Scalable Production of 4-Bromo-2-Methoxybenzaldehyde via Optimized Metal-Halogen Exchange
Introduction to Advanced Intermediate Synthesis
The chemical industry constantly seeks robust methodologies for producing high-value building blocks, and patent CN103025696A presents a transformative approach for the synthesis of 4-bromo-2-methoxybenzaldehyde, a critical scaffold in medicinal chemistry. This intellectual property outlines a streamlined two-step sequence that begins with the selective metal-halogen exchange of 1,4-dibromo-2-fluorobenzene, effectively bypassing the severe cryogenic constraints associated with traditional lithiation protocols. By shifting the reaction temperature window to a manageable 0°C to 5°C range, the process not only enhances operational safety but also drastically simplifies the engineering requirements for commercial reactors. Furthermore, the strategic selection of potassium carbonate for the subsequent nucleophilic aromatic substitution step elegantly mitigates the formation of Cannizzaro byproducts, a persistent impurity challenge in aldehyde functionalization. For R&D directors and procurement strategists alike, this methodology represents a significant leap forward in process intensification, offering a reliable pathway to secure high-purity intermediates essential for complex API manufacturing.
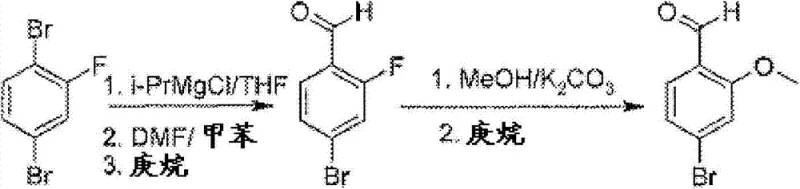
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 4-bromo-2-methoxybenzaldehyde has been plagued by significant technical hurdles that compromise both yield and scalability in an industrial setting. Prior art methods often relied on the formylation of m-bromoanisole, a route notorious for its poor regioselectivity which necessitates difficult and costly purification steps to isolate the desired isomer. Alternatively, existing literature describes routes utilizing butyllithium in diethyl ether at cryogenic temperatures of -78°C, creating substantial safety liabilities due to the pyrophoric nature of the reagent and the high energy consumption required for cooling. Another reported approach involving tributylmagnesate complexes requires the in-situ preparation of reactive species using two equivalents of butyllithium, adding unnecessary complexity and cost to the supply chain. Moreover, previous attempts at the methoxy substitution using sodium methoxide frequently resulted in moderate yields around 38% due to competitive Cannizzaro reactions that degrade the valuable aldehyde functionality. These cumulative inefficiencies render conventional methods unsuitable for the rigorous demands of modern cost reduction in pharma manufacturing.
The Novel Approach
The innovative process disclosed in the patent data fundamentally re-engineers the synthetic route by leveraging the superior selectivity of isopropylmagnesium chloride for metal-halogen exchange. This reagent allows the critical exchange reaction to proceed efficiently at 0°C, eliminating the need for expensive cryogenic infrastructure while maintaining high chemoselectivity between the bromine and fluorine substituents. Following the formylation with DMF, the intermediate 2-fluoro-4-bromobenzaldehyde is purified via crystallization from heptane, a solvent choice that aligns perfectly with green chemistry principles and facilitates easy recovery. The subsequent substitution step utilizes potassium carbonate in methanol at a mild 50°C, a condition specifically optimized to drive the SNAr reaction while suppressing base-catalyzed aldehyde degradation. This optimized sequence delivers an impressive overall yield of 57%, demonstrating a clear advantage over the fragmented and low-yielding processes of the past. By integrating these improvements, the novel approach establishes a new benchmark for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Selective Metal-Halogen Exchange and SNAr
The core of this technological breakthrough lies in the precise control of organometallic reactivity, specifically the preferential exchange of the bromine atom over the fluorine atom on the aromatic ring. Isopropylmagnesium chloride acts as a highly effective magnesium-halogen exchange reagent that targets the carbon-bromine bond due to its lower bond dissociation energy compared to the carbon-fluorine bond. This selectivity is crucial because it preserves the fluorine handle necessary for the subsequent nucleophilic aromatic substitution, ensuring that the molecular architecture remains intact for the final transformation. The reaction is conducted in a mixture of THF and toluene, a solvent system that stabilizes the Grignard species while providing a suitable medium for the subsequent quenching and workup procedures. By maintaining the temperature strictly between 0°C and 5°C, the process minimizes potential side reactions such as Wurtz-type couplings or over-metalation, which could otherwise lead to intractable impurity profiles. This controlled environment ensures that the resulting aryl magnesium species is generated with high fidelity, ready for immediate trapping with the formyl source.
