Revolutionizing Clopidogrel Intermediate Production with High-Boiling Solid Solvent Technology
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the relentless demand for high-quality active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this sector is detailed in patent CN110862372A, which introduces a groundbreaking methodology for synthesizing the key clopidogrel intermediate, (S)-2-(2-thiophene ethylamino)-(2-chlorphenyl)-methyl acetate. This compound serves as a critical building block for clopidogrel bisulfate, a widely prescribed antiplatelet agent used to prevent thrombotic events in patients with acute coronary syndrome. The patent highlights a significant departure from traditional synthetic routes by employing succinonitrile as a novel reaction medium, addressing longstanding challenges related to reaction kinetics, optical purity, and process scalability that have plagued the industry for years.
For R&D directors and process chemists, the implications of this technology are profound. Conventional methods often struggle with maintaining the stereochemical integrity of the chiral center during the nucleophilic substitution step, leading to racemization and reduced therapeutic efficacy. By leveraging the unique physicochemical properties of succinonitrile, this new process not only enhances the specific optical rotation of the final product but also streamlines the purification workflow. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advantages is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines to ensure consistent supply of high-purity materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate has relied on solvents such as acetonitrile, tetrahydrofuran, or even solvent-free conditions, each presenting distinct operational drawbacks that hinder efficient cost reduction in API manufacturing. When acetonitrile is utilized, its relatively low boiling point necessitates prolonged reflux times, often exceeding 40 hours, to drive the reaction to completion. This extended heating leads to significant solvent volatilization and loss, requiring frequent replenishment and complicating the containment of volatile organic compounds (VOCs). Furthermore, literature data indicates that yields in these conventional systems can vary wildly, ranging from as low as 14% to a maximum of 94%, creating unpredictability in production planning.
Alternatively, solvent-free approaches attempt to mitigate environmental concerns but introduce severe engineering challenges. As the reaction progresses, the viscosity of the system increases dramatically due to the consumption of the liquid starting material and the formation of solid products. This phenomenon results in poor mass transfer, uneven stirring, and the risk of materials solidifying on reactor walls, which ultimately compromises both the yield and the optical purity of the product. In aqueous systems, while reaction times may be shorter, the risk of hydrolysis and severe racemization is heightened, with specific optical rotation values dropping drastically to levels incompatible with high-grade pharmaceutical standards.
The Novel Approach
The innovative process disclosed in the patent overcomes these deficiencies by utilizing succinonitrile, a high-boiling solid solvent with a melting point of 54-56°C and a boiling point of 265-267°C. This unique solvent profile allows the reaction to proceed at elevated temperatures (around 95°C) without the risk of solvent evaporation or the need for pressurized equipment. The result is a dramatic reduction in reaction time, often completing within 11 hours compared to the 40+ hours required for acetonitrile. Moreover, the stable thermal environment provided by succinonitrile preserves the chiral integrity of the molecule, consistently delivering products with specific optical rotations near 110 degrees and HPLC purities exceeding 99.8%.
From a commercial perspective, this shift represents a substantial optimization of the production lifecycle. The elimination of solvent loss and the reduction in reaction duration directly translate to lower energy consumption and increased reactor throughput. For supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates, this technology offers a more predictable and robust manufacturing window. The ability to achieve such high purity levels with minimal downstream processing also reduces the burden on quality control laboratories, accelerating the release of batches for subsequent cyclization into the final clopidogrel API.
Mechanistic Insights into Succinonitrile-Mediated Nucleophilic Substitution
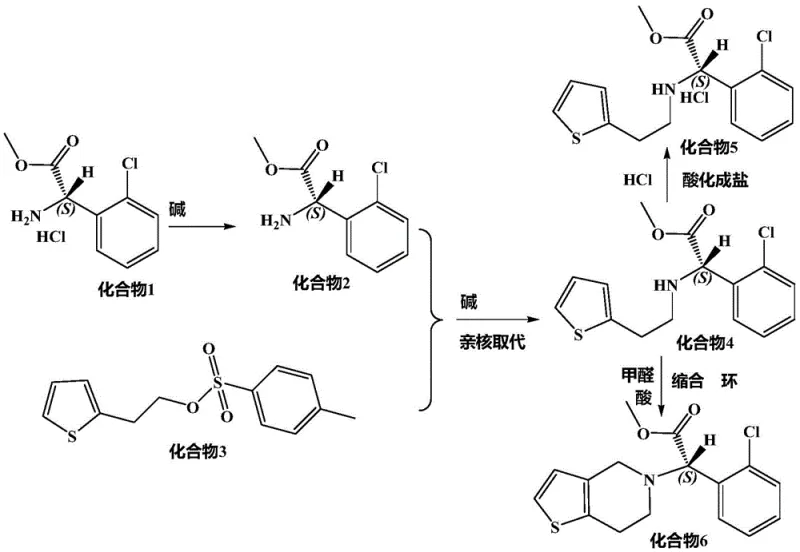
The core of this technological advancement lies in the nucleophilic substitution reaction between (S)-o-chlorophenyl glycine methyl ester and p-toluenesulfonic acid thiophene-2-ethyl ester. In traditional low-boiling solvents, the thermal energy required to overcome the activation barrier of this substitution often promotes competing side reactions, including the epimerization of the alpha-carbon. Succinonitrile acts not merely as a passive medium but as a stabilizing matrix that facilitates efficient molecular collision while suppressing thermal degradation pathways. Its high dielectric constant and ability to dissolve both organic reactants and inorganic acid-binding agents create a homogeneous reaction phase that is crucial for consistent kinetics.
