Advanced Synthesis of Chiral Diphosphonic Diamines for Asymmetric Catalysis and Commercial Scale-up
The landscape of asymmetric catalysis is constantly evolving, driven by the demand for more stable and efficient chiral ligands that can operate under mild conditions. Patent CN102382138B, published in early 2014, introduces a significant advancement in this field with the development of a novel class of chiral diphosphonic diamine compounds. These molecules, characterized by a bis{N-2-[(4R)-4,5-dihydro-4-R-2-oxazolinyl]-diphenyl-phenylphosphonic diamide} scaffold, represent a strategic fusion of oxazoline chirality and phosphonamide stability. For R&D directors and process chemists, this patent offers a compelling alternative to traditional phosphine ligands, which often suffer from oxidation issues and high costs associated with precious metal coordination. The technology described herein provides a robust pathway for generating high-value chiral intermediates suitable for critical C-C bond-forming reactions, such as the Henry reaction, thereby addressing the persistent industry need for reliable pharma intermediates supplier solutions that balance performance with manufacturability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral ligands for asymmetric catalysis has relied heavily on phosphine-based architectures, which, while effective, present substantial logistical and safety challenges in a commercial setting. Traditional methods often require the handling of highly air-sensitive reagents that necessitate inert atmosphere gloveboxes, significantly increasing operational complexity and capital expenditure. Furthermore, many conventional routes depend on expensive transition metal catalysts like palladium or rhodium to facilitate coupling reactions, creating a bottleneck in cost reduction in pharmaceutical intermediates manufacturing due to the volatility of precious metal prices and the stringent regulatory requirements for residual metal removal in final API products. The instability of P-C bonds in certain environments also limits the shelf-life and storage conditions of these ligands, complicating supply chain logistics for global chemical distributors who require long-term stability for inventory management.
The Novel Approach
The methodology outlined in CN102382138B circumvents these hurdles by employing a phosphonamide backbone, which inherently possesses greater oxidative stability due to the presence of the phosphoryl (P=O) group. This novel approach utilizes a straightforward two-step synthesis that begins with the cyclization of 2-cyanoaniline and D-amino alcohols, followed by a coupling reaction with phenylphosphonic dichloride. By shifting away from precious metal catalysis in the ligand synthesis itself and utilizing abundant zinc chloride as a Lewis acid promoter, the process drastically simplifies the reaction setup. This shift not only enhances the safety profile by allowing reactions to proceed in standard glassware under reflux but also aligns perfectly with the goals of green chemistry by reducing the reliance on scarce resources. The resulting compounds exhibit excellent solubility profiles in common organic solvents, making them versatile candidates for various homogeneous catalytic applications without the need for specialized formulation techniques.
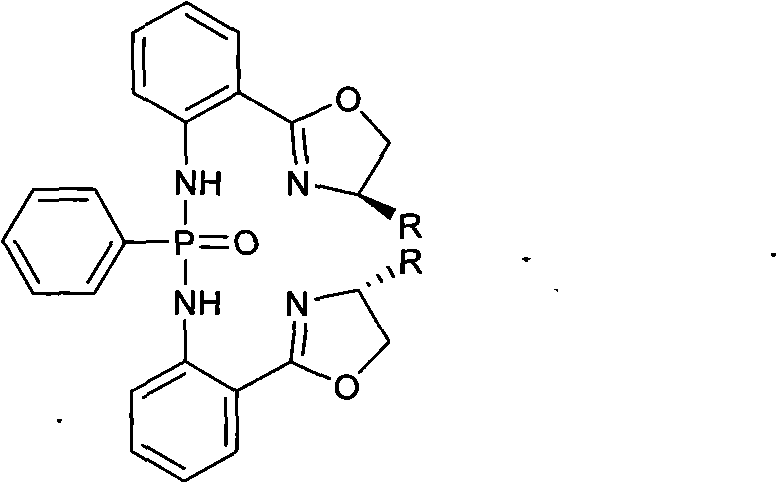
Mechanistic Insights into ZnCl2-Catalyzed Oxazoline Formation
The core of this synthetic strategy lies in the efficient construction of the chiral oxazoline ring, a critical structural motif that dictates the stereochemical outcome of subsequent catalytic reactions. The mechanism involves the activation of the nitrile group in 2-cyanoaniline by the Lewis acidic zinc chloride catalyst, which facilitates a nucleophilic attack by the hydroxyl group of the D-amino alcohol. This cyclization step is thermodynamically driven by the formation of the stable five-membered oxazoline ring and the elimination of ammonia, although in this specific solvent system, the reaction proceeds through a coordinated intermediate complex. The choice of chlorobenzene as the solvent is particularly astute, as its high boiling point allows the reaction mixture to be maintained at reflux temperatures for extended periods (24 hours), ensuring complete conversion of the starting materials despite the steric hindrance presented by bulky R-groups on the amino alcohol side chain.
Following the formation of the oxazoline intermediate, the introduction of the phosphonamide moiety occurs via a nucleophilic substitution at the phosphorus center of phenylphosphonic dichloride. The nitrogen atom of the aniline derivative attacks the electrophilic phosphorus, displacing a chloride ion, which is subsequently scavenged by triethylamine to drive the equilibrium forward. This step creates the robust P-N bond that characterizes the final diphosphonic diamine structure.  The structural integrity of the final product is confirmed by the retention of chirality at the C4 position of the oxazoline ring, which is crucial for inducing asymmetry in downstream reactions. The presence of the P=O group also introduces hydrogen-bond accepting capabilities, which can play a pivotal role in substrate activation during catalysis, offering a dual-functionality that pure phosphine ligands often lack. This mechanistic understanding allows process engineers to fine-tune reaction parameters, such as the stoichiometry of triethylamine, to minimize the formation of mono-substituted byproducts and maximize the yield of the desired bis-substituted target.
