Advanced Chiral Zinc-Nitrogen Coordination Compounds for Industrial Asymmetric Synthesis
The landscape of asymmetric catalysis is continually evolving, driven by the urgent need for more efficient and selective chiral inducers in the synthesis of high-value pharmaceutical intermediates. Patent CN102153571B introduces a groundbreaking chiral zinc-nitrogen coordination compound that addresses critical limitations in current catalytic systems. This specific metal-organic complex, characterized by its unique Zn-N bonding architecture, offers a robust platform for enantioselective transformations, particularly in cyanosilylation reactions. For R&D directors and procurement specialists, understanding the structural integrity and synthetic accessibility of this compound is paramount for integrating it into existing production pipelines. The patent details a reproducible methodology that leverages abundant zinc salts and chiral amino alcohols, signaling a shift towards more sustainable and cost-effective catalytic solutions. By focusing on the precise coordination environment around the zinc center, this technology promises to enhance reaction selectivity while minimizing the reliance on expensive noble metals. As we delve into the technical specifics, it becomes clear that this innovation represents a significant leap forward for manufacturers seeking reliable chiral zinc catalyst suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for generating chiral catalysts often rely heavily on scarce transition metals such as rhodium, palladium, or ruthenium, which introduce substantial cost volatility and supply chain risks for large-scale manufacturing. Furthermore, the synthesis of conventional chiral ligands frequently involves multi-step organic synthesis with low overall yields, creating bottlenecks in the production of fine chemical intermediates. Many existing zinc-based systems suffer from instability under ambient conditions or require rigorous exclusion of moisture and oxygen that is difficult to maintain in industrial reactors. The purification of these traditional catalysts often necessitates complex chromatographic separations that are not easily scalable, leading to increased waste generation and higher operational expenditures. Additionally, the catalytic activity of older generation complexes often diminishes over time or requires high loading amounts to achieve acceptable conversion rates, negatively impacting the economic feasibility of the process. These cumulative inefficiencies highlight the critical need for a more robust, accessible, and high-performance catalytic system that can withstand the rigors of commercial production environments.
The Novel Approach
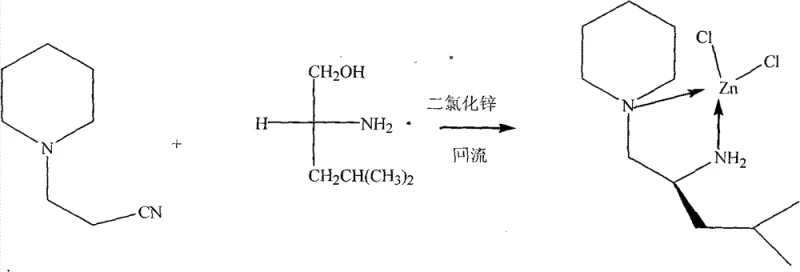
The methodology outlined in patent CN102153571B presents a streamlined synthetic route that circumvents many of the drawbacks associated with conventional catalyst preparation. By utilizing hexahydropyridine propionitrile and D-leucinol as precursors, the process capitalizes on readily available starting materials that significantly reduce raw material costs and lead times. The reaction proceeds under reflux in chlorobenzene with a large excess of anhydrous zinc chloride, driving the coordination equilibrium towards the formation of the desired chiral complex with high efficiency. This one-pot synthesis strategy minimizes the number of isolation steps required, thereby reducing solvent consumption and waste generation compared to multi-step ligand synthesis protocols. The resulting complex exhibits remarkable stability and can be purified effectively using standard column chromatography techniques, yielding high-quality single crystals suitable for structural characterization. This novel approach not only simplifies the manufacturing workflow but also enhances the overall atom economy of the process, making it an attractive option for cost reduction in asymmetric catalysis manufacturing.
