Advanced Phosphorescent Manganese Complexes for Next-Generation OLED Manufacturing
Advanced Phosphorescent Manganese Complexes for Next-Generation OLED Manufacturing
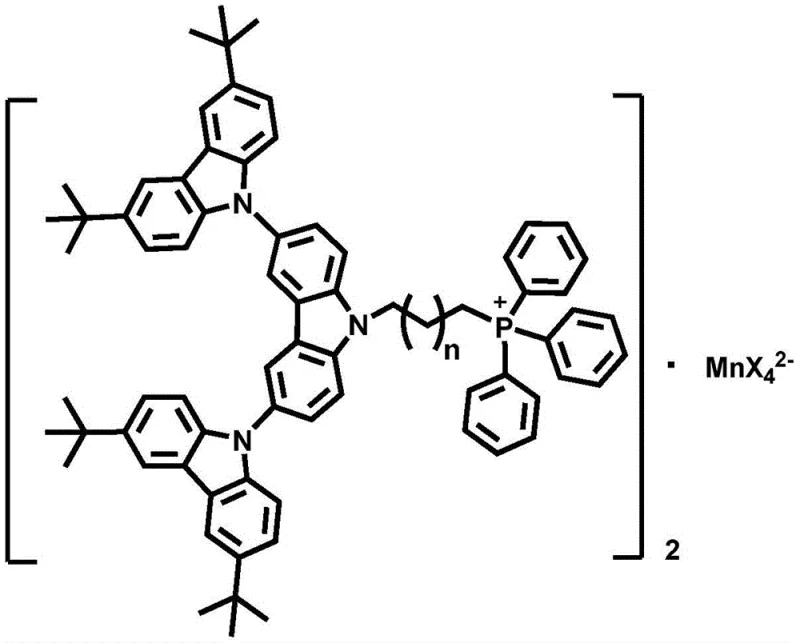
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that balance high performance with economic viability. Traditional phosphorescent emitters relying on noble metals like iridium and platinum have long dominated the market, yet their scarcity and high cost present persistent supply chain vulnerabilities. A groundbreaking solution emerges from patent CN108409787B, which details the synthesis and application of a novel phosphorescent manganese complex featuring a tricarbazole functionalized dendritic structure. This innovation represents a paradigm shift towards earth-abundant metal-based emitters that do not compromise on efficiency. By integrating a hole-transporting dendritic unit with a four-coordinate manganese center, this technology achieves dual functionality as both an emissive and charge-transport material. For global procurement leaders and R&D directors, this development signals a new era of cost-effective, high-efficiency display materials that mitigate the risks associated with precious metal dependency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fabrication of high-efficiency PhOLEDs has relied heavily on doping phosphorescent heavy metal complexes into host matrices. While effective, this host-guest system suffers from inherent thermodynamic instability, often leading to phase separation during device operation or storage. Such phase separation drastically shortens the operational lifetime of the display panel, a critical failure mode for consumer electronics. Furthermore, at high current densities, these conventional systems experience severe Triplet-Triplet Annihilation (TTA), causing efficiency roll-off and rapid device decay. The reliance on iridium and platinum not only inflates raw material costs but also introduces significant volatility into the supply chain, as these metals are geographically concentrated and subject to market fluctuations. Additionally, the synthesis of complex organometallic emitters often requires rigorous purification steps to remove trace metal impurities, adding layers of complexity and expense to the manufacturing process.
The Novel Approach
The technology disclosed in patent CN108409787B circumvents these challenges through a sophisticated molecular design that merges the benefits of dendrimers with the photophysical properties of manganese. Instead of simple doping, the phosphorescent manganese complex is constructed with a bulky tricarbazole dendritic cation that inherently prevents molecular aggregation. This structural feature effectively suppresses concentration quenching, allowing the material to function efficiently even in neat films or high-concentration blends. The use of manganese, an abundant transition metal, fundamentally alters the cost structure of the emitter, offering a sustainable alternative to noble metals. Moreover, the carbazole dendrons serve as efficient hole-transport units, simplifying the device architecture by reducing the need for separate hole-transport layers. This multifunctional approach streamlines the layer stack, potentially lowering production costs and improving the overall reliability of the organic electroluminescent device.
