiral rhodium catalyst (e.g., Cp*Rh complex), silver salt (e.g., AgNO3), and copper salt (e.g., Cu(OAc)2) to the mixture in precise molar ratios.
- Heat the reaction to 40-80°C for 2-12 hours, then quench with water, extract with dichloromethane, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology offers tangible strategic benefits that extend beyond mere technical performance. The primary advantage lies in the significant streamlining of the supply chain for chiral intermediates. By utilizing a direct C-H activation strategy, the process eliminates the need for multiple pre-functionalization steps often required in traditional cross-coupling routes, thereby reducing the number of unit operations and the associated inventory holding costs. The use of readily available starting materials, such as substituted isoquinolines and naphthalenes, ensures a stable and secure supply base, mitigating the risks associated with sourcing exotic or proprietary precursors. Furthermore, the high atom economy of the reaction means that less raw material is wasted, directly contributing to cost reduction in manufacturing without compromising on the quality of the final output.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the high catalytic efficiency of the rhodium system drastically reduce the material cost per kilogram of product. Unlike kinetic resolution methods that discard half of the starting material, this asymmetric synthesis converts the majority of the substrate into the desired enantiomer, leading to substantial savings in raw material expenditure. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedure lowers solvent usage and waste disposal costs, creating a more economically sustainable production model.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against variations in reaction parameters ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream customers. The broad substrate scope allows for the flexible production of various analogues using the same core technology platform, enabling rapid response to changing market demands or custom synthesis requests. This flexibility reduces the lead time for high-purity axial chiral compounds, allowing manufacturers to accelerate their own development timelines and bring products to market faster.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with standard laboratory equipment and showing clear potential for translation to multi-kilogram or ton-scale reactors. The use of common solvents like acetonitrile and the absence of highly toxic reagents simplify compliance with environmental regulations and safety standards. The high selectivity of the reaction minimizes the formation of byproducts, reducing the burden on wastewater treatment facilities and aligning with green chemistry principles, which is increasingly important for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this axial chiral halogenation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the process capabilities and limitations for potential partners and licensees.
Q: What are the advantages of this Rh-catalyzed method over traditional Pd-catalyzed kinetic resolution?
A: Unlike traditional Pd-catalyzed methods which often suffer from low yields (max 50% theoretical for kinetic resolution) and moderate enantioselectivity, this Rh-catalyzed asymmetric C-H activation achieves yields up to 99% and ee values exceeding 97%, offering a much more efficient route to single-enantiomer products.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes readily available starting materials, operates at mild temperatures (40-80°C), and uses common solvents like acetonitrile, making it highly amenable to commercial scale-up without requiring exotic reagents or extreme conditions.
Q: What types of halogenated compounds can be synthesized using this technology?
A: The method demonstrates broad substrate universality, successfully synthesizing axial chiral iodo-, bromo-, and chloro-biaryl compounds with various substituents including alkyl, alkoxy, and fused ring systems like naphthyl and phenanthryl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Halogenated Biaryl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to drive innovation in the pharmaceutical and agrochemical sectors. Our team of expert chemists has thoroughly evaluated the rhodium-catalyzed C-H activation process described in CN114957103A and is fully equipped to translate this laboratory-scale breakthrough into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yield observed in small-scale experiments are maintained during large-scale manufacturing. Our state-of-the-art facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of axial chiral intermediate meets the exacting standards required for global regulatory submissions.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of specific chiral ligands or bulk production of halogenated biaryl intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-performance chiral building blocks for your most demanding applications.