Mechanistic Insights into Zn-N Coordination and Catalytic Cycle
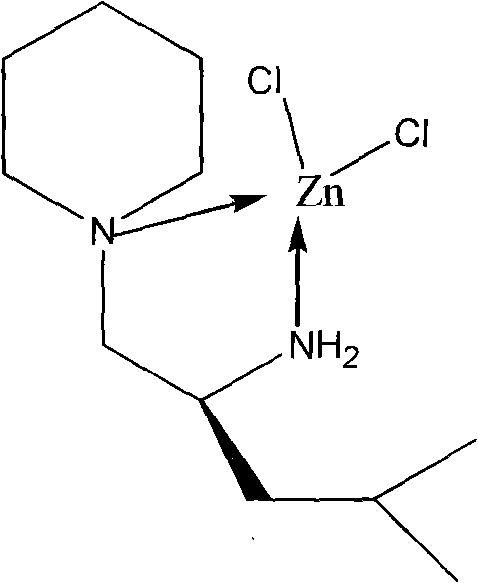
The core innovation of this technology lies in the specific coordination geometry established between the zinc center and the nitrogen-containing chiral ligand. The zinc atom, acting as a Lewis acid, coordinates with the nitrogen atoms from the hexahydropyridine ring and the amino group of the leucinol derivative, creating a rigid chiral environment essential for stereocontrol. This specific arrangement facilitates the activation of the cyanosilane reagent by increasing the electrophilicity of the silicon center, thereby promoting nucleophilic attack on the carbonyl substrate. The steric bulk provided by the isobutyl group of the leucinol moiety plays a crucial role in differentiating the enantiofaces of the incoming aldehyde, ensuring high levels of enantioselectivity in the product formation. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction conditions for diverse substrates beyond benzaldehyde. The stability of the Zn-N bond under the reaction conditions ensures that the catalyst maintains its integrity throughout the catalytic cycle, preventing decomposition and loss of activity.
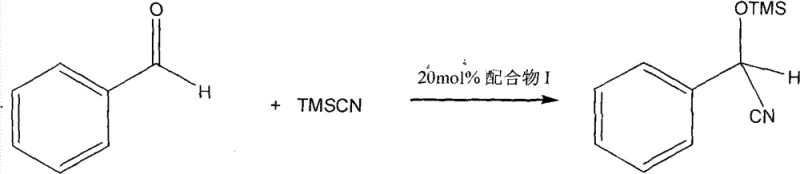
Furthermore, the catalytic performance of this complex in the cyanosilylation of benzaldehyde demonstrates its practical utility in synthesizing valuable chiral intermediates. The reaction proceeds with a conversion rate of 64% under mild conditions, indicating a balance between reactivity and selectivity that is often difficult to achieve. The mechanism likely involves the formation of a pentacoordinate silicon intermediate stabilized by the chiral zinc complex, which lowers the activation energy for the C-C bond formation step. Impurity control is inherently managed by the high specificity of the catalyst, which minimizes side reactions such as homocoupling or non-selective addition. For quality assurance teams, this means a cleaner reaction profile and reduced burden on downstream purification processes. The ability to tune the electronic properties of the ligand by modifying the substituents offers a pathway for further optimization, allowing for the customization of the catalyst for specific high-purity pharmaceutical intermediates.
How to Synthesize Chiral Zinc-Nitrogen Complex Efficiently
The synthesis of this high-performance catalyst is designed to be robust and scalable, adhering to strict anhydrous and anaerobic protocols to ensure maximum yield and purity. The process begins with the precise weighing of anhydrous zinc chloride and the organic precursors, followed by their dissolution in dry chlorobenzene under an inert atmosphere. The mixture is then subjected to prolonged reflux, allowing the thermodynamic equilibrium to favor the formation of the stable coordination complex. Detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are critical for reproducing the results described in the patent documentation. Adhering to these protocols ensures that the resulting catalyst possesses the necessary structural fidelity to perform effectively in asymmetric transformations. For process chemists, mastering these synthesis parameters is the first step towards integrating this technology into a commercial manufacturing workflow.
- Prepare the reaction mixture by combining hexahydropyridine propionitrile and D-leucinol under strict anhydrous and anaerobic conditions with excess anhydrous ZnCl2.
- Conduct the reflux reaction in chlorobenzene solvent for 24 hours to ensure complete coordination and formation of the complex.