Following the formation of the aldehyde intermediate, the mechanism shifts to a nucleophilic aromatic substitution where the fluoride leaving group is displaced by a methoxide nucleophile. A critical mechanistic insight provided by this patent is the avoidance of strong alkoxide bases like sodium methoxide, which are known to induce Cannizzaro disproportionation of the aldehyde group. Instead, the use of potassium carbonate provides a sufficiently basic environment to generate the active methoxide species in situ from methanol, yet remains mild enough to prevent the attack on the carbonyl carbon of the aldehyde. This subtle balance of basicity is what allows the reaction to proceed to completion with minimal formation of alcohol or carboxylic acid byproducts, as illustrated in the comparative reaction pathways below. The result is a cleaner reaction profile that significantly reduces the burden on downstream purification units, directly impacting the cost of goods sold.
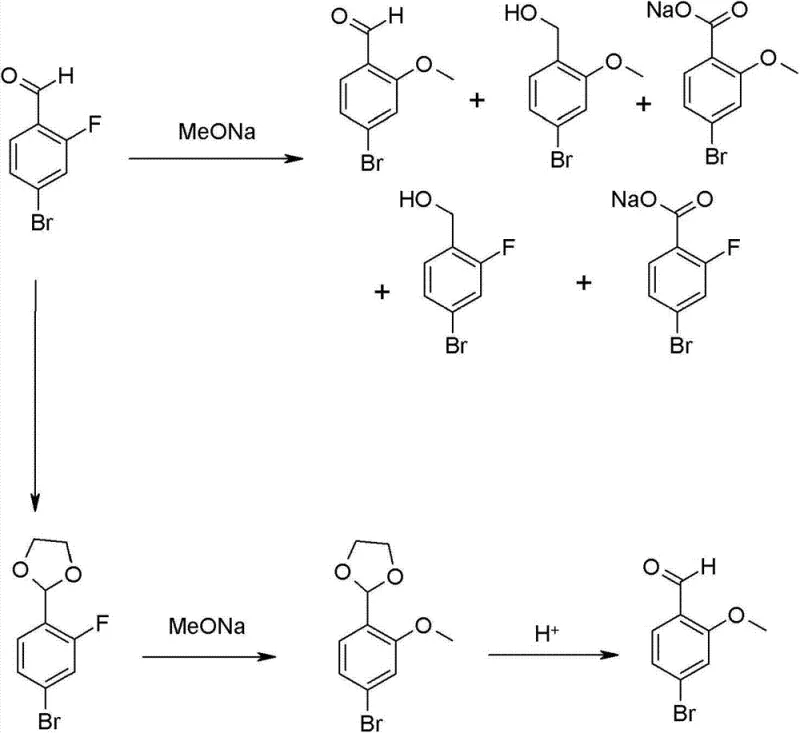
How to Synthesize 4-Bromo-2-Methoxybenzaldehyde Efficiently
Implementing this synthesis requires strict adherence to the specified thermal and stoichiometric parameters to maximize the yield of the intermediate and final product. The process begins with the careful addition of the dibromo-fluoro starting material to the Grignard reagent under inert atmosphere, followed by the introduction of DMF to install the aldehyde functionality. After aqueous workup and crystallization, the solid intermediate is subjected to the substitution conditions using methanol and potassium carbonate with controlled heating. For a comprehensive understanding of the exact operational parameters, the detailed standardized synthesis steps are outlined in the guide below.
