Advanced Synthetic Route for High-Purity (E,E)-Geranyllinalool Production
Introduction to Advanced Diterpenoid Synthesis
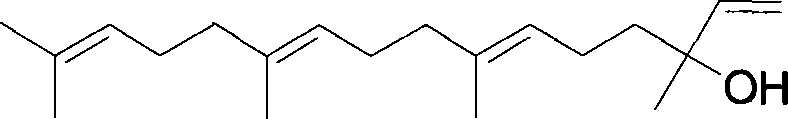
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the production of high-value linear diterpenoids, particularly (E,E)-Geranyllinalool, which serves as a critical precursor for gastric mucosal protective agents like Teprenone. As detailed in patent CN101070270A, a novel synthetic strategy has been developed that addresses long-standing challenges regarding isomer purity and process scalability. This method leverages (E)-Nerolidol as a strategic starting material, utilizing a sequence of protection, regioselective oxidation, and chain extension to achieve the target molecular architecture with high fidelity. The significance of this compound extends beyond pharmaceuticals into the realm of high-end fragrances and insect pheromones, necessitating a supply chain capable of delivering exceptional purity without the burden of complex purification protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (E,E)-Geranyllinalool has been plagued by significant technical hurdles associated with stereoisomer control, particularly when employing Carroll reaction pathways starting from linalool. In these traditional processes, the reaction between linalool and methyl acetoacetate typically yields a mixture of cis and trans isomers of geranyl acetone, which must then be separated via rectification. However, the boiling points of these cis-trans isomers are remarkably close, rendering the separation process energy-intensive, technically demanding, and often resulting in substantial yield losses as nearly half of the material may be discarded as the unwanted isomer. Furthermore, subsequent steps to extend the carbon chain often replicate this issue, requiring multiple difficult distillation stages that increase both the capital expenditure for equipment and the operational costs of manufacturing, thereby limiting the economic viability for general enterprises.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a streamlined route that circumvents the need for arduous isomer separation by leveraging the inherent stereochemistry of (E)-Nerolidol. By protecting the hydroxyl group early in the sequence and employing a highly regioselective oxidation step using selenium dioxide and tert-butyl hydroperoxide, the process ensures that the trans-methyl group is functionalized without disturbing the existing double bond geometry. This strategic approach allows for the precise construction of the carbon skeleton through a coupling reaction with isopropyl methyl ketone, followed by a controlled elimination step that establishes the final double bond with the desired (E,E) configuration. The final deprotection step is conducted under mild acidic conditions, ensuring the integrity of the sensitive polyene chain is maintained while delivering the final product in high purity.

Mechanistic Insights into Regioselective Oxidation and Chain Extension
The cornerstone of this synthetic success lies in the mechanistic precision of the allylic oxidation step, where selenium dioxide acts in concert with tert-butyl hydroperoxide to selectively oxidize the trans-methyl group at the 12-position. This reaction proceeds through a cyclic transition state that favors the abstraction of the allylic hydrogen from the specific methyl group that is trans to the adjacent double bond, thereby installing a hydroxyl functionality with high regioselectivity. This transformation is critical because it sets the stage for the subsequent carbon-carbon bond formation without generating a complex mixture of regioisomers that would be difficult to separate. The preservation of the (E)-configuration at the existing double bonds during this oxidative process is paramount, as any isomerization would compromise the biological activity and olfactory properties of the final diterpenoid product.
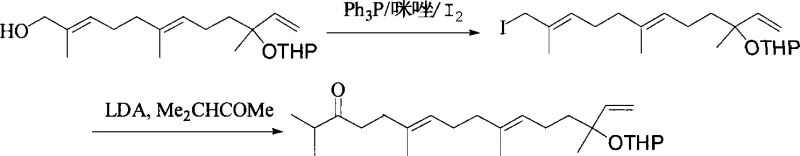
Following the oxidation and subsequent halogenation, the chain extension is achieved through a sophisticated enolate coupling mechanism involving lithium diisopropylamide (LDA) and isopropyl methyl ketone. The LDA selectively deprotonates the ketone to form a kinetic enolate, which then attacks the allylic iodide intermediate in an SN2-like fashion to extend the carbon chain by four carbons. This step is performed under strictly controlled low-temperature conditions to prevent side reactions such as elimination or polymerization, ensuring that the newly formed ketone retains the desired stereochemical arrangement. The subsequent reduction of this ketone to an alcohol, followed by tosylation and base-catalyzed elimination, elegantly installs the terminal vinyl group, completing the construction of the geranyl linalool backbone with minimal risk of geometric isomerization.
