Advancing API Intermediate Synthesis: A Robust Nickel-Catalyzed Route to Gamma-Alkenyl Ketones
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic methodologies. A significant breakthrough in this domain is documented in patent CN110776407A, which discloses a novel preparation method for gamma-alkenyl ketones. This technology leverages the abundant and inexpensive feedstock 1,3-butadiene, transforming it into high-value added synthetic building blocks through a highly efficient nickel-catalyzed process. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift away from traditional, waste-generating alkylation methods towards a more atom-economical paradigm. The ability to construct carbon-carbon bonds directly from simple ketones and dienes without stoichiometric by-products addresses critical pain points in modern API synthesis, offering a pathway to reduce both environmental footprint and raw material costs.
Traditionally, the alpha-alkylation of ketones has relied heavily on the use of strong, stoichiometric bases such as LDA or alkali metal hydrides to generate enolates. These conventional approaches often suffer from significant limitations, including poor functional group tolerance, harsh reaction conditions, and the generation of substantial salt waste during quenching. Furthermore, controlling regioselectivity when using unsymmetrical ketones or dienes like 1,3-butadiene can be notoriously difficult, often leading to complex mixtures that require energy-intensive purification steps. The reliance on precious metal catalysts in some alternative transition-metal mediated processes further exacerbates cost concerns, making the final intermediate prohibitively expensive for large-volume production. These inefficiencies create bottlenecks in the supply chain, increasing lead times and complicating the regulatory approval process due to impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The legacy methods for constructing gamma-alkenyl ketone scaffolds typically involve multi-step sequences or the use of pre-functionalized coupling partners, which inherently lower the overall atom economy. The necessity for cryogenic conditions to manage reactive enolates adds significant operational complexity and energy consumption to the manufacturing process. Moreover, the sensitivity of these traditional reagents to moisture and oxygen necessitates specialized equipment and rigorous exclusion protocols, which are not always feasible in standard multipurpose manufacturing plants. The resulting impurity profiles often include regioisomers and over-alkylated by-products, demanding extensive chromatographic purification that drastically reduces yield and throughput. For supply chain managers, these factors translate into higher volatility in production schedules and increased dependency on specialized contract manufacturing organizations capable of handling hazardous reagents.
The Novel Approach
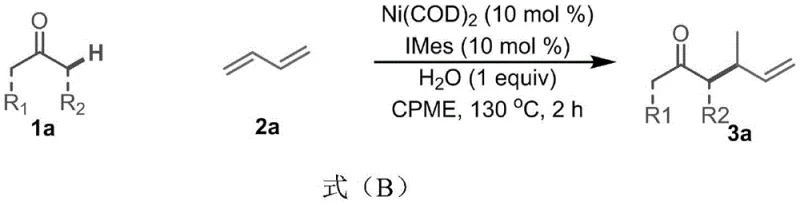
In stark contrast, the methodology described in the patent introduces a streamlined, one-pot transformation that utilizes a nickel(0) catalyst system paired with an N-heterocyclic carbene ligand (IMes). This novel approach enables the direct alpha-butenylation of simple ketones using 1,3-butadiene under remarkably mild and neutral conditions. A defining feature of this chemistry is the use of water or simple alcohols as essential additives, which play a crucial role in protonating the nickel-allyl intermediates and facilitating the catalytic cycle without the need for external strong bases. This shift to neutral pH conditions dramatically expands the substrate scope, allowing for the inclusion of sensitive functional groups that would otherwise decompose under basic conditions. The reaction proceeds with high regioselectivity and excellent yields, as demonstrated by the conversion of acetophenone derivatives to the corresponding gamma-alkenyl ketones in yields reaching up to 82%.
The mechanistic elegance of this transformation lies in the dual activation of both the ketone and the diene by the nickel center. As illustrated in the catalytic cycle, the Ni(0) species coordinates with 1,3-butadiene to form a pi-allyl complex, which is subsequently protonated by the water additive to generate a nickel-hydride species. This active hydride species then facilitates the deprotonation of the ketone to form a nickel-enolate, setting the stage for the critical carbon-carbon bond-forming event. The subsequent reductive elimination releases the desired gamma-alkenyl ketone product and regenerates the active Ni(0) catalyst, ensuring high turnover numbers and minimizing metal waste. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for scale-up, as it highlights the delicate balance between catalyst loading, additive concentration, and temperature required to maintain high efficiency.
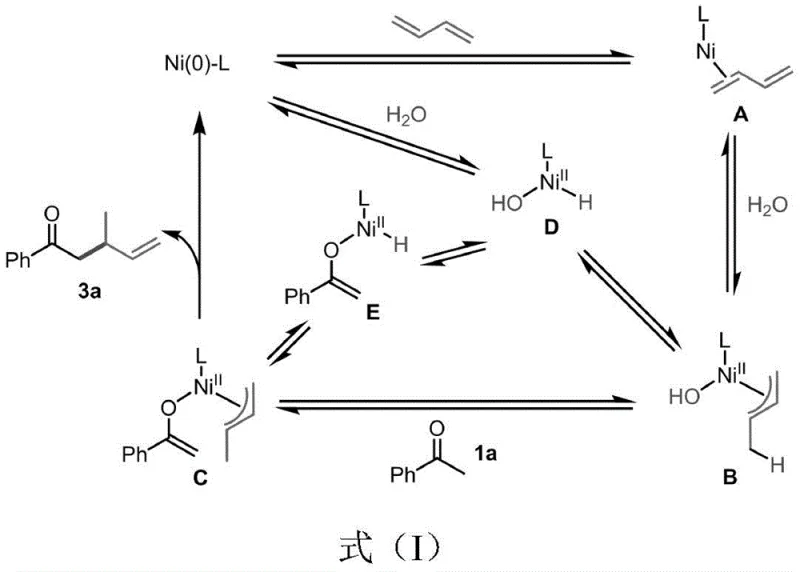
From a commercial perspective, the implications of this mechanistic pathway are profound. The reliance on nickel, a base metal, rather than palladium or rhodium, immediately lowers the raw material cost profile of the catalyst system. Furthermore, the use of 1,3-butadiene, a commodity chemical produced on a scale of over 10 million tons annually for the rubber industry, ensures a stable and inexpensive supply of the carbon source. The elimination of strong bases not only improves safety but also simplifies the downstream processing, as there is no need for extensive aqueous workups to remove inorganic salts. This streamlined workflow directly contributes to cost reduction in pharmaceutical intermediate manufacturing by reducing solvent usage, waste disposal fees, and processing time. For procurement teams, this translates to a more resilient supply chain less susceptible to fluctuations in the price of precious metals or specialized reagents.
