Advanced Selenium-Catalyzed Synthesis of Mono-Thioethers for Commercial Scale-Up and Pharmaceutical Applications
The chemical landscape for sulfur-containing organic compounds is undergoing a significant transformation, driven by the urgent need for greener and more efficient synthetic methodologies in the pharmaceutical and agrochemical sectors. Patent CN102127038B introduces a groundbreaking approach for the synthesis of mono-thioether compounds, utilizing a selenium-catalyzed carbonylation strategy that fundamentally alters the economic and technical feasibility of producing these critical intermediates. Unlike traditional methods that often rely on hazardous hydrogen sulfide or expensive transition metals, this innovation leverages carbon monoxide and water in the presence of elemental selenium to achieve high-yield transformations under remarkably mild conditions. For R&D directors and process chemists, this represents a pivotal shift towards atom-economical pathways that minimize waste generation while maximizing structural diversity. The ability to synthesize complex thioethers from readily available disulfides and alkyl halides opens new avenues for drug discovery, particularly for molecules requiring specific sulfur linkages that were previously difficult or costly to access. This report analyzes the technical depth of this patent to provide actionable insights for procurement and supply chain strategists looking to optimize their intermediate sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioether compounds has been plagued by significant operational challenges that hinder scalability and increase overall production costs for fine chemical manufacturers. Classical approaches typically involve the nucleophilic substitution reaction between alkali metal sulfides, such as potassium sulfide or sodium sulfide, and alkyl halides in anhydrous ethanol. While chemically straightforward, these methods suffer from severe drawbacks including prolonged reaction times, stringent requirements for anhydrous conditions, and the generation of substantial inorganic salt waste that complicates downstream purification. Furthermore, the use of strong bases and volatile organic solvents often necessitates specialized corrosion-resistant equipment and rigorous safety protocols to manage the release of toxic hydrogen sulfide gas. Alternative methods involving phase transfer catalysts or microwave radiation have attempted to mitigate some of these issues but often introduce new complexities regarding catalyst removal and energy consumption. For supply chain managers, these inefficiencies translate into longer lead times, higher capital expenditure for safety infrastructure, and inconsistent batch-to-batch quality due to the sensitivity of the reaction conditions to moisture and oxygen.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN102127038B offers a streamlined one-pot reaction system that operates under atmospheric pressure and moderate temperatures ranging from 20°C to 100°C. By employing elemental selenium as a non-metallic catalyst in conjunction with carbon monoxide and water, this novel route bypasses the need for hazardous sulfide salts and eliminates the generation of corrosive byproducts. The reaction proceeds with exceptional selectivity, often exceeding 99%, which drastically reduces the burden on purification teams and minimizes the loss of valuable starting materials. The integration of water as a reactant rather than just a solvent is a key innovation, facilitating the carbonylation process while maintaining a benign reaction environment. For procurement specialists, this translates to a dramatic simplification of the raw material list, replacing expensive or regulated reagents with commodity chemicals like carbon monoxide and simple alkyl halides. The robustness of this system allows for greater flexibility in manufacturing schedules and reduces the risk of production delays caused by equipment failure or safety incidents associated with high-pressure or high-temperature processes.
Mechanistic Insights into Selenium-Catalyzed Carbonylation
The core brilliance of this patented technology lies in its unique catalytic cycle that combines the benefits of both homogeneous and heterogeneous catalysis through a dynamic phase-transfer mechanism. Initially, the solid selenium powder is insoluble in the organic reaction medium, but upon the introduction of carbon monoxide and the base promoter, it transforms into a soluble active selenium species that drives the homogeneous catalytic conversion of disulfides and alkyl halides. This in-situ generation of the active catalyst ensures intimate contact between reactants, leading to rapid reaction kinetics and high conversion rates even at relatively low temperatures. As the reaction reaches completion and the system is cooled and exposed to air, the active selenium species revert to their elemental solid form, precipitating out of the solution as a fine powder. This self-separating characteristic is a game-changer for process chemistry, as it allows for the physical removal of the catalyst via simple filtration without the need for complex chromatographic techniques or expensive metal scavengers. Such a mechanism not only ensures high product purity but also enables the potential recycling of the selenium catalyst, further enhancing the economic viability of the process for large-scale operations.
From an impurity control perspective, this mechanistic pathway offers superior selectivity compared to traditional nucleophilic substitutions. The involvement of carbon monoxide in the reaction sequence helps to regulate the reactivity of the sulfur species, preventing over-alkylation or the formation of polysulfide byproducts that are common contaminants in conventional thioether synthesis. The use of mild organic or inorganic bases as co-catalysts, such as sodium acetate or triethylamine, further fine-tunes the reaction environment to favor the formation of the desired mono-thioether structure. For quality assurance teams, this means a cleaner crude product profile that requires fewer crystallization steps to meet stringent pharmaceutical specifications. The ability to tolerate various functional groups on both the disulfide and the alkyl halide substrates demonstrates the versatility of this catalytic system, making it applicable to a wide range of complex intermediates used in the synthesis of antibiotics, antifungals, and other bioactive molecules. Understanding this mechanism is crucial for R&D directors evaluating the feasibility of adapting this route for their specific proprietary compounds.
