Revolutionizing Benzothienopyrrole Manufacturing: A Mild, High-Yield Synthetic Route for Global Supply Chains
Revolutionizing Benzothienopyrrole Manufacturing: A Mild, High-Yield Synthetic Route for Global Supply Chains
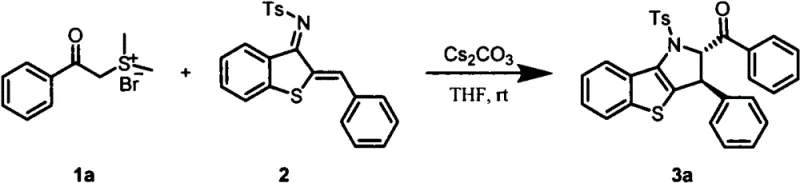
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex nitrogen-containing heterocyclic scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN114591348A introduces a groundbreaking synthetic methodology for generating benzothieno polysubstituted pyrrole skeletons, addressing key limitations in current manufacturing protocols. This innovation leverages a tandem cyclization reaction between thioaurone-derived unsaturated imines and sulfur ylides, catalyzed by cesium carbonate in tetrahydrofuran. By shifting away from harsh conditions and expensive transition metals, this technology offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks aiming to optimize their production portfolios. The method boasts exceptional operational simplicity, proceeding at room temperature with reaction times as short as 10 minutes, while delivering isolated yields approaching 93%. For R&D directors and procurement strategists, this represents a significant opportunity to enhance process robustness and reduce the overall cost of goods sold (COGS) for high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzothienopyrrole frameworks has relied heavily on transition metal catalysis or strong Brønsted acid/base systems that often necessitate rigorous exclusion of moisture and elevated temperatures. Traditional approaches frequently involve multi-step sequences with poor atom economy, leading to substantial waste generation and increased environmental compliance burdens. Furthermore, the reliance on precious metal catalysts introduces significant supply chain vulnerabilities, including price volatility and the complex regulatory requirements associated with residual metal limits in final API products. Previous methodologies, such as those involving guanidine catalysis or specific [4+1] cyclizations reported in earlier literature, often suffer from narrow substrate scope or require prolonged reaction times that hinder throughput. These inefficiencies create bottlenecks in commercial scale-up, where reproducibility and batch-to-batch consistency are paramount. Consequently, manufacturers face challenges in maintaining competitive pricing while adhering to stringent purity specifications required by global regulatory bodies.
The Novel Approach
The methodology disclosed in CN114591348A fundamentally reimagines this synthetic challenge by employing a mild, base-mediated tandem cyclization strategy. By utilizing cesium carbonate as a readily available inorganic base catalyst, the process eliminates the need for specialized ligands or toxic heavy metals, thereby streamlining the purification workflow. The reaction proceeds efficiently in tetrahydrofuran at ambient temperature, drastically reducing energy consumption compared to thermal reflux conditions. This novel approach not only accelerates the reaction kinetics—completing transformations in merely 10 minutes—but also demonstrates remarkable versatility across a range of sulfur ylide substrates, including those with electron-withdrawing chloro and bromo substituents as well as electron-donating methyl groups. For cost reduction in fine chemical manufacturing, this translates to lower utility costs, simplified waste treatment, and reduced raw material inventory holding times. The ability to achieve such high conversion rates under such mild conditions underscores the potential for seamless integration into existing continuous flow or batch processing facilities.
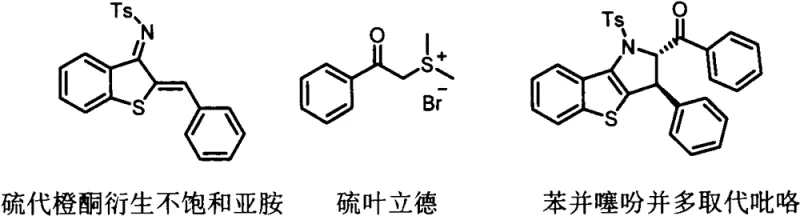
Mechanistic Insights into Base-Catalyzed Tandem Cyclization
The core of this technological advancement lies in the unique reactivity of the thioaurone-derived unsaturated imine acting as a four-carbon synthon in a [4+n] cyclization manifold. The mechanism initiates with the deprotonation of the sulfur ylide by the cesium carbonate base, generating a highly nucleophilic carbanion species. This nucleophile attacks the electrophilic carbon of the conjugated imine system, triggering a cascade of intramolecular rearrangements. The sulfur atom within the thioaurone skeleton plays a pivotal role, leveraging its nucleophilicity and the lability of the C-S bond under these specific conditions to facilitate ring closure. Unlike oxygen analogs, the sulfur atom allows for the construction of the fused benzothiophene system with high regioselectivity. The driving force for this transformation is partly attributed to the restoration of aromaticity within the newly formed pyrrole ring, providing a thermodynamic sink that pushes the equilibrium towards the product. Understanding this mechanistic pathway is crucial for process chemists aiming to troubleshoot potential side reactions or optimize stoichiometry for commercial scale-up of complex pharmaceutical intermediates.
