Revolutionizing N-Sulfonyl Pyrrolidine Synthesis via Advanced Gold Complex Catalysis for Commercial Scale-Up
Revolutionizing N-Sulfonyl Pyrrolidine Synthesis via Advanced Gold Complex Catalysis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery and development. A significant breakthrough in this domain is detailed in Chinese Patent CN113121472A, which discloses a novel method for preparing N-sulfonyl tetrahydropyrrole compounds utilizing a specialized gold complex catalyst. This technology represents a paradigm shift from traditional high-energy processes to mild, room-temperature catalysis, offering profound implications for the manufacturing of high-purity pharmaceutical intermediates. By leveraging an ortho-carborane Schiff base ligand coordinated with gold, this invention achieves exceptional reaction rates and yields while drastically reducing the catalyst loading required, addressing key pain points for both research and production teams globally.
For R&D directors and process chemists, the ability to conduct these transformations under ambient conditions without the need for aggressive heating or stoichiometric amounts of expensive metal salts is a game-changer. The patent highlights that the specific structural integrity of the o-carborane cage imparts unique stability and electronic properties to the gold center, enabling it to activate inert C-O bonds in tetrahydrofuran derivatives with unprecedented efficiency. This technical advancement not only streamlines the synthetic route but also opens new avenues for exploring complex molecular architectures that were previously difficult to access due to thermal instability of substrates. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge catalytic technologies is essential for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-sulfonyl pyrrolidine compounds has been fraught with significant operational challenges and economic inefficiencies that hinder large-scale production. Traditional methodologies often rely on the substitution reaction between sulfonyl chlorides and pyrrolidine, or the alkylation of sulfonamides with 1,4-dibromobutane, both of which generate substantial amounts of hazardous waste and require rigorous purification steps to remove halide byproducts. Furthermore, alternative approaches involving the direct reaction of sulfonamides with tetrahydrofuran derivatives have typically demanded harsh reaction conditions, specifically temperatures exceeding 100°C, to overcome the kinetic barrier of C-O bond cleavage. These high-temperature protocols not only consume excessive energy but also pose safety risks and can lead to the decomposition of sensitive functional groups on the substrate, thereby limiting the scope of applicable starting materials. Additionally, prior art methods frequently necessitate the use of large equivalents of silver salts alongside gold catalysts to facilitate the reaction, which dramatically inflates the raw material costs and complicates the downstream removal of heavy metal residues from the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology described in patent CN113121472A introduces a streamlined, one-pot catalytic system that operates effectively at room temperature. The core innovation lies in the utilization of a gold complex featuring an ortho-carborane Schiff base ligand, which acts as a highly efficient promoter for the coupling of sulfonamides and tetrahydrofuran compounds. This novel approach eliminates the need for external heating sources, allowing the reaction to proceed smoothly at ambient temperatures (rt) within a timeframe of 6 to 10 hours, which significantly reduces the energy footprint of the manufacturing process. Moreover, the catalyst loading is remarkably low, ranging from 0.005 to 0.01 molar equivalents relative to the substrate, which translates to substantial cost savings on precious metal usage compared to conventional protocols that require stoichiometric or near-stoichiometric amounts of metal additives. The versatility of this system is further evidenced by its compatibility with a wide array of substrates, including various substituted benzenesulfonamides and functionalized tetrahydrofurans, ensuring robust performance across diverse chemical landscapes.
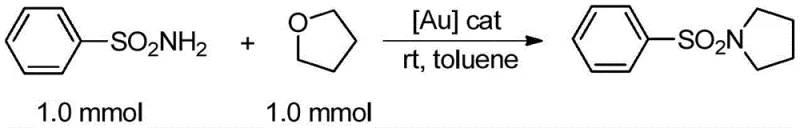
The general reaction scheme illustrates the elegance of this transformation, where the gold complex facilitates the nucleophilic attack of the sulfonamide nitrogen onto the activated tetrahydrofuran ring, resulting in the formation of the N-sulfonyl pyrrolidine scaffold with high atom economy. This level of efficiency and selectivity is critical for cost reduction in fine chemical manufacturing, as it minimizes the formation of side products and simplifies the isolation of the target molecule. By adopting this advanced catalytic strategy, manufacturers can achieve higher throughput and purity levels while adhering to increasingly stringent environmental regulations regarding waste disposal and energy consumption.