- Purify the resulting crude product via column chromatography using petroleum ether and dichloromethane to isolate the white crystalline complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this zinc-based catalytic system offers substantial strategic advantages over traditional noble metal catalysts. The primary benefit stems from the use of zinc, a base metal that is abundant, inexpensive, and less subject to the geopolitical supply constraints that often plague precious metal markets. This shift in raw material dependency translates directly into significant cost savings in manufacturing, as the overall material cost per kilogram of catalyst is drastically reduced. Moreover, the simplified synthetic route reduces the consumption of specialized solvents and reagents, further lowering the operational expenditure associated with catalyst production. For supply chain heads, the reliability of the raw material supply ensures continuity of production, mitigating the risk of delays caused by material shortages. The robustness of the catalyst also implies a longer shelf life and easier handling requirements, reducing the need for specialized storage infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals like palladium or rhodium from the catalyst formulation results in a direct and substantial reduction in raw material costs. By utilizing zinc chloride, which is a commodity chemical, the production budget can be optimized without compromising on catalytic performance. Additionally, the high efficiency of the reaction reduces the required catalyst loading, meaning less material is needed to achieve the same output, further driving down the cost per unit of product. The simplified purification process also reduces labor and utility costs associated with complex separation techniques. These cumulative factors create a compelling economic case for switching to this zinc-nitrogen system for large-scale industrial applications.
- Enhanced Supply Chain Reliability: The reliance on widely available organic precursors such as hexahydropyridine propionitrile and D-leucinol ensures a stable and resilient supply chain. Unlike specialized chiral ligands that may have limited suppliers, these starting materials are commercially accessible from multiple sources, reducing the risk of single-supplier dependency. This diversity in sourcing options allows procurement managers to negotiate better terms and secure consistent delivery schedules. The stability of the final complex also means that inventory can be held for longer periods without degradation, providing a buffer against market fluctuations. This reliability is crucial for maintaining uninterrupted production schedules in the fast-paced pharmaceutical and fine chemical industries.
- Scalability and Environmental Compliance: The synthetic process is inherently scalable, as it utilizes standard unit operations like reflux and column chromatography that are well-understood in industrial settings. The use of chlorobenzene, while requiring careful handling, is a common solvent with established recovery and recycling protocols, aiding in environmental compliance. The reduction in heavy metal waste aligns with increasingly stringent environmental regulations, simplifying the disposal process and reducing associated fees. The ability to scale from gram to kilogram quantities without significant re-optimization makes this technology ideal for commercial scale-up of complex pharmaceutical intermediates. This scalability ensures that the technology can grow with the demand, supporting long-term business expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc-nitrogen complex in industrial settings. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details is essential for making informed decisions about adopting this technology for your specific production needs. The information covers aspects ranging from catalytic performance to handling requirements, ensuring a comprehensive overview for all stakeholders.
Q: What is the primary catalytic application of this zinc-nitrogen complex?
A: The complex demonstrates significant catalytic performance in the cyanosilylation of benzaldehyde, achieving high conversion rates under mild conditions.
Q: How does this synthesis method improve upon traditional coordination compound preparation?
A: The method utilizes a one-pot reflux strategy in chlorobenzene which simplifies the purification process and enhances the yield of the chiral single crystals.
Q: Is this catalyst suitable for large-scale pharmaceutical intermediate production?
A: Yes, the use of stable zinc salts and accessible organic ligands suggests strong potential for scalability and cost-effective manufacturing in fine chemical sectors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc-Nitrogen Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral zinc-nitrogen coordination compound described in patent CN102153571B for the future of asymmetric synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative catalyst can be seamlessly integrated into your supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical applications. We understand the critical importance of consistency and quality in catalyst manufacturing, and our team is dedicated to delivering products that meet the highest industry standards. By partnering with us, you gain access to a wealth of technical expertise and production capacity that can accelerate your project timelines.
We invite you to explore how this advanced catalytic technology can optimize your current manufacturing processes and drive significant value for your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We encourage you to reach out to request specific COA data and route feasibility assessments to verify the compatibility of this catalyst with your existing workflows. Let us help you navigate the complexities of chiral synthesis and secure a competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
