Advanced Selective Oxidation Strategies for High-Purity Indole Derivatives and Commercial Scale-Up
Advanced Selective Oxidation Strategies for High-Purity Indole Derivatives and Commercial Scale-Up
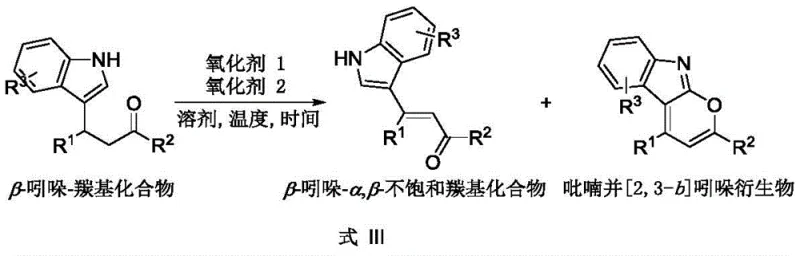
The landscape of organic synthesis for complex heterocyclic scaffolds is constantly evolving, driven by the need for more efficient and selective pathways in the production of high-value pharmaceutical intermediates. A significant breakthrough in this domain is detailed in patent CN112812122B, which discloses a versatile method for the selective synthesis of beta-indole-alpha,beta-unsaturated carbonyl compounds and pyrano[2,3-b]indole derivatives. This technology leverages a sophisticated oxidant-controlled strategy to diverge from a common beta-indole-carbonyl precursor, offering manufacturers unprecedented flexibility in accessing two distinct classes of bioactive scaffolds. By utilizing specific combinations of oxidants such as DDQ and TEMPO salts, the process achieves high conversion rates under mild conditions, addressing critical pain points related to reaction severity and product purity that have long plagued traditional synthetic routes. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for fine chemical intermediates.
![General structural formulas of beta-indole-alpha,beta-unsaturated carbonyl compounds and pyrano[2,3-b]indole derivatives](/insights/img/selective-indole-synthesis-pharma-intermediate-supplier-20260303211301-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha,beta-unsaturated carbonyl systems and fused indole scaffolds has relied heavily on multi-step sequences that often involve harsh reaction conditions, expensive transition metal catalysts, or toxic reagents. Conventional approaches to beta-disubstituted alpha,beta-unsaturated carbonyl compounds frequently suffer from poor regioselectivity and limited substrate scope, restricting their utility in the discovery of new drug candidates. Furthermore, existing methods for synthesizing pyrano[2,3-b]indole cores, which are prevalent in natural products and fluorescent probes, often exhibit low conversion rates and require rigorous purification steps to remove metal residues. These inefficiencies translate directly into increased operational costs and extended lead times for reliable pharmaceutical intermediate suppliers, creating bottlenecks in the supply chain for downstream API manufacturing. The reliance on noble metals also introduces significant environmental compliance challenges regarding waste disposal and heavy metal limits in final drug substances.
The Novel Approach
The methodology presented in the patent data introduces a streamlined, oxidant-driven divergence that fundamentally simplifies the synthetic landscape for these valuable motifs. By employing a single beta-indole-carbonyl starting material, the pathway can be toggled between two distinct products simply by adjusting the oxidant composition, eliminating the need for entirely different synthetic routes. The use of organic oxidants like DDQ (2,3-Dichloro-5,6-dicyano-1,4-benzoquinone) and TEMPO tetrafluoroborate allows the reaction to proceed at ambient temperatures, drastically reducing energy consumption compared to thermal cyclization methods. This approach not only enhances the atom economy of the process but also significantly simplifies the post-reaction workup, as the byproducts are generally easier to separate than metal complexes. Such innovation supports substantial cost savings in electronic chemical manufacturing and pharma sectors by reducing the complexity of the production workflow.
Mechanistic Insights into Oxidant-Controlled Divergent Synthesis
The core of this technological advancement lies in the precise manipulation of oxidative potential to direct the reaction trajectory. When DDQ is used as the sole oxidant, it facilitates a dehydrogenative process that generates the conjugated double bond system characteristic of the beta-indole-alpha,beta-unsaturated carbonyl compound (Formula I). This transformation likely proceeds via a hydride abstraction mechanism followed by elimination, preserving the indole nitrogen while establishing the exocyclic unsaturation. Conversely, the introduction of a second oxidant, specifically TEMPO+BF4-, alters the electronic environment sufficiently to promote an intramolecular oxidative cyclization. In this scenario, the oxygen atom of the carbonyl side chain attacks the electron-deficient position on the indole ring, facilitated by the synergistic action of the dual oxidant system, to close the pyran ring and form the pyrano[2,3-b]indole derivative (Formula II). This mechanistic understanding is crucial for process chemists aiming to optimize reaction parameters for specific substrates.