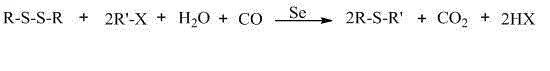
How to Synthesize Mono-Thioether Compounds Efficiently
To implement this advanced synthesis route effectively, process engineers must adhere to specific operational parameters that maximize yield and catalyst efficiency while ensuring safety. The standard procedure involves charging a reactor with the disulfide substrate, alkyl halide, selenium powder, and a selected organic solvent such as DMF or toluene, followed by the addition of water and a base promoter. Continuous flow of carbon monoxide is maintained throughout the heating phase, which typically lasts between 1 to 24 hours depending on the specific substrate reactivity. Upon completion, the critical step of switching the gas feed from carbon monoxide to air triggers the precipitation of the selenium catalyst, allowing for its immediate removal by filtration. The detailed standardized synthesis steps, including precise molar ratios, temperature gradients, and workup procedures, are outlined in the structured guide below to ensure reproducibility and compliance with Good Manufacturing Practices.
- Prepare the reaction mixture by combining disulfide compounds, halogenated hydrocarbons, selenium catalyst, and water in an organic solvent such as DMF or toluene.
- Maintain the reaction at atmospheric pressure with continuous carbon monoxide flow at temperatures between 20°C and 100°C for 1 to 24 hours.
- Cool the mixture, switch gas flow to air to precipitate the catalyst, filter the solid selenium, and purify the filtrate via extraction and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this selenium-catalyzed technology presents a compelling value proposition centered around cost optimization and risk mitigation. The shift from high-pressure, high-temperature processes to atmospheric conditions significantly lowers the barrier to entry for manufacturing, allowing for the utilization of standard glass-lined or stainless-steel reactors without the need for specialized autoclaves. This reduction in capital intensity directly impacts the unit cost of the final intermediate, making it more competitive in the global market. Furthermore, the use of non-metallic selenium as a catalyst avoids the regulatory and cost burdens associated with heavy metal residues, which often require extensive and expensive purification steps to meet ICH guidelines for pharmaceutical ingredients. The simplicity of the one-pot design also reduces labor costs and cycle times, enabling faster turnaround from raw material intake to finished goods. These factors collectively contribute to a more resilient supply chain capable of responding swiftly to market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recycle the selenium catalyst significantly lower the direct material costs associated with production. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified workup procedure minimizes solvent usage and waste disposal fees. The avoidance of complex purification trains required for removing heavy metal traces further drives down operational expenditures, resulting in substantial overall cost savings for the manufacturing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By relying on readily available and stable raw materials such as elemental selenium, carbon monoxide, and common alkyl halides, manufacturers can mitigate the risks associated with supply disruptions of exotic or regulated reagents. The robustness of the reaction conditions ensures consistent batch quality, reducing the incidence of failed batches that can delay shipments and strain customer relationships. This stability allows for more accurate forecasting and inventory management, ensuring a continuous flow of critical intermediates to downstream API producers and formulation companies.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, including the absence of toxic three-waste emissions and the use of water as a reactant, align perfectly with increasingly stringent environmental regulations globally. The ease of scaling from laboratory to commercial production is facilitated by the lack of exothermic hazards and the simplicity of the equipment requirements. This environmental compatibility not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate sustainability profile, which is becoming a key differentiator in supplier selection processes for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium-catalyzed synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the fit of this technology within their existing manufacturing infrastructure and for procurement professionals negotiating supply agreements based on realistic performance metrics.
Q: What are the primary advantages of using selenium catalyst over traditional sulfide synthesis methods?
A: The selenium-catalyzed method operates under atmospheric pressure and mild temperatures, eliminating the need for high-pressure equipment and harsh reagents like potassium sulfide, thereby reducing safety risks and operational costs.
Q: How does this patent address the issue of catalyst recovery and product purity?
A: The process utilizes a unique phase-transfer mechanism where selenium dissolves during the reaction and precipitates as a solid powder upon exposure to air post-reaction, allowing for easy filtration and high-purity product isolation without complex metal scavenging.
Q: Is this synthesis method suitable for large-scale industrial production of pharmaceutical intermediates?
A: Yes, the one-pot reaction design, use of inexpensive raw materials, and absence of toxic three-waste emissions make it highly scalable and compliant with modern environmental regulations for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mono-Thioether Supplier
As a leading CDMO and manufacturer in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate innovative patent technologies like CN102127038B into commercial reality. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can handle the specific nuances of selenium-catalyzed reactions with precision and safety. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect and quantify trace impurities, guaranteeing that every batch of mono-thioether intermediate meets the exacting standards required by the global pharmaceutical market. Our commitment to process optimization allows us to deliver high-quality products that support your drug development timelines and commercial manufacturing needs.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and reduce your overall cost of goods sold. Let us be your partner in bringing high-performance sulfur-containing intermediates to market with speed and reliability.
