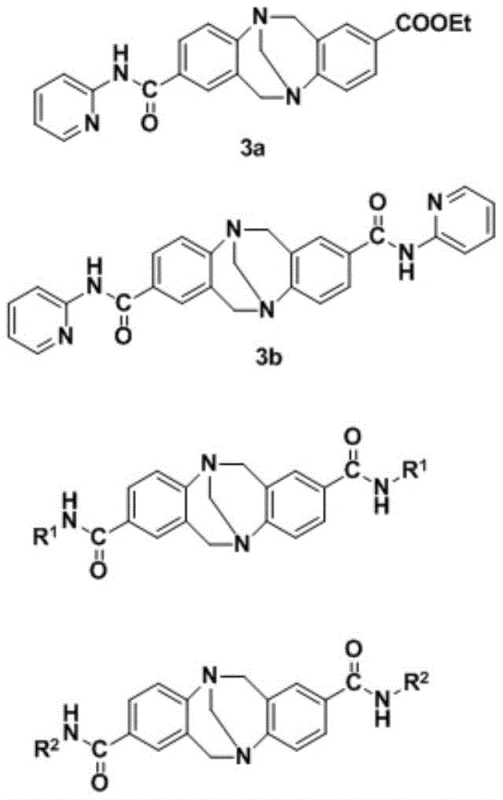
Advanced Amide-Troger's Base Derivatives: Enabling Scalable Synthesis of Anticancer Intermediates
The pharmaceutical industry is constantly seeking novel catalytic systems that can streamline the synthesis of complex heterocyclic scaffolds, particularly those with potential anticancer activity. Patent CN108997341B introduces a groundbreaking approach involving the synthesis and application of Amide-Troger's base derivatives. These unique molecular structures serve as highly efficient organocatalysts for the construction of naphthoquinopyran derivatives, a class of compounds renowned for their diverse biological activities including anticancer, antimalarial, and anti-inflammatory properties. The innovation lies not only in the novel chemical structure of the catalyst itself but also in the robustness of the synthetic methodology employed to create it. By utilizing a straightforward amidation strategy involving azacyclic amines and sodium hydride in anhydrous solvents, this technology offers a pathway to high-purity intermediates that are critical for downstream drug discovery programs. The implications for R&D teams are profound, as this method bypasses the need for expensive and toxic transition metal catalysts often required in similar transformations.
Furthermore, the structural rigidity imparted by the Troger's base backbone provides a unique chiral environment that can potentially influence the stereoselectivity of subsequent reactions, although the primary focus of this patent is on the catalytic efficiency in forming the naphthoquinopyran core. For procurement and supply chain professionals, the adoption of such metal-free organocatalysts represents a strategic shift towards greener chemistry and reduced regulatory burden regarding heavy metal residues in final API products. The patent data underscores the versatility of these derivatives, demonstrating their efficacy across a range of substrates with varying electronic and steric properties. This versatility ensures that the technology is not limited to a single niche application but can be adapted for the synthesis of a broad library of bioactive molecules, thereby enhancing the value proposition for pharmaceutical manufacturers looking to diversify their pipeline with novel anticancer candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of naphthoquinopyran derivatives and similar complex heterocycles has relied heavily on transition metal catalysis or harsh acidic and basic conditions that often compromise the integrity of sensitive functional groups. Conventional methods frequently employ catalysts such as iron oxide nanoparticles, ionic liquids, or Lewis acids which, while effective, introduce significant challenges in terms of product purification and environmental compliance. The removal of trace metal residues from the final pharmaceutical intermediate is a costly and time-consuming process, often requiring multiple chromatography steps or specialized scavenging resins that drive up the overall cost of goods. Moreover, many traditional protocols suffer from long reaction times and moderate yields, which negatively impacts the throughput of process development laboratories. The use of toxic solvents or reagents in these older methods also poses safety risks to operators and creates substantial waste disposal liabilities for manufacturing facilities. These limitations create a bottleneck in the rapid scale-up of promising drug candidates, delaying their entry into clinical trials and ultimately affecting the time-to-market for new therapies.
