Advanced Metal-Free Catalysis for Trifluoromethyl Indole Acetonitrile Commercialization
Introduction to Breakthrough Indole Acetonitrile Technology
The landscape of fine chemical synthesis is constantly evolving, driven by the demand for more efficient, safer, and environmentally sustainable manufacturing processes. A significant advancement in this field is documented in patent CN113717090B, which introduces a novel preparation method for trifluoromethyl-containing all-carbon quaternary carbon center indole acetonitrile compounds. These molecules represent a critical class of intermediates with profound implications for both the pharmaceutical and agrochemical sectors. The incorporation of a trifluoromethyl group alongside a sterically demanding all-carbon quaternary center presents unique challenges in organic synthesis, yet these structural motifs are essential for enhancing the metabolic stability, lipophilicity, and biological activity of final active ingredients. This patent provides a robust solution to these synthetic hurdles, offering a pathway that bypasses traditional limitations associated with constructing such complex molecular architectures.
For R&D directors and procurement specialists, understanding the nuances of this technology is paramount. The method described leverages a metal-free Lewis acid catalytic system, specifically utilizing tris(pentafluorophenyl)borane, to facilitate the nucleophilic substitution of hydroxyl groups with cyano groups. This approach not only achieves exceptional yields but also aligns with modern green chemistry principles by avoiding the use of toxic inorganic cyanides. As a reliable agrochemical intermediate supplier, recognizing the potential of such innovations allows for strategic planning in supply chain development, ensuring access to high-purity building blocks that can accelerate the discovery and production of next-generation fungicides and pharmaceuticals.
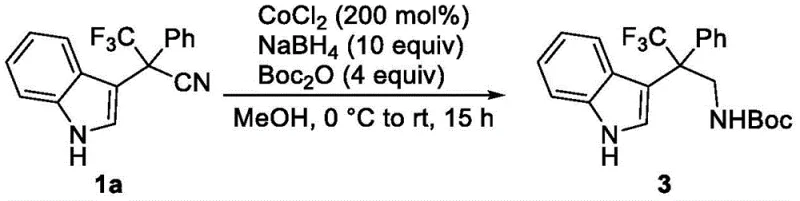
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole acetonitrile derivatives has relied heavily on nucleophilic substitution reactions involving halogenated precursors and inorganic cyanide salts such as sodium cyanide or potassium cyanide. While effective in certain contexts, these traditional methodologies suffer from severe drawbacks that impact both operational safety and economic efficiency. The use of inorganic cyanides poses significant toxicity risks, requiring stringent safety protocols, specialized containment equipment, and complex waste treatment procedures to neutralize hazardous byproducts. Furthermore, the construction of all-carbon quaternary centers via these methods is often plagued by steric hindrance, leading to sluggish reaction kinetics and poor yields. The necessity for harsh reaction conditions can also compromise the integrity of sensitive functional groups elsewhere in the molecule, limiting the scope of substrates that can be effectively utilized.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113717090B represents a paradigm shift in how these valuable intermediates are manufactured. By employing trifluoromethyl indole alcohol compounds as starting materials and trimethylsilyl cyanide (TMSCN) as the cyanide source, the process circumvents the hazards associated with inorganic cyanides. The reaction is catalyzed by tris(pentafluorophenyl)borane, a potent Lewis acid that activates the hydroxyl group for substitution under relatively mild thermal conditions. This metal-free catalytic system ensures that the final product is free from transition metal contamination, a critical quality attribute for pharmaceutical applications. The versatility of this approach is demonstrated by its compatibility with a wide range of substituents on the indole ring, allowing for the rapid generation of diverse chemical libraries.
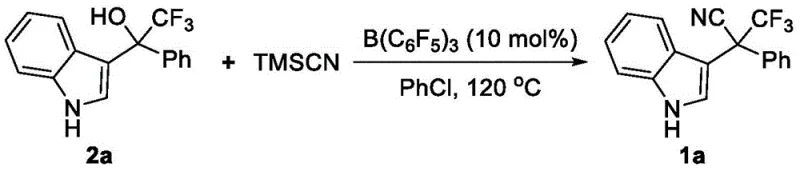
Mechanistic Insights into Lewis Acid Catalyzed Cyanation
The core of this technological breakthrough lies in the mechanistic efficiency of the tris(pentafluorophenyl)borane catalyst. As a strong Lewis acid, the boron center coordinates with the oxygen atom of the hydroxyl group in the trifluoromethyl indole alcohol substrate. This coordination significantly enhances the leaving group ability of the hydroxyl moiety, facilitating the formation of a carbocation intermediate stabilized by the adjacent indole ring and the electron-withdrawing trifluoromethyl group. Subsequently, the cyanide ion, released from the trimethylsilyl cyanide reagent, attacks this electrophilic center to form the new carbon-carbon bond. This mechanism is particularly advantageous for constructing sterically crowded quaternary centers, as the Lewis acid activation lowers the energy barrier for the substitution step, enabling reactions that would otherwise be kinetically prohibited.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or transition-metal-catalyzed pathways. The absence of metal catalysts eliminates the risk of metal-induced side reactions or residual metal impurities that often require expensive scavenging resins to remove. Furthermore, the use of TMSCN minimizes the formation of polymeric byproducts commonly associated with free cyanide ions. The reaction proceeds with high chemoselectivity, preserving other sensitive functional groups such as esters or halogens that might be present on the aromatic rings. This high level of control translates directly into simplified downstream processing, as the crude reaction mixtures are cleaner and easier to purify via standard silica gel chromatography, resulting in products with superior purity profiles suitable for sensitive biological applications.
