Advanced TiCl4-Catalyzed Cyclization for High-Purity Naphthofuran Derivatives Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. A significant breakthrough in this domain is detailed in Chinese Patent CN109574967B, which discloses a novel method for synthesizing naphthofuran derivatives using titanium tetrachloride (TiCl4) as a dehydration reagent. Naphthofuran skeletons are critical structural motifs found in numerous bioactive natural products and synthetic drugs, exhibiting potent properties such as cytotoxicity, antioxidant activity, and antifungal capabilities. Furthermore, these derivatives possess unique photochromic characteristics, making them valuable for advanced material applications. The traditional synthesis routes often suffer from severe limitations, including the requirement for excessive amounts of strong acids, high temperatures, or ultralow temperatures, which complicate scale-up and increase operational costs. This patent introduces a streamlined approach where naphthoxy ketones undergo dehydration and cyclization under the influence of TiCl4 in a mild solvent system. By shifting the paradigm from harsh thermal conditions to Lewis acid catalysis at ambient temperatures, this technology offers a compelling value proposition for manufacturers aiming to optimize their production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
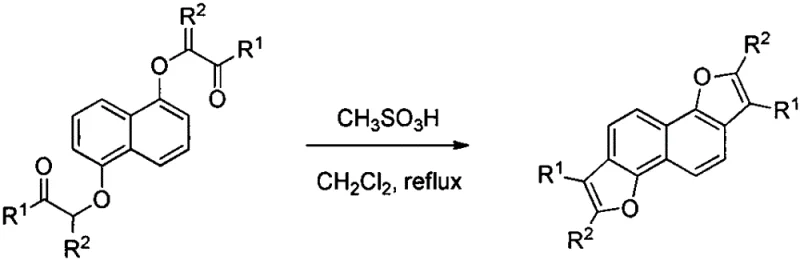
Historically, the construction of the naphthofuran ring system via dehydration cyclization of naphthoxy ketones has been plagued by inefficient and hazardous reaction conditions. Literature precedents, such as those cited in the background of the patent, reveal that open-chain naphthoxy ketones typically require refluxing in dichloromethane for extended periods using a large excess of methanesulfonic acid as the dehydrating agent. Alternatively, the use of phosphorus oxychloride necessitates refluxing for up to six hours, leading to significant energy consumption and potential safety hazards associated with handling corrosive reagents at elevated temperatures. For cyclic naphthoxy ketones, the challenge is even more pronounced; condensation often demands the use of polyphosphoric acid at temperatures as high as 130°C for several hours. These harsh environments not only degrade heat-sensitive functional groups but also promote unwanted side reactions, resulting in lower yields and complex impurity profiles that are difficult to separate. The reliance on such aggressive conditions creates a bottleneck for industrial scalability, increasing the burden on equipment maintenance and waste treatment systems.
The Novel Approach
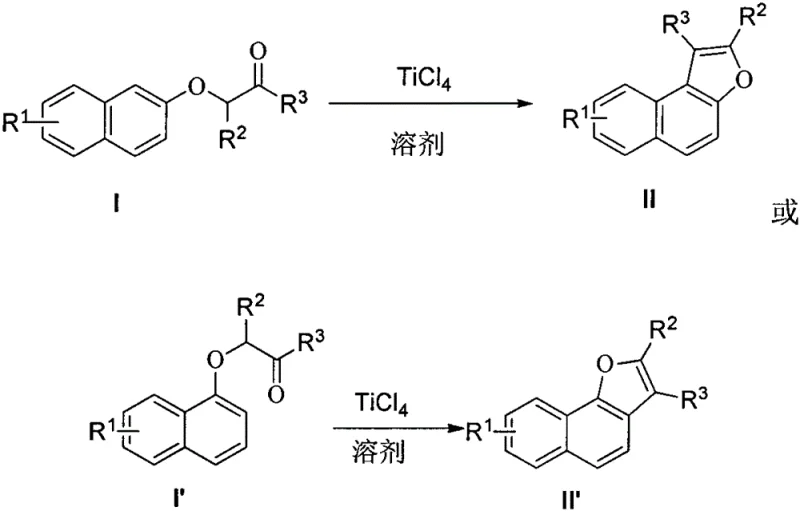
In stark contrast to these legacy methods, the technology described in CN109574967B utilizes titanium tetrachloride to facilitate the cyclization under remarkably mild conditions. The process involves dissolving the naphthoxy ketone substrate in a dry, inert solvent such as dichloromethane and slowly adding a solution of TiCl4. Remarkably, the reaction proceeds efficiently at room temperature (or within a range of 0-120°C, with room temperature being preferred), completing within a short timeframe of 0.1 to 2 hours. This drastic reduction in thermal energy input translates directly to lower operational expenditures and a safer working environment. Moreover, the method exhibits excellent substrate tolerance, accommodating both acyclic and various cyclic ketones (including 5, 6, 7, and 8-membered rings) without the need for specialized high-pressure or high-temperature reactors. The simplicity of the workup procedure, involving a standard aqueous quench and extraction, further underscores the practicality of this approach for large-scale manufacturing, offering a clear pathway to cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into TiCl4-Catalyzed Dehydrative Cyclization
The efficacy of this synthesis lies in the potent Lewis acidity of titanium tetrachloride, which activates the carbonyl group of the naphthoxy ketone towards nucleophilic attack. Upon coordination of the TiCl4 to the carbonyl oxygen, the electrophilicity of the carbonyl carbon is significantly enhanced. This activation facilitates an intramolecular electrophilic aromatic substitution where the electron-rich naphthalene ring attacks the activated carbonyl center. The subsequent elimination of water, driven by the affinity of titanium for oxygen species, leads to the formation of the furan ring fused to the naphthalene core. This mechanism avoids the generation of free carbocations that are common in strong Brønsted acid catalysis, thereby reducing the likelihood of skeletal rearrangements or polymerization side reactions. The result is a highly regioselective transformation that consistently yields the desired naphthofuran structure with minimal isomeric impurities.