- Perform metal-halogen exchange on 1,4-dibromo-2-fluorobenzene using isopropylmagnesium chloride at 0°C, followed by formylation with DMF.
- Crystallize the intermediate 2-fluoro-4-bromobenzaldehyde from heptane to ensure high purity before the next step.
- React the intermediate with methanol and potassium carbonate at 50°C to effect SNAr substitution, minimizing Cannizzaro side reactions.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that extend well beyond simple chemical yield, addressing key pain points in global supply chain management. The elimination of cryogenic requirements and pyrophoric reagents translates directly into reduced capital expenditure for manufacturing facilities, as standard stainless steel reactors can be utilized without specialized low-temperature cooling jackets. Furthermore, the substitution of hazardous solvents like diethyl ether with safer alternatives such as toluene and heptane significantly lowers the risk profile of the operation, leading to lower insurance premiums and simplified regulatory compliance. The robustness of the crystallization steps ensures consistent product quality, reducing the variability that often plagues batch-to-batch production in fine chemical manufacturing. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of multinational pharmaceutical clients.
- Cost Reduction in Manufacturing: The transition from butyllithium to isopropylmagnesium chloride eliminates the need for handling extremely hazardous pyrophoric materials, thereby reducing the specialized safety infrastructure and training costs associated with their use. Additionally, the ability to run the reaction at 0°C rather than -78°C results in substantial energy savings, as the facility does not need to maintain extreme cryogenic conditions for extended periods. The simplified workup procedure, which avoids complex protection and deprotection sequences, further reduces the consumption of auxiliary chemicals and solvent volumes. These operational efficiencies culminate in a significantly lower cost of production, making the final intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: By utilizing commercially available and stable reagents like isopropylmagnesium chloride, the process removes dependencies on custom-synthesized or hard-to-source catalysts that can bottleneck production schedules. The use of common industrial solvents such as toluene and heptane ensures that raw material procurement is straightforward and less susceptible to market volatility compared to specialized ether solvents. Moreover, the high selectivity of the reaction minimizes the generation of difficult-to-remove impurities, ensuring a consistent supply of high-quality material that meets stringent customer specifications without delay. This reliability is critical for maintaining continuous manufacturing lines and avoiding costly production stoppages.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing solvent systems that are easily recovered and recycled through standard distillation units, thereby minimizing waste generation and environmental impact. The avoidance of heavy metal catalysts and the use of benign inorganic bases like potassium carbonate simplify the wastewater treatment process, ensuring compliance with increasingly strict environmental regulations. The crystallization-driven purification strategy is highly scalable and does not rely on chromatography, which is often a bottleneck in large-scale production. Consequently, this route supports the seamless transition from pilot plant trials to multi-ton commercial production with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthetic route for 4-bromo-2-methoxybenzaldehyde. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of adopting this technology within your existing manufacturing framework.
Q: Why is isopropylmagnesium chloride preferred over butyllithium for this synthesis?
A: Isopropylmagnesium chloride allows the reaction to proceed at 0°C rather than the cryogenic -78°C required for butyllithium, significantly reducing energy costs and improving operational safety by avoiding pyrophoric reagents.
Q: How does this process minimize the formation of Cannizzaro byproducts?
A: By utilizing potassium carbonate as the base for the nucleophilic aromatic substitution instead of strong alkoxides like sodium methoxide, the process suppresses the disproportionation of the aldehyde group, leading to higher selectivity.
Q: What solvents are utilized to ensure scalability?
A: The process replaces diethyl ether, which poses explosion hazards on large scale, with safer alternatives like toluene and heptane, facilitating easier solvent recovery and safer commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-2-Methoxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends heavily on the availability of high-quality, cost-effective intermediates like 4-bromo-2-methoxybenzaldehyde. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch conforms to the highest industry standards, providing you with the confidence needed to advance your drug development programs. Our commitment to technical excellence allows us to adapt quickly to changing project needs while maintaining unwavering quality control.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized process can enhance your supply chain efficiency and reduce your overall manufacturing costs.