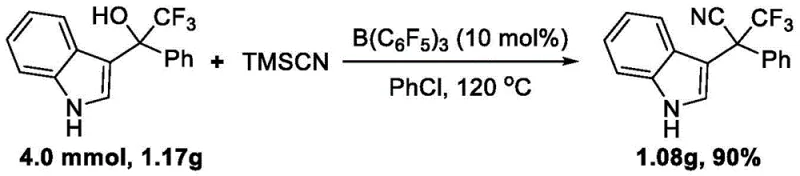
How to Synthesize Trifluoromethyl Indole Acetonitrile Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the precise weighing of the trifluoromethyl indole alcohol substrate and the tris(pentafluorophenyl)borane catalyst, typically used in a molar ratio ranging from 1:0.05 to 1:0.1. These components are dissolved in chlorobenzene, which has been identified as the optimal solvent for balancing solubility and reaction rate. Trimethylsilyl cyanide is then added, usually in a slight excess to drive the equilibrium towards product formation. The detailed standardized synthesis steps are provided in the guide below, outlining the specific temperatures, times, and workup procedures validated in the patent examples.
- Combine trifluoromethyl indole alcohol substrate with tris(pentafluorophenyl)borane catalyst in chlorobenzene solvent.
- Add trimethylsilyl cyanide (TMSCN) as the safe cyanide source and heat the mixture to 120°C.
- Monitor reaction progress via TLC, then purify the crude product using silica gel column chromatography to obtain high-purity target compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The elimination of toxic inorganic cyanides and transition metal catalysts fundamentally alters the cost structure and risk profile of manufacturing these intermediates. By removing the need for specialized hazardous waste disposal and expensive metal scavenging steps, the overall cost of goods sold can be significantly reduced. Additionally, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results. This reliability is crucial for maintaining a steady supply of critical raw materials for downstream drug or agrochemical production.
- Cost Reduction in Manufacturing: The metal-free nature of this catalytic system delivers substantial cost savings by obviating the need for precious metal catalysts and the associated purification infrastructure. Traditional methods often require palladium or copper catalysts, which are not only expensive to purchase but also necessitate rigorous removal processes to meet regulatory limits for residual metals in pharmaceutical ingredients. By utilizing an organoboron catalyst, manufacturers can streamline their purification workflows, reducing solvent consumption and processing time. Furthermore, the high yields reported in the patent, often exceeding 90%, minimize raw material waste, contributing to a more economical and sustainable production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The reagents employed in this process, such as trimethylsilyl cyanide and chlorobenzene, are commercially available and stable, ensuring a secure supply chain不受 geopolitical or logistical disruptions that often affect specialized reagents. The simplicity of the operation, which does not require inert atmosphere techniques beyond standard nitrogen protection, allows for execution in a wider range of manufacturing facilities. This flexibility reduces dependency on single-source suppliers for exotic catalysts and enables faster scaling of production capacity to meet market demand. Consequently, lead times for high-purity agrochemical intermediates can be drastically shortened, providing a competitive edge in fast-moving markets.
- Scalability and Environmental Compliance: The patent data explicitly validates the scalability of this method through successful gram-scale experiments, demonstrating that the reaction performance holds up when moving from milligram to multi-gram quantities. This proven scalability de-risks the technology transfer from laboratory to pilot plant and eventually to commercial production. Moreover, the avoidance of heavy metals and highly toxic cyanide salts aligns perfectly with increasingly stringent environmental regulations globally. Facilities adopting this greener chemistry approach can expect smoother regulatory approvals and reduced environmental compliance costs, positioning them as leaders in sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in patent CN113717090B, ensuring accuracy and relevance for decision-makers evaluating this technology for their supply chains.
Q: What are the safety advantages of this synthesis method compared to traditional cyanation?
A: Unlike conventional methods that utilize highly toxic inorganic cyanides like potassium cyanide, this patented process employs trimethylsilyl cyanide (TMSCN). This significantly reduces handling risks and simplifies waste treatment protocols, enhancing overall operational safety for manufacturing teams.
Q: Does this process require expensive transition metal catalysts?
A: No, the method utilizes tris(pentafluorophenyl)borane, a metal-free Lewis acid catalyst. This eliminates the need for costly transition metals and removes the complex downstream processing steps typically required to reduce residual metal impurities to ppm levels, thereby streamlining production.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent data demonstrates successful gram-scale amplification experiments maintaining high yields up to 90%. The use of robust reaction conditions and standard purification techniques like silica gel chromatography indicates strong feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Indole Acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes described in patent CN113717090B for the production of high-value fluorinated intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this metal-free catalysis are fully realized in a practical manufacturing setting. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific indole derivatives or bulk supply of standardized intermediates, our infrastructure is designed to support your growth from early-stage development to full-scale commercialization.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of fluorinated chemistry and secure a reliable source of high-quality intermediates for your next breakthrough product.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