Mechanistic Insights into Dendritic Manganese Coordination
The core of this innovation lies in the precise coordination chemistry that stabilizes the manganese center within an organic-inorganic hybrid framework. The synthesis begins with the construction of a robust tricarbazole core via Ullmann coupling, creating a rigid, three-dimensional scaffold. This scaffold is then functionalized with an alkyl chain terminating in a phosphonium group, which serves as the anchoring point for the manganese anion. The resulting cationic ligand interacts with manganese dihalides (MnX2) to form a tetrahedral or octahedral complex, depending on the specific halide and crystal field environment. In the solid state, the dendritic arms create a protective shell around the emissive manganese center, isolating it from neighboring molecules and preventing non-radiative decay pathways. This isolation is crucial for maintaining high photoluminescence quantum yields (PLQY) and ensuring that the triplet excitons generated during electroluminescence are harvested efficiently.
Furthermore, the electronic properties of the carbazole units play a pivotal role in the device's performance. Carbazole is well-known for its high triplet energy level and excellent hole-transporting capability. By covalently linking these units to the manganese center, the material facilitates balanced charge injection and transport. The energy transfer from the carbazole ligand to the manganese center is highly efficient, acting as an antenna effect that sensitizes the metal-centered emission. This mechanism ensures that even if the direct excitation of the manganese ion is weak, the absorption cross-section of the large organic ligand captures the energy and funnels it to the emissive state. The result is a material that exhibits strong green or red phosphorescence in the solid state, with tunable emission characteristics based on the choice of halide ligands (F, Cl, Br, I) coordinated to the manganese.
How to Synthesize Phosphorescent Manganese Complex Efficiently
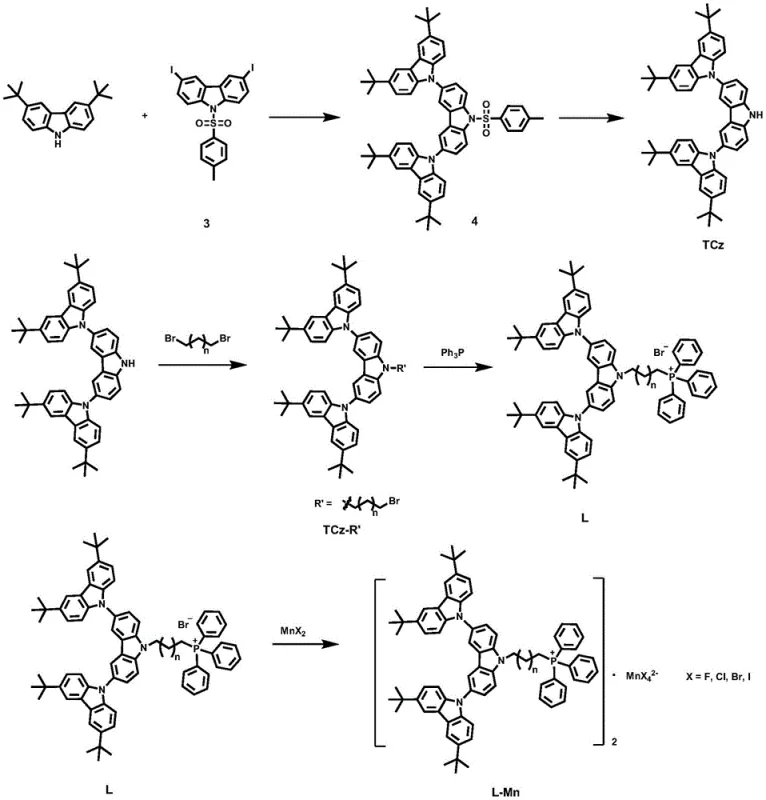
The preparation of this advanced material follows a logical, five-step sequence that is amenable to scale-up. The process leverages well-established organic transformations, minimizing the need for exotic reagents or extreme conditions. The initial Ullmann coupling builds the dendritic core, followed by a deprotection step to reveal the reactive nitrogen center. Subsequent alkylation introduces the linker arm, which is then quaternized with triphenylphosphine to generate the cationic ligand. The final step involves a simple stirring reaction with manganese bromide in dichloromethane at room temperature, yielding the target complex with high purity. This straightforward pathway reduces the technical barriers to entry for manufacturers looking to adopt this new class of emitters.
- Perform Ullmann coupling of 3,6-diiodo-N-tosylcarbazole with 3,6-di-tert-butylcarbazole using copper powder and K2CO3 in nitrobenzene at 170°C.
- Execute deprotection of the tosyl group using KOH in a DMSO-THF-H2O solvent system to yield the tricarbazole core (TCz).