How to Synthesize (E,E)-Geranyllinalool Efficiently
The execution of this seven-step synthesis requires careful attention to reaction conditions, particularly regarding temperature control during the lithiation and coupling phases to ensure high yields and purity. The process begins with the protection of the starting alcohol, followed by the critical oxidation and coupling steps described previously, and concludes with a mild deprotection to reveal the final active pharmaceutical ingredient intermediate. Operators must ensure that anhydrous conditions are maintained during the organometallic steps to prevent quenching of the reactive intermediates, while the oxidation steps require careful monitoring to avoid over-oxidation to the corresponding aldehyde or acid. For a detailed breakdown of the specific reagent quantities, solvent choices, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Protect the hydroxyl group of (E)-Nerolidol using dihydropyran (DHP) and PPTS catalyst to form the THP ether.
- Perform regioselective allylic oxidation on the trans-methyl group using SeO2 and t-BuOOH to introduce a hydroxyl group.
- Convert the new hydroxyl to an iodide, then couple with isopropyl methyl ketone using LDA to extend the carbon chain.
- Reduce the resulting ketone with NaBH4, convert the secondary alcohol to a tosylate, and eliminate to form the final double bond.
- Remove the THP protecting group under mild acidic conditions (PPTS/MeOH) to yield pure (E,E)-Geranyllinalool.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers transformative advantages by fundamentally altering the cost structure associated with producing high-purity diterpenoid intermediates. By eliminating the need for multiple high-efficiency distillation columns required to separate cis-trans isomers in traditional methods, the process significantly reduces both the capital investment required for plant infrastructure and the ongoing energy consumption associated with fractional distillation. This simplification of the purification train translates directly into lower manufacturing costs and a more robust supply chain that is less susceptible to bottlenecks caused by complex separation units, thereby enhancing the overall reliability of supply for downstream pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The avoidance of difficult isomer separation steps removes a major cost driver from the production process, as traditional rectification of close-boiling isomers is both energy-intensive and results in significant material loss. Furthermore, the use of readily available and relatively inexpensive reagents such as selenium dioxide, dihydropyran, and common ketones ensures that the raw material costs remain stable and predictable, allowing for substantial cost savings in fine chemical manufacturing without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: Because the synthesis relies on commodity chemicals and avoids the need for specialized, hard-to-source chiral catalysts or exotic reagents, the risk of supply disruption is drastically minimized. The robustness of the reaction conditions, which tolerate standard industrial solvents and do not require extreme pressures or temperatures, means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet fluctuating market demand for teprenone precursors and fragrance ingredients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction steps that are well-understood in chemical engineering terms, such as liquid-liquid extractions and standard crystallizations or chromatography, which are easily adapted from laboratory to pilot to commercial scale. Additionally, the high selectivity of the reactions minimizes the generation of complex waste streams containing difficult-to-separate isomers, simplifying waste treatment protocols and supporting stricter environmental compliance standards in modern chemical production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (E,E)-Geranyllinalool, derived directly from the technical specifications and experimental data provided in the patent literature. These insights are intended to clarify the operational benefits of this specific synthetic route for stakeholders evaluating its potential integration into their existing supply chains or R&D pipelines. Understanding these nuances is essential for making informed decisions about sourcing strategies and process development partnerships.
Q: Why is this synthetic route superior to traditional Carroll reaction methods?
A: Traditional methods often rely on the Carroll reaction which produces mixtures of cis and trans isomers that have very similar boiling points, making separation via rectification extremely difficult and costly. This novel route starts with (E)-Nerolidol and utilizes stereocontrolled reactions that preserve the E-configuration throughout, eliminating the need for complex isomer separation steps.
Q: Can this process be scaled for industrial production of Teprenone intermediates?
A: Yes, the patent explicitly states that the reaction conditions are mild and the reagents are cheap and easy to obtain, making it highly suitable for industrial production. The avoidance of difficult distillation steps significantly simplifies the engineering requirements for large-scale manufacturing.
Q: Is it possible to produce optically active (E,E)-Geranyllinalool using this method?
A: Absolutely. Since the configuration of the tertiary carbon at the 3-position of the starting material (E)-Nerolidol is not affected during the reaction sequence, using optically active (E)-Nerolidol as the raw material will directly result in optically active (E,E)-Geranyllinalool, which is valuable for specific chiral drug syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E,E)-Geranyllinalool Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates like (E,E)-Geranyllinalool play in the successful manufacture of life-saving pharmaceuticals and premium consumer products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality and stringent purity specifications. We utilize rigorous QC labs to verify the geometric purity of our diterpenoid products, guaranteeing that the (E,E)-isomer content meets the exacting standards required for downstream synthesis of drugs like Teprenone.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis tailored to your volume requirements. Whether you require specific COA data to validate our quality systems or detailed route feasibility assessments for custom derivatives, we are committed to providing the transparency and technical expertise necessary to secure your supply chain and accelerate your time to market.