Furthermore, the control over impurity formation is a critical aspect of this technology, particularly for clients requiring high-purity OLED material or API intermediates. The mild reaction conditions prevent the degradation of sensitive substituents on the naphthalene ring or the ketone side chain, which might otherwise decompose under the harsh acidic reflux conditions of traditional methods. The use of anhydrous dichloromethane as the solvent ensures that the Lewis acid remains active throughout the reaction without premature hydrolysis, while the inert gas protection prevents moisture ingress that could lead to inconsistent reaction rates. Post-reaction, the titanium species are easily quenched with saturated ammonium chloride, forming water-soluble salts that are removed during the aqueous workup. This clean separation profile ensures that the final organic layer contains predominantly the target naphthofuran derivative, simplifying the downstream purification process and ensuring a superior impurity profile for the final product.
How to Synthesize Naphthofuran Derivatives Efficiently
To implement this synthesis effectively, precise control over stoichiometry and addition rates is essential to manage the exothermic nature of the Lewis acid complexation. The patent outlines a standardized protocol where the naphthoxy ketone is first dissolved in dry dichloromethane under nitrogen, followed by the dropwise addition of the TiCl4 solution to maintain temperature control. Detailed standardized synthesis steps see the guide below.
- Dissolve the naphthoxy ketone substrate in anhydrous dichloromethane under an inert nitrogen atmosphere.
- Slowly add a solution of titanium tetrachloride (TiCl4) in dichloromethane to the reaction mixture at room temperature.
- Quench the reaction with saturated ammonium chloride, extract with dichloromethane, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this TiCl4-mediated synthesis route offers tangible strategic benefits beyond mere technical feasibility. The shift from high-temperature, long-duration reactions to a rapid, room-temperature process fundamentally alters the cost structure of production. By eliminating the need for prolonged heating and the use of massive excesses of corrosive acids like polyphosphoric acid, the process significantly reduces utility costs and reactor occupancy time. This efficiency allows for higher throughput within existing infrastructure, effectively increasing capacity without capital expenditure on new equipment. Additionally, the simplified workup and purification steps reduce the consumption of solvents and stationary phases, contributing to substantial cost savings in raw materials and waste disposal.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the elimination of energy-intensive heating steps and the reduction in reagent consumption. Traditional methods often require refluxing for hours or even days, whereas this novel approach completes in under two hours at room temperature. This drastic reduction in reaction time lowers energy bills and increases the turnover rate of production vessels. Furthermore, the high regioselectivity minimizes the formation of hard-to-remove by-products, reducing the loss of material during purification and improving the overall mass balance of the process. The use of common, inexpensive solvents like dichloromethane and readily available TiCl4 ensures that raw material costs remain stable and predictable.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are widely available in the global market. Unlike specialized catalysts or reagents that may face supply bottlenecks, titanium tetrachloride and dichloromethane are standard industrial chemicals with robust supply lines. The mild reaction conditions also reduce the wear and tear on production equipment, leading to fewer unplanned maintenance downtimes and more consistent delivery schedules. The ability to run the reaction at ambient temperature removes the dependency on complex heating or cooling systems, making the process less vulnerable to utility failures and ensuring continuous operation even in facilities with limited infrastructure.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of extreme thermal or pressure conditions. The exotherm is manageable through controlled addition rates, posing minimal risk of thermal runaway, which is a common concern with strong acid dehydrations. From an environmental perspective, the reduction in reaction time and the avoidance of heavy metal catalysts or excessive acid waste align with green chemistry principles. The simpler aqueous workup generates less hazardous waste compared to the neutralization of large volumes of polyphosphoric acid or methanesulfonic acid, facilitating easier compliance with increasingly stringent environmental regulations and reducing the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making.
Q: What are the advantages of using TiCl4 over traditional acids for naphthofuran synthesis?
A: Unlike traditional methods requiring harsh conditions like refluxing methanesulfonic acid or high-temperature polyphosphoric acid, the TiCl4 method operates at room temperature with shorter reaction times, significantly reducing energy consumption and side reactions.
Q: Is this synthesis method suitable for both cyclic and acyclic substrates?
A: Yes, the patent demonstrates that this protocol is highly versatile, successfully cyclizing both open-chain naphthoxy ketones and various cyclic naphthoxy ketones (5 to 8-membered rings) with good yields.
Q: How does this method impact the purity profile of the final intermediate?
A: The mild reaction conditions and high regioselectivity of the TiCl4 catalyst minimize the formation of by-products and polymerization, resulting in a cleaner crude product that is easier to purify to high specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthofuran Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of next-generation therapeutics and advanced materials. Our team of expert chemists has thoroughly evaluated the TiCl4-catalyzed route described in CN109574967B and validated its potential for commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial supply is seamless. Our state-of-the-art facilities are equipped to handle Lewis acid chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch of naphthofuran derivative meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your supply chain.