Mechanistic Insights into Ni-Catalyzed Alpha-Butenylation
Deep diving into the reaction kinetics reveals that water is not merely a passive solvent component but an active participant in the catalytic turnover. Isotopic labeling experiments using D2O confirm the incorporation of deuterium into the product, validating the proposed protonation steps involving the nickel-hydride species. This insight allows for fine-tuning of the reaction conditions; for instance, maintaining the optimal molar ratio of ketone to butadiene to water (preferably 1:3:1) is critical for maximizing yield and minimizing side reactions such as butadiene dimerization. The robustness of the IMes ligand in stabilizing the nickel center against decomposition under thermal stress (up to 130°C) further underscores the suitability of this system for continuous flow processing or large-batch reactor operations. Such mechanistic clarity provides R&D directors with the confidence to transfer this technology from the laboratory bench to pilot plant scales with predictable outcomes.
How to Synthesize Gamma-Alkenyl Ketone Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized protocol established in the patent examples. The process begins with the preparation of the catalyst solution under an inert atmosphere to prevent oxidation of the Ni(0) species. Following the addition of substrates and the critical water additive, the reaction mixture is heated to promote the insertion and elimination steps. The simplicity of the workup, involving filtration through silica to remove metal residues followed by standard chromatography or crystallization, makes this method particularly attractive for GMP manufacturing. Detailed standardized operating procedures for this synthesis are outlined below to ensure reproducibility and quality control.
- Prepare the catalyst system by mixing Ni(COD)2 (10 mol%) and IMes ligand (10 mol%) in CPME solvent under inert atmosphere.
- Add the ketone substrate (1.0 equiv), 1,3-butadiene (3.0 equiv), and water (1.0 equiv) to the reaction mixture sequentially.
- Heat the reaction mixture to 130°C for 2 hours, then cool, filter to remove metal ions, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this technology offers a compelling value proposition that extends beyond mere chemical novelty. The shift from precious metal catalysis to base metal nickel catalysis represents a strategic move towards cost sustainability. By eliminating the need for expensive palladium or rhodium complexes, manufacturers can significantly reduce the bill of materials without compromising on reaction efficiency. Additionally, the use of commodity-grade 1,3-butadiene ensures that the primary carbon source remains affordable and readily available globally, mitigating risks associated with sourcing specialized fine chemicals. The operational safety improvements derived from avoiding pyrophoric bases also contribute to lower insurance premiums and reduced regulatory burden, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with inexpensive nickel complexes, combined with the use of bulk chemical feedstocks like 1,3-butadiene, drives down the overall cost of goods sold. The neutral reaction conditions eliminate the need for costly anhydrous solvents and specialized drying equipment, while the simplified workup reduces solvent consumption and waste treatment expenses. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: Utilizing 1,3-butadiene, a high-volume petrochemical derivative, insulates the production process from the supply volatility often seen with niche fine chemical reagents. The robustness of the catalyst system allows for flexible sourcing of raw materials, and the tolerance for various ketone substrates means that production lines can be easily adapted to synthesize different intermediates without major retooling. This flexibility ensures consistent delivery schedules and reduces the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The atom-economic nature of this reaction minimizes waste generation, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The absence of stoichiometric salt by-products simplifies effluent treatment, and the use of safer reagents reduces the hazard profile of the manufacturing site. These factors facilitate easier regulatory approval for new drug applications and support the long-term sustainability of the manufacturing operation, making it a preferred choice for environmentally conscious partners.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the implementation and scope of this technology, we have compiled a set of answers based on the technical data provided in the patent documentation. These questions cover critical aspects such as catalyst loading, substrate compatibility, and safety considerations, providing a quick reference for technical teams evaluating this route for their specific projects. Understanding these details is essential for making informed decisions about process adoption and risk assessment.
Q: What are the key advantages of this nickel-catalyzed method over traditional alkylation?
A: This method operates under neutral pH conditions using water as an additive, avoiding hazardous strong bases. It utilizes inexpensive nickel catalysts instead of precious metals and achieves high regioselectivity with commodity chemical 1,3-butadiene.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of stable solvents like CPME, mild reaction temperatures (130°C), and the avoidance of pyrophoric reagents make this protocol highly amenable to commercial scale-up and safe industrial operation.
Q: What is the substrate scope for this reaction?
A: The reaction demonstrates broad compatibility with various ketones including acetophenone derivatives, cyclic ketones, and heteroaromatic ketones, tolerating functional groups such as halides, esters, and ethers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Alkenyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed methodology for the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to scale diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of gamma-alkenyl ketone meets the highest industry standards, supporting your regulatory filings and clinical trial needs with reliable, high-quality material.
We invite you to collaborate with us to leverage this advanced synthetic route for your next generation of therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this technology can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
