Advanced Chiral Phosphamide Ligands for High-Enantioselective Pharmaceutical Manufacturing
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in patent CN102070669B, which discloses a novel class of chiral phosphamide compounds featuring a chiral oxazoline backbone. These compounds, specifically 2-[(4R)-4,5-dihydro-4-R-2-oxazolinyl]diphenyl phosphamides, represent a significant leap forward in ligand design, offering exceptional catalytic activity and enantioselectivity. For R&D directors and process chemists, the structural versatility of these ligands—where R can be varied from isobutyl to benzyl groups—provides a powerful toolkit for optimizing stereoselective transformations. The ability to achieve high enantiomeric excess (ee) values in critical reactions like the cyanosilylation of aldehydes positions this technology as a cornerstone for modern chiral drug manufacturing.
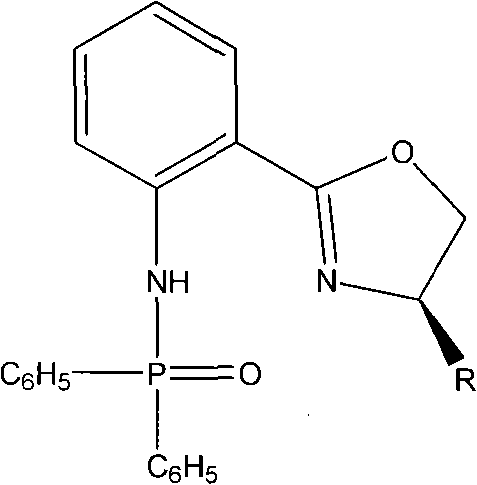
From a supply chain perspective, the robustness of the synthetic route described in the patent ensures that these high-value catalysts can be produced reliably. The methodology avoids exotic reagents, relying instead on established organic transformations that are amenable to scale-up. This reliability is crucial for procurement managers seeking to mitigate risks associated with catalyst availability. By integrating these chiral phosphamides into existing synthetic workflows, manufacturers can potentially streamline purification processes, as higher selectivity often translates to fewer downstream separation steps. Consequently, this technology not only enhances the chemical efficiency of asymmetric synthesis but also aligns with broader industry goals of cost reduction and sustainable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to asymmetric cyanosilylation and similar nucleophilic additions have long relied on a variety of chiral Lewis acids and organocatalysts, many of which suffer from significant drawbacks in an industrial setting. Conventional metal-based catalysts often require stringent anhydrous conditions that are difficult to maintain on a multi-kilogram scale, leading to batch-to-batch variability and inconsistent enantioselectivity. Furthermore, many established protocols utilize expensive transition metals such as titanium or rare earth elements, which introduce concerns regarding heavy metal contamination in the final active pharmaceutical ingredient (API). The removal of these trace metals often necessitates additional purification steps, such as specialized scavenging or recrystallization, which drastically increases processing time and operational costs. Additionally, older ligand systems frequently exhibit narrow substrate scopes, requiring extensive re-optimization when switching between different aldehyde substrates, thereby slowing down the overall drug development timeline.
The Novel Approach
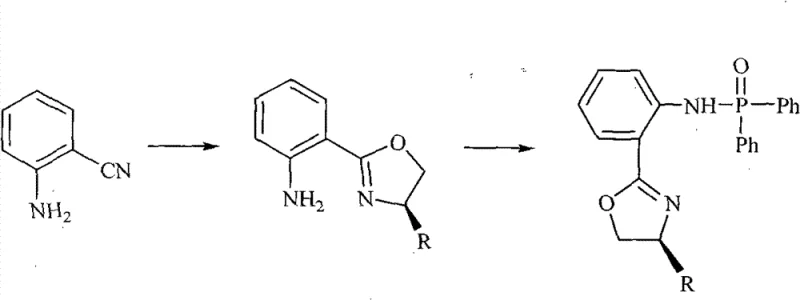
The methodology outlined in patent CN102070669B presents a transformative alternative by utilizing a structurally rigid chiral phosphamide framework that combines the stability of phosphorus chemistry with the stereochemical control of oxazolines. This novel approach employs a two-step synthesis that is both operationally simple and highly effective. The first step involves a zinc-catalyzed cyclization to form the chiral oxazoline intermediate, followed by a phosphorylation step to install the diphenylphosphinyl group. This route avoids the use of air-sensitive reagents typically associated with organolithium or Grignard chemistry, making the process safer and more manageable in standard reactor setups. The resulting catalysts demonstrate remarkable tolerance to reaction conditions, maintaining high activity even in the presence of minor impurities. By shifting the paradigm towards these robust organic-inorganic hybrid ligands, manufacturers can achieve consistent, high-yielding transformations that significantly reduce the complexity of process validation.