Furthermore, the choice of acid-binding agent plays a synergistic role in this mechanism. The patent specifies the use of dipotassium hydrogen phosphate or potassium carbonate in conjunction with the solvent. These bases effectively neutralize the acid byproduct generated during the substitution without inducing the harsh alkaline conditions that typically trigger racemization. By maintaining a controlled pH environment within the succinonitrile matrix, the process ensures that the nucleophilic attack occurs selectively at the desired position, preserving the (S)-configuration essential for biological activity. This precise control over the reaction microenvironment is what enables the commercial scale-up of complex chiral intermediates with such remarkable fidelity.
How to Synthesize (S)-2-(2-thiophene ethylamino)-(2-chlorphenyl)-methyl acetate Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage pathway that transforms readily available starting materials into the target intermediate with exceptional efficiency. The process begins with the alkalization and dissociation of the hydrochloride salt to generate the free amine, followed by the critical nucleophilic substitution step in succinonitrile, and concludes with acidification to isolate the stable hydrochloride salt. This structured approach minimizes the handling of unstable intermediates and maximizes overall yield. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Alkalize (S)-o-chlorophenyl glycine methyl ester hydrochloride in water using sodium carbonate, extract with dichloromethane, and concentrate to obtain the free base.
- Perform nucleophilic substitution by reacting the free base with p-toluenesulfonic acid thiophene-2-ethyl ester in succinonitrile solvent at 95°C with a phosphate buffer.
- Acidify the resulting mixture with hydrochloric acid in ethyl acetate to precipitate the final hydrochloride salt, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this succinonitrile-based process offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the solvent management system. Unlike acetonitrile, which requires extensive recovery infrastructure to capture evaporated losses, succinonitrile remains in the liquid phase throughout the reaction and can be easily separated and recycled due to its high boiling point. This closed-loop capability significantly reduces raw material costs and minimizes the environmental footprint associated with solvent disposal, aligning with increasingly stringent global sustainability mandates.
- Cost Reduction in Manufacturing: The implementation of this novel solvent system eliminates the hidden costs associated with long reaction cycles and solvent replenishment. By shortening the reaction time from over 40 hours to approximately 11 hours, manufacturers can achieve a higher turnover rate for their reactor assets, effectively increasing production capacity without capital expenditure on new equipment. Additionally, the high purity of the crude product reduces the volume of solvents and reagents needed for recrystallization, further driving down the cost of goods sold (COGS) through qualitative process intensification.
- Enhanced Supply Chain Reliability: Consistency is the cornerstone of a resilient supply chain. The variability inherent in older synthesis methods often leads to batch failures or off-spec material that requires rework, causing delays in delivery schedules. This new process demonstrates remarkable robustness, consistently delivering yields above 90% and optical purity within a narrow, predictable range. Such reliability allows procurement teams to negotiate firmer delivery contracts and maintain lower safety stock levels, knowing that the production process is less susceptible to the fluctuations that typically disrupt the supply of critical cardiovascular intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals unforeseen engineering hurdles, particularly regarding heat transfer and mixing. The succinonitrile method avoids the viscosity issues of solvent-free reactions, ensuring that agitation and temperature control remain effective even at multi-ton scales. Furthermore, the reduction in VOC emissions and the potential for solvent recycling make this process inherently more compliant with environmental regulations, reducing the risk of regulatory shutdowns and facilitating smoother audits by international health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making.
Q: Why is succinonitrile preferred over acetonitrile for this synthesis?
A: Succinonitrile has a significantly higher boiling point (265-267°C) compared to acetonitrile, which prevents solvent loss during long heating periods and maintains consistent reaction concentration, leading to higher yields and reduced racemization.
Q: How does this process improve optical purity?
A: The use of succinonitrile minimizes side reactions and thermal degradation associated with lower boiling solvents, preserving the chiral center and achieving specific optical rotation values around 110 degrees, compared to lower values in aqueous or solvent-free systems.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process addresses key scalability issues such as viscosity and stirring difficulties found in solvent-free methods, making it robust for industrial production with simplified post-reaction processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the succinonitrile process are fully realized in a GMP-compliant environment. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify critical parameters such as specific optical rotation and chiral purity, guaranteeing that every batch meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to optimize your supply chain for cardiovascular therapeutics. By leveraging our technical proficiency in chiral synthesis and process optimization, we can help you secure a stable source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this innovative technology can enhance your competitive position in the market.