The Novel Approach
In stark contrast, the methodology described in patent CN108997341B utilizes Amide-Troger's base derivatives as metal-free organocatalysts, effectively eliminating the issues associated with heavy metal contamination. The synthesis of the catalyst itself is achieved under mild conditions using sodium hydride as a base in anhydrous tetrahydrofuran or DMF, which are standard solvents readily available in most chemical supply chains. This novel approach ensures that the reaction environment remains controlled and inert, minimizing side reactions and degradation of the starting materials. The resulting catalysts demonstrate high activity even at relatively low loading levels, facilitating the efficient condensation of naphthoquinones with aldehydes and active methylene compounds. The operational simplicity of heating the reaction mixture to reflux under an inert atmosphere allows for easy scalability from gram-scale laboratory experiments to multi-kilogram pilot production runs. By avoiding the use of expensive noble metals or complex ligand systems, this technology significantly lowers the raw material costs while simultaneously improving the safety profile of the manufacturing process. The high yields reported in the patent examples suggest that this method is not only chemically superior but also economically advantageous for large-scale production.
Mechanistic Insights into Amide-Troger's Base Catalyzed Cyclization
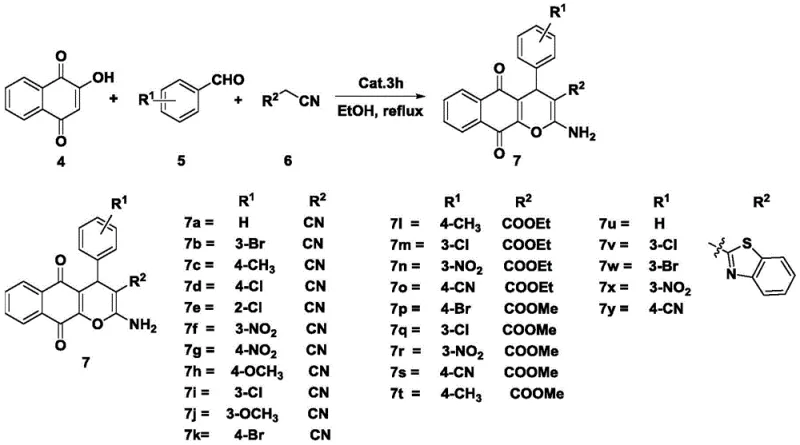
The catalytic mechanism of Amide-Troger's base derivatives in the synthesis of naphthoquinopyran derivatives is rooted in their ability to activate carbonyl groups through hydrogen bonding or nucleophilic interactions, thereby facilitating the Knoevenagel condensation and subsequent cyclization steps. The rigid V-shaped structure of the Troger's base scaffold positions the amide functional groups in a specific spatial orientation that enhances their interaction with the substrate molecules. This pre-organization reduces the entropy of activation, allowing the reaction to proceed more rapidly and with greater selectivity compared to flexible linear analogues. The presence of electron-withdrawing or electron-donating groups on the aromatic rings of the catalyst can be tuned to modulate its acidity or basicity, providing a handle for further optimization of the catalytic performance. During the reaction, the catalyst likely stabilizes the transition state of the Michael addition step, which is often the rate-determining step in the formation of the pyran ring. This stabilization prevents the formation of by-products and ensures that the reaction pathway proceeds cleanly towards the desired naphthoquinopyran core. Understanding these mechanistic details is crucial for R&D directors who need to predict the behavior of the catalyst with new substrates or under different process conditions.
Impurity control is another critical aspect where this mechanistic understanding pays dividends. The mild reaction conditions employed, specifically the use of anhydrous solvents and controlled heating, minimize the risk of hydrolysis or oxidation of sensitive intermediates. The patent data indicates that the crude products obtained from these reactions are of high purity, often requiring only simple filtration or a single column chromatography step for final purification. This is a significant advantage over methods that generate complex mixtures of regioisomers or polymeric by-products. The ability to consistently produce high-purity intermediates is essential for meeting the stringent quality specifications required by regulatory agencies for pharmaceutical ingredients. Furthermore, the robustness of the catalyst under reflux conditions suggests that it is thermally stable and does not decompose during the reaction, which prevents the introduction of catalyst-derived impurities into the final product. This level of control over the chemical process translates directly into reduced waste generation and lower overall production costs, aligning with the principles of green chemistry and sustainable manufacturing.
How to Synthesize Amide-Troger's Base Derivatives Efficiently
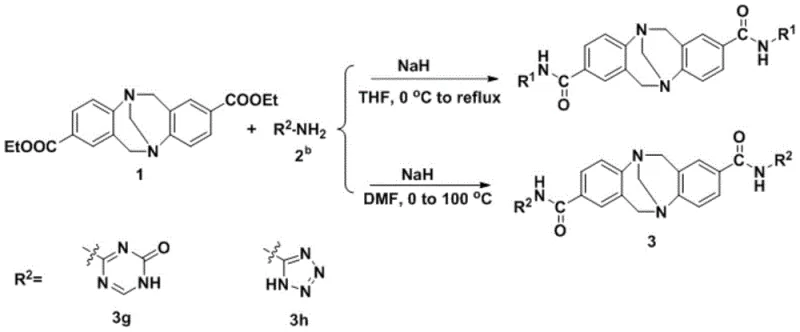
The synthesis of these high-value catalytic intermediates follows a streamlined protocol designed for reproducibility and safety. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to prevent moisture interference, which is critical when using strong bases like sodium hydride. The detailed standardized synthesis steps are provided in the guide below to ensure consistent quality and yield across different production batches. Adhering to these protocols allows manufacturers to leverage the full potential of this technology while maintaining strict control over process parameters.