Mechanistic Insights into Chiral Phosphamide Catalysis
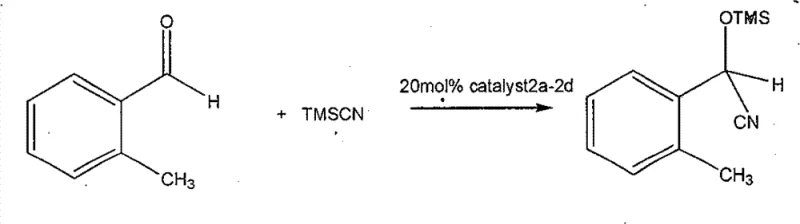
The exceptional performance of these chiral phosphamide compounds stems from their unique ability to act as bidentate ligands, coordinating to metal centers through both the phosphoryl oxygen and the oxazoline nitrogen atoms. This coordination creates a well-defined chiral environment around the catalytic center, effectively shielding one face of the substrate while exposing the other to the incoming nucleophile. In the context of the cyanosilylation of 2-tolualdehyde, as demonstrated in the patent data, the catalyst activates the carbonyl group of the aldehyde via Lewis acid coordination, increasing its electrophilicity. Simultaneously, the bulky R-groups on the oxazoline ring (such as the isobutyl group in catalyst 2a or the phenyl group in 2c) exert steric pressure that directs the approach of the trimethylsilyl cyanide (TMSCN). This dual activation and steric guidance mechanism is responsible for the observed enantioselectivities, which can reach up to 99% ee depending on the specific R-substituent employed.
Impurity control is another critical aspect where the mechanistic understanding of this system offers distinct advantages. The high specificity of the catalyst-substrate interaction minimizes side reactions such as oligomerization or non-selective background reactions that often plague less selective systems. The patent data indicates that the reaction proceeds efficiently at moderate temperatures (20-30°C), which further suppresses thermal degradation pathways and the formation of byproducts. For quality control teams, this means a cleaner crude reaction profile, simplifying the analytical burden and reducing the load on purification columns. The stability of the phosphamide bond under reaction conditions ensures that the catalyst does not decompose into phosphoric acid derivatives that could complicate downstream workups. This mechanistic robustness is a key factor in translating laboratory success to commercial viability, ensuring that the purity specifications required for pharmaceutical intermediates are consistently met.
How to Synthesize Chiral Phosphamide Efficiently
The synthesis of these high-performance ligands is designed for reproducibility and scalability, utilizing standard organic synthesis techniques that are familiar to process chemists. The protocol begins with the condensation of 2-cyanoaniline and a chiral D-amino alcohol, a reaction that is catalyzed by anhydrous zinc chloride in chlorobenzene. This step is critical for establishing the chiral center and forming the oxazoline ring, which serves as the stereochemical director. Following the isolation of the intermediate, the second step involves coupling with diphenylphosphinyl chloride in the presence of triethylamine. Detailed standardized synthesis steps, including precise stoichiometry, temperature profiles, and purification methods, are essential for achieving the reported yields and optical purities.