Mechanistic Insights into Ortho-Carborane Gold Complex Catalysis
The exceptional catalytic performance observed in this system can be attributed to the unique electronic and steric environment created by the ortho-carborane Schiff base ligand surrounding the gold center. The o-carborane cage is a three-dimensional aromatic cluster known for its extreme thermal and chemical stability, as well as its strong electron-withdrawing character, which modulates the Lewis acidity of the coordinated gold atom. This modulation enhances the ability of the gold complex to coordinate with the oxygen atom of the tetrahydrofuran ring, thereby weakening the adjacent C-O bond and rendering it susceptible to nucleophilic attack by the sulfonamide. Unlike simple phosphine or N-heterocyclic carbene ligands, the rigid and bulky nature of the carborane framework prevents the aggregation of gold species into inactive clusters, ensuring that the catalyst remains homogeneous and active throughout the reaction duration. This stability is further corroborated by the fact that the catalyst can be prepared via a simple one-pot procedure involving the condensation of o-carborane dicarbaldehyde with arylamines followed by metallation with AuBr, yielding a robust complex that is insensitive to air and moisture.
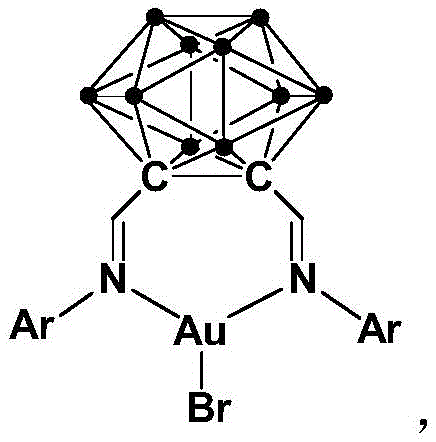
From an impurity control perspective, the high selectivity of this catalyst is paramount for producing high-purity OLED material or pharmaceutical intermediates where trace metal contamination is strictly regulated. The mechanism proceeds through a well-defined catalytic cycle where the gold center activates the ether substrate without promoting competing side reactions such as polymerization or over-oxidation, which are common pitfalls in acid-catalyzed ether cleavages. The specific geometry of the ligand, as depicted in the structural formula, creates a protective pocket around the metal center that guides the approach of the nucleophile, ensuring regioselective ring opening of the tetrahydrofuran. This precise control over the reaction pathway results in cleaner reaction profiles, reducing the burden on downstream purification units and enhancing the overall yield of the desired N-sulfonyl pyrrolidine product. For quality assurance teams, this means a more consistent product batch-to-batch with a simplified impurity profile that is easier to characterize and control.
How to Synthesize N-Sulfonyl Pyrrolidine Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to both laboratory scale optimization and industrial scale-up. The process begins with the in situ or pre-formed preparation of the gold complex, followed by its introduction to a mixture of the sulfonamide and tetrahydrofuran substrate in a non-polar solvent such as toluene. The reaction is allowed to stir at room temperature, monitoring progress via TLC or HPLC until completion, typically achieved within 8 hours for most substrates. The simplicity of the work-up procedure, which involves concentration and column chromatography, further underscores the operational ease of this method compared to multi-step traditional syntheses. For detailed standard operating procedures and specific parameter adjustments for different substrates, please refer to the standardized synthesis guide below.
- Prepare the catalyst by reacting ortho-carborane dicarbaldehyde with arylamine in toluene at 60-100°C, followed by the addition of AuBr at room temperature.
- Dissolve the sulfonamide compound and tetrahydrofuran derivative in toluene solvent within a reaction vessel.
