Advanced Synthesis of Fluorescent Mononuclear Copper Complexes for High-Performance Optical Applications
The landscape of optoelectronic materials is undergoing a significant transformation, driven by the urgent need for cost-effective yet high-performance luminescent compounds. Patent CN110330527B introduces a groundbreaking advancement in this sector with the disclosure of a novel mononuclear copper complex exhibiting robust fluorescence properties. This innovation addresses critical bottlenecks in the manufacturing of display and lighting materials by replacing expensive noble metals with abundant copper while maintaining exceptional thermal stability and optical output. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards sustainable and economically viable production strategies for next-generation electronic chemicals. The detailed crystallographic data and synthetic methodology provided in the patent offer a clear roadmap for integrating these materials into commercial supply chains, ensuring reliability and consistency in high-volume manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of high-efficiency phosphorescent and fluorescent materials has relied heavily on iridium, platinum, or other precious metal complexes. These conventional pathways are fraught with significant economic and logistical challenges, primarily due to the exorbitant cost and geopolitical scarcity of the central metal ions. Furthermore, the synthetic routes for these noble metal complexes often require harsh reaction conditions, including high temperatures, inert atmospheres, and complex purification steps to remove toxic metal residues. Such rigorous requirements not only inflate the operational expenditure but also complicate the supply chain, leading to extended lead times and vulnerability to raw material shortages. Additionally, many existing copper-based alternatives have historically suffered from poor thermal stability or rapid degradation under operational conditions, limiting their practical utility in commercial devices where longevity is paramount.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110330527B presents a streamlined and highly efficient synthetic route that circumvents these traditional hurdles. By utilizing a specific benzimidazole phosphine ligand coordinated with cuprous chloride and triphenylphosphine, the inventors have achieved a four-coordinate distorted tetrahedral configuration that stabilizes the copper center effectively. This novel approach operates under remarkably mild conditions, requiring temperatures of only 40-50°C in a binary solvent system of dichloromethane and acetonitrile. The simplicity of the workup procedure, which involves mere filtration and natural volatilization for crystallization, drastically reduces processing time and energy consumption. This method not only lowers the barrier to entry for manufacturing but also ensures a high degree of reproducibility, making it an ideal candidate for the commercial scale-up of complex electronic chemicals.
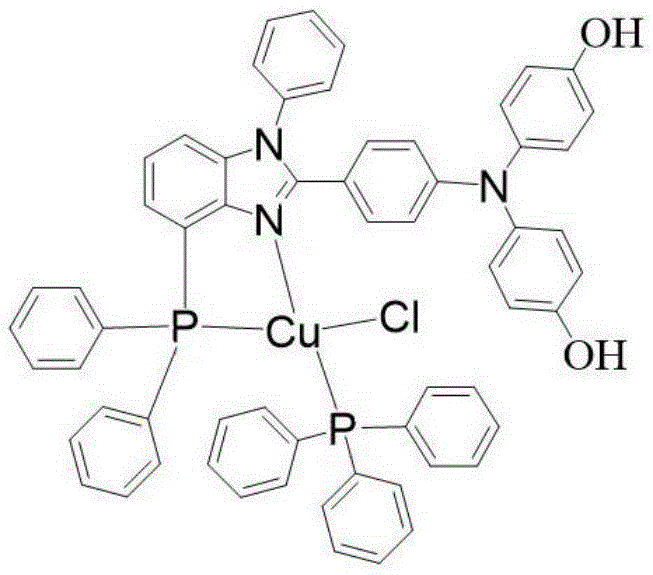
Mechanistic Insights into Cu(I) Coordination and Fluorescence
The core of this technological breakthrough lies in the precise molecular architecture of the resulting complex, designated as C61H47CuClN3O2P2. The central copper(I) ion is coordinated by a chlorine atom, nitrogen and phosphorus atoms from the benzimidazole phosphine ligand, and a phosphorus atom from the auxiliary triphenylphosphine ligand. This specific arrangement creates a rigid structural framework that minimizes non-radiative decay pathways, thereby enhancing fluorescence quantum yield. The crystal belongs to the triclinic system with space group P-1, and the unit cell parameters (a=12.73 Å, b=18.42 Å, c=23.22 Å) indicate a densely packed structure that contributes to its impressive thermal resilience. Understanding this coordination geometry is vital for R&D teams aiming to replicate or modify the structure for specific emission wavelengths, as slight perturbations in the ligand field can significantly alter the electronic properties of the metal center.
