Advanced Manufacturing of Ceftazidime Intermediates: Overcoming Solvation and Impurity Challenges
The pharmaceutical landscape for third-generation cephalosporins relies heavily on the consistent supply of high-quality intermediates, particularly for Ceftazidime, a potent antibiotic effective against Pseudomonas aeruginosa and other resistant gram-negative bacilli. A pivotal advancement in this domain is detailed in patent CN101293891B, which outlines a robust method for preparing Ceftazidime intermediates, specifically focusing on the conversion of Ceftazidime tert-butyl ester solvates into stable hydrates and subsequently into Ceftazidime dihydrochloride. This technology addresses critical bottlenecks in the existing manufacturing infrastructure, offering a pathway to higher purity and operational simplicity. By shifting away from problematic solvent systems like methylene dichloride and methanol, this process mitigates the formation of persistent impurities that have long plagued production lines. For global procurement teams and R&D directors, understanding this shift is essential, as it represents a move towards more sustainable and reliable pharmaceutical intermediate sourcing strategies that align with stringent regulatory standards.
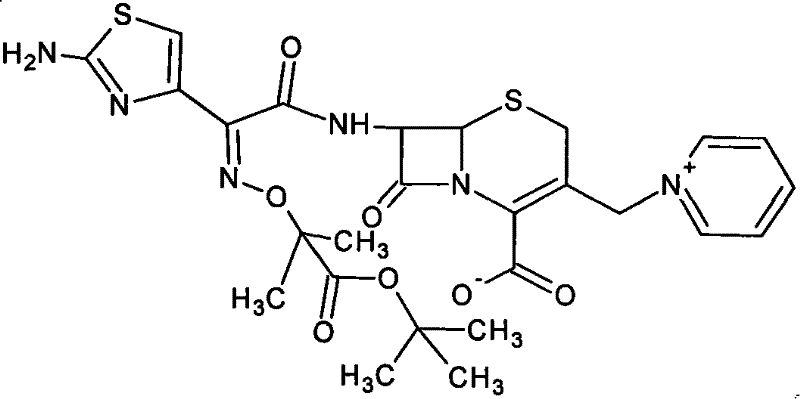
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied on the active ester method, as described in patents like U.S. Pat 5182383, to synthesize Ceftazidime tert-butyl ester. While this method offers mild reaction conditions, it introduces severe downstream complications due to its reliance on methylene dichloride and methanol as solvents. The resulting product is a solvated compound containing these residual solvents, which poses significant storage instability and handling risks. More critically, during the subsequent hydrolysis step under strong acidic conditions, the residual methanol acts as a nucleophile, reacting with the carboxylic acid moiety to form Ceftazidime methyl ester. This specific impurity possesses physicochemical properties nearly identical to the target molecule, making it exceptionally difficult to remove through standard purification techniques like crystallization or chromatography. Furthermore, alternative hydrolysis routes using trifluoroacetic acid (U.S. Pat 5831085) suffer from low yields, while methods using only concentrated hydrochloric acid (U.S. Pat 4954624) are too aggressive, leading to rapid degradation of the sensitive beta-lactam structure.
The Novel Approach
The methodology presented in CN101293891B fundamentally reengineers the solvent and acid systems to bypass these historical pitfalls. The innovation begins with a solvent exchange strategy where the unstable solvated compound is converted into a Ceftazidime tert-butyl ester hydrate using a mixed system of acetone and water. This step effectively purges the problematic methanol and dichloromethane residues, yielding a stable, off-white solid that is far superior for storage and transport. Following this stabilization, the process employs a novel mixed acid hydrolysis system comprising concentrated hydrochloric acid and formic acid. This specific combination allows for precise control over the reaction kinetics at moderate temperatures (10-20°C), ensuring complete de-protection of the tert-butyl group without inducing the degradation associated with pure strong acids. The final crystallization using acetone further ensures high purity, effectively eliminating the methyl ester impurity at its source rather than attempting to remove it post-formation.
Mechanistic Insights into Mixed Acid Hydrolysis and Solvent Exchange
The core chemical breakthrough lies in the modulation of acidity and nucleophilicity within the reaction matrix. In traditional strong acid hydrolysis, the high concentration of protons facilitates rapid cleavage of the tert-butyl ester but simultaneously activates residual alcohols (like methanol) to attack the carbonyl carbon, forming esters. By introducing formic acid alongside hydrochloric acid, the process creates a buffered acidic environment that maintains sufficient proton activity for ester cleavage while reducing the overall aggressiveness that leads to beta-lactam ring opening. The mechanism involves the protonation of the tert-butyl oxygen, followed by the elimination of isobutylene and the formation of the free carboxylic acid. Crucially, the absence of methanol in the acetone-water system removes the competing nucleophile entirely. Additionally, the use of acetone for crystallization leverages the differential solubility of the dihydrochloride salt versus potential organic impurities, driving the equilibrium towards the formation of the desired crystalline lattice.