- Add the gold complex catalyst (0.005-0.01 molar ratio) to the mixture and stir at room temperature for 6-10 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this gold complex catalysis technology offers tangible strategic benefits that extend beyond mere technical superiority. The shift to room temperature operations fundamentally alters the cost structure of production by eliminating the need for energy-intensive heating infrastructure and reducing the cooling loads associated with exothermic quenches. Furthermore, the drastic reduction in catalyst loading means that the consumption of expensive gold resources is minimized, directly impacting the bill of materials in a positive way. Since the catalyst is highly stable and can be synthesized from readily available precursors like o-carborane dicarbaldehyde and simple anilines, the supply chain for the catalyst itself is robust and less prone to disruption compared to exotic ligand systems. This reliability ensures continuous production schedules and mitigates the risk of delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The elimination of high-temperature requirements and the significant decrease in catalyst loading lead to substantial cost savings in both energy and raw materials. By avoiding the use of stoichiometric silver salts and reducing the reaction time through enhanced catalytic activity, the overall processing costs are significantly lowered. The simplified purification process also reduces solvent consumption and waste treatment expenses, contributing to a leaner and more economical manufacturing model that enhances profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: The use of cheap and easily obtainable raw materials, such as commercially available sulfonamides and tetrahydrofurans, ensures a stable supply chain foundation. The robustness of the catalyst against air and moisture simplifies storage and handling logistics, reducing the need for specialized inert atmosphere equipment during transport and warehousing. This resilience translates to reduced lead time for high-purity pharmaceutical intermediates, allowing for faster response to market demands and more flexible inventory management strategies for global clients.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this process make it inherently scalable from gram to ton quantities without the engineering challenges associated with high-pressure or high-temperature reactors. The reduction in hazardous byproducts and heavy metal waste aligns with modern green chemistry initiatives, facilitating easier regulatory approval and environmental compliance. This sustainability advantage is increasingly becoming a key differentiator in vendor selection processes for multinational corporations committed to reducing their carbon footprint and environmental impact.
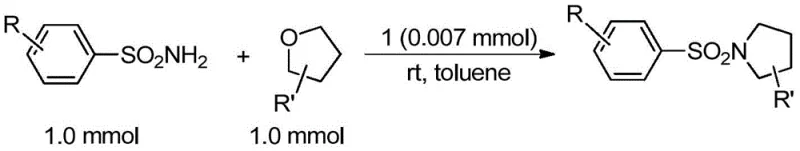
The broad substrate scope demonstrated in the patent data confirms that this platform technology is not limited to a single molecule but is applicable to a wide library of derivatives, enhancing its commercial value. The ability to tolerate various functional groups, including halogens, nitro groups, and methoxy groups, allows for the late-stage functionalization of complex molecules, which is highly desirable in medicinal chemistry campaigns. This versatility ensures that the manufacturing process remains relevant and adaptable as drug candidates evolve through the development pipeline.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial viability of this synthesis method, we have compiled a set of answers based on the detailed experimental data provided in the patent literature. These insights are intended to clarify the operational parameters and potential applications for partners considering this technology for their supply chain. Understanding these nuances is crucial for making informed decisions about process integration and resource allocation.
Q: What are the advantages of using the ortho-carborane gold complex over traditional silver/gold salts?
A: Unlike traditional methods requiring high temperatures (>100°C) and large equivalents of silver salts, this novel gold complex operates efficiently at room temperature with significantly lower catalyst loading, reducing energy consumption and metal waste.
Q: Is this catalytic system suitable for diverse sulfonamide substrates?
A: Yes, the patent demonstrates high substrate universality, successfully catalyzing reactions with various substituted benzenesulfonamides (including electron-donating and electron-withdrawing groups) and different tetrahydrofuran derivatives with high yields.
Q: How does this method impact the environmental profile of N-sulfonyl pyrrolidine production?
A: By eliminating the need for harsh reaction conditions and excessive metal additives, this method simplifies the work-up process, reduces solvent usage for purification, and minimizes heavy metal contamination in the final product, aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Sulfonyl Pyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ortho-carborane gold complex system in driving the next generation of pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-sulfonyl pyrrolidine intermediate delivered meets the highest global standards for safety and efficacy. We are committed to leveraging our technical expertise to optimize these gold-catalyzed routes for maximum efficiency and cost-effectiveness for our clients.
We invite you to collaborate with us to explore how this cutting-edge synthesis method can enhance your product portfolio and streamline your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term business goals in the competitive pharmaceutical landscape.