From an impurity control perspective, the mild nature of these oxidative conditions plays a vital role in maintaining high product quality. Traditional acid or base-catalyzed condensations often lead to polymerization or degradation of the sensitive indole nucleus, generating complex impurity profiles that are difficult to characterize and remove. In contrast, the neutral to slightly acidic conditions provided by the DDQ/TEMPO system minimize side reactions such as over-oxidation or ring-opening. The high selectivity observed in the experimental data, with yields reaching up to 92% for certain substrates, indicates a clean reaction profile that reduces the burden on downstream purification units. For quality assurance teams, this translates to a more robust process capable of consistently meeting stringent purity specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize Beta-Indole Derivatives Efficiently
Implementing this selective synthesis protocol in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and solvent choice to maximize the desired outcome. The general procedure involves dissolving the beta-indole-carbonyl precursor in a chlorinated solvent such as dichloromethane, followed by the sequential addition of the oxidant system. For the unsaturated carbonyl product, a 1:1 molar ratio of substrate to DDQ is typically sufficient, whereas the cyclized pyranoindole product benefits from the addition of 1.5 equivalents of TEMPO+BF4- alongside the DDQ. The reaction progress is rapid, often reaching completion within minutes at room temperature, which allows for high-throughput screening of various R-group substitutions. Detailed standardized operating procedures for scaling this chemistry are essential for ensuring reproducibility across different batches.
- Prepare the reactor by adding the beta-indole-carbonyl precursor and the selected oxidant system (either DDQ alone or DDQ combined with TEMPO+BF4-) into an appropriate organic solvent such as dichloromethane.
- Stir the reaction mixture at a controlled temperature ranging from 0 to 60 degrees Celsius for a duration between 1 to 300 minutes, monitoring the conversion until the starting material is fully consumed.
- Quench the reaction with aqueous sodium carbonate, perform liquid-liquid extraction with ethyl acetate, dry the organic phase over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxidant-controlled synthesis offers tangible benefits that extend beyond mere chemical elegance. The elimination of transition metal catalysts removes the need for expensive scavenging resins and extensive testing for residual metals, which are significant cost drivers in the production of high-purity OLED material and pharmaceutical intermediates. Furthermore, the ability to access two distinct product families from a common intermediate enhances supply chain resilience, allowing manufacturers to pivot production based on market demand without retooling entire production lines. The mild reaction conditions also imply lower energy overheads and reduced safety risks associated with high-pressure or high-temperature operations, contributing to a safer and more sustainable manufacturing environment. These factors collectively drive down the total cost of ownership for these critical building blocks.
- Cost Reduction in Manufacturing: The process utilizes commercially available organic oxidants that are generally less expensive than noble metal catalysts like palladium or rhodium, leading to direct material cost optimization. Additionally, the simplified workup procedure, which involves standard aqueous quenching and extraction, reduces the consumption of specialized purification media and solvents. By avoiding the generation of heavy metal waste, facilities can also realize significant savings in waste treatment and environmental compliance fees. The high yields reported in the patent examples further ensure that raw material utilization is maximized, minimizing the financial impact of starting material loss.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to variations in utility supply, such as steam pressure or cooling water temperature, ensuring consistent batch-to-batch quality. The wide substrate scope demonstrated in the patent data suggests that the method can accommodate various functional groups without requiring bespoke optimization for each new derivative. This flexibility allows suppliers to maintain a broader inventory of intermediates with greater confidence, reducing the risk of stockouts for downstream clients. Moreover, the short reaction times enable faster turnaround from order to shipment, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram scale is straightforward due to the absence of exothermic hazards typically associated with strong mineral acids or reactive organometallics. The use of dichloromethane, while requiring proper containment, is a well-understood solvent in industrial settings with established recovery protocols. The overall green chemistry profile is improved by the high atom efficiency and the avoidance of toxic metal residues, aligning with increasingly strict global regulations on pharmaceutical manufacturing emissions. This ensures long-term viability of the supply source without the risk of regulatory shutdowns or costly process modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this selective oxidation technology. Understanding these details is vital for process development teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: How is selectivity controlled between the unsaturated carbonyl and the pyranoindole skeleton?
A: Selectivity is precisely governed by the oxidant system employed. Using DDQ alone favors the formation of the beta-indole-alpha,beta-unsaturated carbonyl compound, whereas combining DDQ with a second oxidant like TEMPO+BF4- drives the oxidative cyclization to form the pyrano[2,3-b]indole derivative.
Q: What are the typical reaction conditions required for this synthesis?
A: The process operates under remarkably mild conditions, typically utilizing dichloromethane as the solvent at temperatures between 0 and 60 degrees Celsius. Reaction times are short, often completing within 5 to 30 minutes for model substrates, which facilitates rapid throughput.
Q: Is this method suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to its simple operational procedure, absence of transition metal catalysts, and straightforward workup involving standard extraction and chromatography, making it ideal for cost reduction in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN112812122B for maintaining a competitive edge in the global fine chemicals market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify identity and assay. Whether you require the unsaturated carbonyl precursors or the cyclized pyranoindole scaffolds, our infrastructure is designed to support your volume requirements with unwavering consistency.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our available indole derivatives and to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in driving efficiency and quality in your supply chain.