From an impurity control perspective, this mechanism is transformative. The formation of the hydrate intermediate serves as a "chemical reset," ensuring that the starting material for the hydrolysis step is chemically uniform and free of reactive solvent residues. The controlled temperature range of 10-20°C is vital; it is low enough to suppress thermal degradation pathways but high enough to allow the formic acid-assisted hydrolysis to proceed at a practical rate. This balance results in a product profile where the Ceftazidime methyl ester content is negligible (often below detection limits), and the overall purity exceeds 99%, as evidenced by HPLC analysis in the patent examples. This level of control is essential for meeting the rigorous impurity profiles required by pharmacopoeias for injectable antibiotics.
How to Synthesize Ceftazidime Dihydrochloride Efficiently
The synthesis protocol derived from this patent offers a clear, scalable route for manufacturing facilities aiming to upgrade their production capabilities. The process is divided into two distinct phases: the stabilization of the tert-butyl ester and the subsequent acid hydrolysis. The initial phase requires careful management of the acetone-to-water ratio to ensure complete conversion to the hydrate form without premature hydrolysis. The second phase demands precise temperature control during the addition of the mixed acid system to manage the exotherm and maintain reaction selectivity. For a comprehensive breakdown of the operational parameters, including specific stirring times, washing protocols, and drying conditions, please refer to the standardized synthesis guide below.
- Convert the Ceftazidime tert-butyl ester solvated compound into a stable hydrate form by stirring in a mixed solvent system of acetone and water for 1 to 5 hours.
- Hydrolyze the resulting tert-butyl ester hydrate in a mixed acid system comprising concentrated hydrochloric acid and formic acid at a controlled temperature of 10 to 20°C.
- Induce crystallization of the final Ceftazidime dihydrochloride product by adding acetone to the hydrolysis mixture, followed by filtration and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates directly into risk mitigation and cost optimization. The elimination of methylene dichloride and methanol not only simplifies the environmental compliance burden but also reduces the costs associated with solvent recovery and hazardous waste disposal. By preventing the formation of the methyl ester impurity, the need for complex and yield-loss-inducing purification steps is drastically reduced, leading to a more efficient overall mass balance. Furthermore, the stability of the intermediate hydrate form allows for more flexible inventory management, decoupling the production of the ester from the immediate hydrolysis step and enabling better response to market demand fluctuations.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by streamlining the solvent system and eliminating the need for expensive purification treatments required to remove methyl ester impurities. The use of acetone, a widely available and cost-effective solvent, replaces more expensive or regulated solvents, lowering the raw material input costs. Additionally, the high yield and simplified work-up procedure reduce the consumption of utilities and labor hours per kilogram of finished product, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: The ability to produce a stable intermediate hydrate enhances supply chain resilience by allowing for the stockpiling of key precursors without degradation concerns. This stability reduces the risk of batch failures due to raw material variability, ensuring a consistent flow of materials to the final API synthesis stage. The robustness of the mixed acid hydrolysis also means that the process is less sensitive to minor operational deviations, resulting in higher batch success rates and more predictable delivery schedules for downstream partners.
- Scalability and Environmental Compliance: This method is explicitly designed for industrial suitability, utilizing standard equipment and avoiding extreme conditions that limit scale-up. The reduction in chlorinated solvent usage aligns with modern green chemistry initiatives, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing site. The straightforward crystallization process ensures that the technology can be transferred seamlessly from pilot plants to multi-ton commercial reactors without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ceftazidime intermediate synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing. Understanding these nuances helps stakeholders make informed decisions about process adoption and supplier qualification.
Q: Why is the mixed acid system (HCl + Formic Acid) preferred over pure hydrochloric acid for Ceftazidime hydrolysis?
A: Pure concentrated hydrochloric acid creates an overly aggressive acidic environment that leads to rapid hydrolysis rates and significant product degradation. The addition of formic acid moderates the reaction kinetics, allowing for a controlled de-protection of the tert-butyl group at 10-20°C without compromising the structural integrity of the beta-lactam ring.
Q: How does this process eliminate the 'methyl ester' impurity common in traditional methods?
A: Traditional active ester methods often utilize methanol as a solvent, which reacts with the carboxylic acid group under strong acidic conditions to form a stubborn methyl ester impurity. By switching to an acetone-water system for the intermediate hydrate preparation, methanol is completely excluded from the reaction matrix, thereby fundamentally preventing the formation of this difficult-to-remove byproduct.
Q: What are the storage advantages of the Ceftazidime tert-butyl ester hydrate over the solvated compound?
A: The solvated compounds formed in dichloromethane and methanol are unstable and difficult to store long-term due to residual solvent volatility and reactivity. Converting these into the hydrate form using acetone and water yields an off-white solid with significantly improved stability, lower residual solvent content, and better handling characteristics for downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftazidime Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final antibiotic depends entirely on the precision of its intermediates. Our technical team has extensively analyzed advanced synthesis routes like the one described in CN101293891B to ensure our production capabilities meet the highest global standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities or full-scale commercial supply, our output remains consistent. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities like methyl esters, guaranteeing that every batch of Ceftazidime intermediate we supply supports your regulatory filings and patient safety goals.
We invite you to collaborate with us to optimize your supply chain for Ceftazidime production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects, ensuring a partnership built on transparency, quality, and technical excellence.