- Reflux 2-cyanoaniline with D-amino alcohol in chlorobenzene using anhydrous ZnCl2 catalyst under oxygen-free conditions for 24 hours to form the oxazoline intermediate.
- Purify the intermediate via aqueous workup, chloroform extraction, and column chromatography to isolate the pure oxazoline-aniline derivative.
- React the purified intermediate with diphenylphosphinyl chloride in toluene and triethylamine under reflux for 24 hours to yield the final chiral phosphamide target.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chiral phosphamide technology offers tangible benefits that extend beyond mere chemical performance. The primary advantage lies in the simplification of the supply chain for chiral building blocks. Because the catalysts are derived from readily available amino alcohols and commodity anilines, the risk of raw material shortages is significantly minimized compared to technologies relying on complex, proprietary chiral pools. This accessibility translates directly into cost stability and predictable lead times, allowing for more accurate production planning. Furthermore, the high efficiency of the catalyst reduces the overall loading required per batch, meaning that a smaller quantity of catalyst can drive the conversion of larger amounts of substrate, effectively lowering the cost per kilogram of the final product.
- Cost Reduction in Manufacturing: The implementation of this catalytic system eliminates the need for expensive stoichiometric chiral auxiliaries or precious metal catalysts that are difficult to recover. By utilizing a catalytic amount of a robust ligand that can be synthesized from low-cost starting materials, the direct material costs are substantially reduced. Additionally, the high enantioselectivity reduces the burden on chiral separation technologies, such as preparative HPLC or resolution crystallization, which are often the most expensive steps in chiral manufacturing. The elimination of these costly purification stages results in significant operational savings and a lower carbon footprint for the manufacturing process.
- Enhanced Supply Chain Reliability: The synthetic route described in the patent utilizes reagents that are widely available in the global chemical market, reducing dependency on single-source suppliers for exotic materials. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum, allows for production in a wider range of manufacturing facilities, thereby diversifying the potential supply base. This flexibility ensures continuity of supply even in the face of regional disruptions, providing a strategic buffer for long-term production contracts. The stability of the final phosphamide products also allows for extended shelf-life, facilitating inventory management and reducing waste due to degradation.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot production without fundamental changes to the reaction engineering. The use of common solvents like toluene and chlorobenzene, which have established recovery and recycling protocols in most chemical plants, simplifies waste management and solvent handling. Moreover, the avoidance of heavy metals in the final catalytic step aligns with increasingly stringent environmental regulations regarding metal residues in pharmaceuticals. This compliance reduces the regulatory burden and accelerates the approval process for new drug applications that utilize intermediates produced via this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral phosphamide technology. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, offering a clear picture of what partners can expect when integrating this chemistry into their pipelines. Understanding these nuances is vital for making informed decisions about process adoption and supplier selection.
Q: What represents the primary advantage of these chiral phosphamide ligands over traditional metal complexes?
A: These ligands offer superior enantioselectivity (up to 99% ee) in cyanosilylation reactions while utilizing readily available starting materials, reducing the dependency on scarce precious metal catalysts.
Q: Are the starting materials for this synthesis commercially scalable?
A: Yes, the synthesis relies on commodity chemicals such as 2-cyanoaniline and various D-amino alcohols (leucinol, valinol, etc.), ensuring a robust and continuous supply chain for large-scale production.
Q: How does the R-group variation affect the catalytic performance?
A: The steric bulk of the R-group (isobutyl, isopropyl, phenyl, or benzyl) directly influences the chiral pocket geometry, allowing fine-tuning of stereoselectivity for specific substrate classes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral catalysts play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity chiral phosphamide compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR capabilities. Our dedication to quality ensures that every batch of catalyst performs consistently, providing the reliability that R&D and production teams demand for critical synthetic steps.
We invite you to collaborate with us to optimize your asymmetric synthesis processes and achieve your cost and efficiency targets. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how switching to our catalytic solutions can improve your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a supply chain that prioritizes innovation, quality, and long-term strategic value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