Furthermore, the photophysical mechanism is predominantly driven by Metal-to-Ligand Charge Transfer (MLCT), a phenomenon where electron density shifts from the copper d-orbitals to the pi-antibonding orbitals of the ligand upon excitation. The patent data specifies a strong emission peak at 603nm when excited at 400nm, placing the emission in the orange-red region of the visible spectrum. This specific spectral output is highly desirable for full-color display applications and solid-state lighting. The thermal stability, evidenced by the onset of weight loss only at 268.5°C, suggests that the complex can withstand the thermal stresses encountered during device fabrication processes such as vacuum deposition. For quality control managers, this robustness translates to lower rejection rates and higher yield consistency, reinforcing the material's viability as a high-purity OLED material precursor.
How to Synthesize Mononuclear Copper Complex Efficiently
The preparation method described in the patent is designed for operational simplicity without compromising on the purity or crystallinity of the final product. The process leverages the high solubility of the precursors in the chosen solvent system to facilitate rapid coordination kinetics. By strictly controlling the molar ratios and temperature, the reaction avoids the formation of polynuclear byproducts that often plague copper chemistry. The following section outlines the standardized protocol derived directly from the patent examples, serving as a foundational guide for laboratory replication and process engineering validation. Detailed standardized synthesis steps are provided in the guide below.
- Combine cuprous chloride, benzimidazole phosphine ligand, and triphenylphosphine in a dichloromethane and acetonitrile mixed solvent system.
- Heat the reaction mixture to 40-50°C and maintain continuous stirring for 2-3 hours to ensure complete coordination.
- Filter the solution, allow natural volatilization for 72-96 hours to grow crystals, then wash and dry under vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-based technology offers profound strategic advantages over legacy noble metal systems. The primary benefit stems from the substitution of scarce and volatile precious metals with copper, a base metal with a stable and abundant global supply chain. This fundamental shift in raw material sourcing mitigates the risk of price spikes and supply disruptions, ensuring a more predictable cost structure for long-term production planning. Moreover, the mild reaction conditions eliminate the need for specialized high-temperature reactors or extensive safety protocols associated with hazardous reagents, thereby reducing capital expenditure on equipment and lowering overall operational overheads.
- Cost Reduction in Manufacturing: The economic implications of switching to this copper complex are substantial, driven largely by the elimination of expensive iridium or platinum precursors. Since the synthesis utilizes readily available cuprous chloride and triphenylphosphine, the raw material costs are drastically simplified and reduced. Additionally, the energy efficiency of the process, operating at merely 40-50°C, results in significantly lower utility costs compared to high-temperature syntheses. The straightforward purification via crystallization further minimizes solvent usage and waste treatment expenses, contributing to a leaner and more cost-effective manufacturing model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Reliance on a reliable electronic chemical supplier is contingent upon the stability of the upstream raw material market. Copper and its common salts are commodities with well-established global distribution networks, unlike the niche supply chains for organometallic noble metal precursors. This abundance ensures that production schedules are not held hostage by raw material shortages. Furthermore, the simplicity of the synthesis allows for flexible manufacturing locations, reducing logistics complexity and reducing lead time for high-purity electronic chemicals. This resilience is critical for maintaining continuous operation in high-volume industries where downtime is financially catastrophic.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower, aligning with increasingly stringent global regulations on industrial emissions and waste. The absence of heavy metal contaminants simplifies effluent treatment, and the high atom economy of the coordination reaction reduces chemical waste generation. From a scalability perspective, the lack of extreme pressure or temperature requirements means that existing standard chemical processing infrastructure can be utilized for scale-up. This ease of translation from bench to plant accelerates time-to-market for new products and ensures that cost reduction in electronic chemical manufacturing is sustainable over the long term without compromising environmental standards.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the performance and handling of this mononuclear copper complex. These insights are derived directly from the experimental data and characterization results presented in the patent documentation, providing a factual basis for assessing its suitability for your specific application requirements. The following questions address key concerns regarding stability, optical performance, and process adaptability.
Q: What is the thermal stability profile of this copper complex?
A: Thermogravimetric analysis indicates the complex maintains structural integrity up to 268.5°C before the organic ligand begins to decompose, demonstrating superior thermal stability for device integration.
Q: What are the fluorescence emission characteristics?
A: Under an excitation wavelength of 400nm at 293K, the complex exhibits a strong emission peak at 603nm, attributed to Metal-to-Ligand Charge Transfer (MLCT).
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes mild temperatures (40-50°C), common solvents, and a simple filtration-crystallization workup, making it highly amenable to commercial scale-up without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mononuclear Copper Complex Supplier
As the demand for advanced optical materials continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass market availability is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of material performs consistently in your final devices. We understand the critical nature of supply continuity in the electronics sector and are committed to delivering high-quality intermediates that meet the exacting standards of the global industry.
We invite you to engage with our technical team to explore how this innovative copper complex can optimize your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits specific to your operation. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both technological advancement and financial growth.