- Prepare the reaction vessel by adding azacyclic amine and sodium hydride under an inert atmosphere with anhydrous tetrahydrofuran.
- Dissolve the intermediate in anhydrous tetrahydrofuran and slowly drop the mixture into the reaction vessel while stirring at room temperature.
- Heat the container to reflux under inert atmosphere to obtain the target Amide-Troger's base derivative product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Amide-Troger's base technology offers tangible benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts from the process removes a significant cost driver associated with the purchase of expensive noble metals and the subsequent validation of their removal. This shift to organocatalysis simplifies the supply chain by reducing reliance on specialized metal suppliers and mitigates the risk of supply disruptions caused by geopolitical factors affecting metal mining and refining. Additionally, the use of common solvents like tetrahydrofuran and DMF ensures that raw materials are readily available from multiple vendors, fostering a competitive pricing environment and enhancing supply security. The mild reaction conditions also reduce energy consumption compared to high-pressure or high-temperature processes, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost structure of producing naphthoquinopyran intermediates. By removing the need for expensive palladium, platinum, or rhodium catalysts, the direct material costs are significantly reduced. Furthermore, the simplified purification process, which avoids complex metal scavenging steps, lowers the consumption of chromatography media and solvents. This reduction in downstream processing requirements translates into substantial cost savings per kilogram of product. The high yields achieved with this method also mean that less starting material is wasted, improving the overall atom economy of the process. These cumulative efficiencies allow for a more competitive pricing strategy for the final pharmaceutical intermediate, making it an attractive option for cost-sensitive generic drug manufacturers as well as innovative biotech firms.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydride, amines, and standard organic solvents ensures a robust and resilient supply chain. Unlike specialized catalysts that may have long lead times or single-source suppliers, the reagents for this synthesis are produced globally in large volumes. This availability reduces the risk of production delays due to raw material shortages. Moreover, the stability of the Amide-Troger's base derivatives allows for easier storage and transportation, minimizing the need for specialized cold chain logistics. The scalability of the process from laboratory to commercial scale ensures that supply can be ramped up quickly to meet market demand without the need for significant capital investment in new equipment. This flexibility is crucial for maintaining continuity of supply in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: The process described in the patent is inherently scalable, utilizing standard reactor configurations and heating methods that are common in chemical manufacturing plants. The absence of toxic heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The reduced generation of hazardous waste lowers the costs associated with environmental management and liability. Additionally, the high selectivity of the reaction minimizes the formation of by-products, further reducing the burden on waste treatment facilities. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, which is becoming an important factor in supplier selection by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the feasibility and benefits of adopting this method for industrial production. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of using Amide-Troger's base derivatives as catalysts?
A: These derivatives offer mild reaction conditions, short reaction times, and high yields compared to traditional metal catalysts, significantly reducing toxicity and purification costs.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly states broad industrialization prospects due to the use of common solvents like THF and DMF and standard heating reflux techniques.
Q: What is the specific application of these derivatives in medicinal chemistry?
A: They serve as efficient organocatalysts for the synthesis of naphthoquinopyran derivatives, which are potential lead compounds for anticancer drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide-Troger's Base Derivative Supplier
The development of Amide-Troger's base derivatives represents a significant advancement in the field of organocatalysis, offering a powerful tool for the synthesis of complex anticancer intermediates. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is equipped to handle the nuances of this chemistry, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand the critical nature of supply chain continuity for pharmaceutical clients and are committed to delivering high-quality intermediates that facilitate your drug development timelines. Our infrastructure is designed to support both custom synthesis projects and large-scale commercial manufacturing, providing a flexible partnership model that adapts to your specific needs.
We invite you to explore the potential of this technology for your next project by requesting a Customized Cost-Saving Analysis from our technical procurement team. By leveraging our expertise, you can optimize your synthesis route for both cost and efficiency. Please contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to be your strategic partner in bringing novel therapeutics to market faster and more economically.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