The structural integrity of the final product is confirmed by the retention of chirality at the C4 position of the oxazoline ring, which is crucial for inducing asymmetry in downstream reactions. The presence of the P=O group also introduces hydrogen-bond accepting capabilities, which can play a pivotal role in substrate activation during catalysis, offering a dual-functionality that pure phosphine ligands often lack. This mechanistic understanding allows process engineers to fine-tune reaction parameters, such as the stoichiometry of triethylamine, to minimize the formation of mono-substituted byproducts and maximize the yield of the desired bis-substituted target.
How to Synthesize Chiral Diphosphonic Diamine Efficiently
The practical execution of this synthesis requires strict adherence to anhydrous and oxygen-free conditions to prevent the hydrolysis of the acid chloride and ensure the longevity of the zinc catalyst. The process begins with the preparation of the chiral oxazoline intermediate, where precise temperature control during the 24-hour reflux is essential to achieve the reported yields of approximately 60%. Following the reaction, the workup involves a careful extraction process using chloroform and water to separate the organic product from inorganic zinc salts, followed by purification via column chromatography to ensure high optical purity. The second stage involves the coupling of this purified intermediate with phenylphosphonic dichloride in a toluene/triethylamine system, again under reflux for 24 hours, to generate the final diphosphonic diamide.
- Reflux 2-cyanoaniline with D-amino alcohol in chlorobenzene using anhydrous ZnCl2 catalyst for 24 hours to form the chiral oxazoline intermediate.
- Purify the intermediate via aqueous workup and column chromatography to remove zinc salts and unreacted starting materials.
- React the purified intermediate with phenylphosphonic dichloride in toluene and triethylamine under reflux for 24 hours to yield the final diphosphonic diamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical efficacy. The primary advantage lies in the significant cost reduction in fine chemical intermediates manufacturing achieved by eliminating the need for expensive noble metal catalysts during the ligand synthesis phase. By utilizing zinc chloride, a commodity chemical with a stable and low market price, the overall bill of materials is drastically reduced, allowing for more competitive pricing strategies in the global marketplace. Furthermore, the use of standard solvents like chlorobenzene and toluene, which are readily available in bulk quantities from major chemical suppliers, mitigates the risk of supply disruptions that often plague specialty solvent markets. This reliability ensures consistent production schedules and reduces the lead time for high-purity chiral ligands, enabling downstream customers to maintain their own manufacturing timelines without interruption.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the substitution of precious metal catalysts with inexpensive zinc salts, which removes the burden of costly metal recovery and purification steps typically required in pharmaceutical manufacturing. Additionally, the high thermal stability of the reagents allows for energy-efficient heating protocols using standard steam or oil baths rather than specialized cryogenic cooling, further lowering utility costs. The simplified purification workflow, which relies on standard liquid-liquid extraction and chromatography rather than complex distillation or crystallization sequences, reduces labor hours and solvent consumption per kilogram of product. These cumulative efficiencies translate into a leaner cost structure that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically 2-cyanoaniline and various D-amino alcohols, are established commodity chemicals with multiple qualified vendors globally, reducing single-source dependency risks. The robustness of the reaction conditions means that the process is less susceptible to minor variations in environmental factors, such as humidity or ambient temperature, which can often cause batch failures in more sensitive chemistries. This resilience ensures a higher first-pass yield and consistent quality output, allowing supply chain planners to forecast inventory levels with greater accuracy. Consequently, manufacturers can maintain lower safety stock levels while still meeting customer demand, optimizing working capital and warehouse utilization.
- Scalability and Environmental Compliance: From an environmental perspective, the avoidance of heavy metals like palladium or platinum simplifies waste stream management and reduces the regulatory burden associated with hazardous waste disposal. The solvents used in the process are amenable to standard recovery and recycling protocols, supporting sustainability initiatives and reducing the overall carbon footprint of the manufacturing operation. The scalability of the reflux conditions is well-understood in chemical engineering, meaning that transitioning from laboratory scale to multi-ton commercial production involves minimal process re-engineering. This ease of scale-up ensures that supply can be rapidly ramped up to meet surges in demand for chiral intermediates without compromising on product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral diphosphonic diamine compounds. Understanding these details is crucial for integrating this technology into existing supply chains and R&D pipelines.
Q: What are the key advantages of this phosphonamide ligand over traditional phosphines?
A: Unlike traditional phosphines which are often air-sensitive and prone to oxidation, the P=O bond in these diphosphonic diamines offers superior stability. Additionally, the oxazoline moiety provides a rigid chiral environment essential for high enantioselectivity in reactions like the Henry reaction.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes common solvents like chlorobenzene and toluene and avoids cryogenic conditions. The use of ZnCl2 as a Lewis acid catalyst is cost-effective compared to precious metal catalysts, facilitating easier scale-up from lab to pilot plant.
Q: What represents the critical quality attribute (CQA) for this intermediate?
A: The optical purity (ee value) of the D-amino alcohol starting material is critical, as it dictates the stereochemistry of the final oxazoline ring. Furthermore, rigorous removal of residual zinc and chloride ions is necessary to prevent downstream catalyst poisoning.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diphosphonic Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation pharmaceuticals and agrochemicals. Our team of expert process chemists has extensively analyzed the synthetic pathways described in CN102382138B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to the market. We are committed to delivering these complex intermediates with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for asymmetric catalysis applications. Our state-of-the-art facilities are equipped to handle the anhydrous and oxygen-free conditions necessary for this synthesis, guaranteeing product consistency and reliability for our global partners.
We invite R&D directors and procurement specialists to collaborate with us to explore how this cost-effective synthetic route can enhance your project economics. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your manufacturing operations.