- Conduct alkylation of TCz with dibromoalkane and t-BuOK in THF to introduce the phosphonium precursor chain.
- React the bromo-intermediate with triphenylphosphine (PPh3) in toluene at 120°C to form the cationic ligand L.
- Coordinate the ligand L with MnBr2·4H2O in dichloromethane at room temperature to finalize the phosphorescent manganese complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this manganese-based technology offers compelling strategic benefits beyond mere performance metrics. The primary advantage is the drastic reduction in raw material risk. Unlike iridium, which is mined in limited quantities and subject to geopolitical tension, manganese is one of the most abundant elements in the Earth's crust. This abundance translates directly into price stability and long-term supply security, essential factors for planning multi-year production cycles in the display industry. Additionally, the synthetic route avoids the use of expensive palladium catalysts often required for cross-coupling reactions in competitor technologies, further driving down the Bill of Materials (BOM) cost. The simplicity of the purification process, which relies on precipitation and filtration rather than column chromatography, significantly reduces solvent consumption and waste generation, aligning with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The elimination of precious metals from the emitter formulation results in substantial cost savings per kilogram of material produced. Since the manganese salt is inexpensive and the organic ligands are synthesized from readily available carbazole derivatives, the overall production cost is a fraction of that for iridium-based counterparts. Furthermore, the high yield of the final complexation step and the ease of isolation mean that manufacturing throughput can be increased without proportional increases in operational expenditure. This economic efficiency allows display manufacturers to produce high-quality panels at a more competitive price point, enhancing market positioning.
- Enhanced Supply Chain Reliability: Diversifying the supply base away from single-source noble metal suppliers mitigates the risk of production stoppages. The precursors for this manganese complex, such as 3,6-di-tert-butylcarbazole and triphenylphosphine, are commodity chemicals available from multiple global vendors. This redundancy ensures that supply disruptions from any single source do not halt the entire manufacturing line. Moreover, the stability of the final complex under ambient conditions simplifies logistics and storage requirements, reducing the need for specialized cold-chain shipping or inert atmosphere handling that is often mandatory for sensitive organometallic reagents.
- Scalability and Environmental Compliance: The synthetic protocol is designed with scalability in mind, utilizing solvents like nitrobenzene, THF, and dichloromethane which are standard in industrial organic synthesis. The reaction conditions, primarily reflux temperatures and room temperature stirring, are easily manageable in large-scale reactors without requiring high-pressure equipment. From an environmental perspective, the avoidance of heavy metal waste streams simplifies effluent treatment. The ability to recycle solvents and the reduced toxicity profile of manganese compared to other heavy metals contribute to a greener manufacturing footprint, helping companies meet their sustainability goals and comply with directives like RoHS and REACH.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphorescent manganese technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear picture of what partners can expect during technology transfer and scale-up.
Q: What are the advantages of manganese complexes over iridium-based OLED emitters?
A: Manganese complexes offer a significant cost advantage due to the earth-abundance of manganese compared to scarce noble metals like iridium. Additionally, the dendritic structure described in patent CN108409787B reduces concentration quenching, enhancing device stability and efficiency without requiring complex host-guest doping strategies.
Q: How does the dendritic structure improve electroluminescent performance?
A: The tricarbazole dendritic unit acts as both a hole transport group and a steric barrier. This architecture prevents the aggregation of emissive centers, thereby minimizing Triplet-Triplet Annihilation (TTA) and concentration quenching, which are common failure modes in traditional phosphorescent polymers.
Q: Is the synthesis of this manganese complex scalable for industrial production?
A: Yes, the synthesis utilizes standard organic reactions such as Ullmann coupling and nucleophilic substitution under relatively mild conditions. The purification steps involve straightforward precipitation and recrystallization, avoiding chromatography, which facilitates large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorescent Manganese Complex Supplier
As the demand for sustainable and cost-effective OLED materials accelerates, NINGBO INNO PHARMCHEM stands ready to support your transition to next-generation emitters. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN108409787B can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phosphorescent manganese complex meets the exacting standards required for high-performance display applications. Our commitment to quality assurance ensures consistent batch-to-batch reproducibility, a critical factor for maintaining yield in OLED panel fabrication.
We invite you to engage with our technical procurement team to discuss how this innovative material can optimize your product lineup. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to manganese-based emitters for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, securing a reliable supply of high-purity electronic chemicals for your future projects.