From an impurity control perspective, the mildness of the cesium carbonate catalyst is instrumental in minimizing degradation pathways often seen with stronger bases or acids. The reaction environment is sufficiently gentle to preserve sensitive functional groups on the aryl rings of the sulfur ylide, such as halides, which are valuable handles for downstream diversification. The high selectivity observed suggests that competing nucleophilic attacks or polymerization of the reactive imine intermediate are effectively suppressed. This results in a cleaner crude reaction profile, which significantly reduces the burden on downstream purification units like chromatography columns or crystallization tanks. For quality assurance teams, this means a more consistent impurity profile and a higher probability of meeting strict specification limits for genotoxic impurities or residual solvents. The mechanistic elegance ensures that the process is not just a laboratory curiosity but a robust industrial protocol capable of delivering high-purity benzothienopyrrole derivatives consistently.
How to Synthesize Benzothieno Polysubstituted Pyrrole Efficiently
Implementing this synthesis in a pilot or production setting requires adherence to precise operational parameters to maximize yield and safety. The protocol is designed for ease of execution, requiring standard glassware and inert atmosphere techniques typical of modern organic synthesis laboratories. The key to success lies in the quality of the reagents, particularly the dryness of the tetrahydrofuran solvent and the activity of the cesium carbonate base. Operators should monitor the reaction progress closely using thin-layer chromatography (TLC) to confirm the rapid consumption of the starting imine, ensuring that the reaction is quenched or worked up immediately upon completion to prevent any potential decomposition. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible results.
- Dissolve thioaurone-derived unsaturated imine and cesium carbonate catalyst in anhydrous tetrahydrofuran (THF) under inert atmosphere, then add the sulfur ylide reagent.
- Stir the reaction mixture at room temperature for approximately 10 minutes until TLC monitoring indicates complete consumption of the starting material.
- Perform liquid-liquid extraction with dichloromethane and water, dry the organic phase, and purify the crude product via silica gel column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift to a base-catalyzed system using commodity chemicals like cesium carbonate and THF mitigates the risks associated with sourcing specialized catalysts that may have long lead times or single-source dependencies. This resilience is critical in maintaining reducing lead time for high-purity pharmaceutical intermediates in a volatile global market. Furthermore, the dramatic reduction in reaction time from hours or days to mere minutes significantly increases asset utilization rates, allowing manufacturing facilities to produce more batches per year without capital expansion. The simplified workup procedure, which avoids complex aqueous washes for metal removal, reduces the volume of hazardous waste generated, aligning with increasingly stringent environmental regulations and sustainability goals. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and ligands directly lowers the raw material cost per kilogram of the final product. Additionally, the ambient temperature operation removes the need for heating or cooling infrastructure, resulting in substantial energy savings. The high yields (87-93%) minimize material loss, ensuring that expensive starting materials are converted efficiently into value-added products. Qualitative analysis suggests that the simplified purification process reduces solvent consumption and labor hours associated with column chromatography or recrystallization, further driving down the operational expenditure.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including thioaurone derivatives and sulfur ylides, are synthesized from readily available precursors, ensuring a stable supply base. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in environmental conditions, leading to higher batch success rates. This reliability allows supply chain planners to maintain leaner inventory levels while still guaranteeing on-time delivery to downstream API manufacturers. The compatibility with standard equipment also means that the technology can be transferred to multiple contract manufacturing organizations (CMOs) without significant retrofitting costs.
- Scalability and Environmental Compliance: The mild nature of the reaction makes it inherently safer for scale-up, reducing the risk of thermal runaways or exothermic events common in aggressive chemical transformations. The use of THF, a solvent with well-established recovery and recycling protocols, supports green chemistry initiatives. By avoiding heavy metals, the facility reduces its liability regarding wastewater treatment and soil contamination, simplifying the permitting process for new production lines. The high atom economy of the tandem cyclization ensures that the majority of the reactant mass ends up in the product, minimizing the generation of by-products that require disposal.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process parameters and product specifications. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for conducting a thorough feasibility study and integrating this methodology into your existing R&D pipeline.
Q: What are the primary advantages of this Cs2CO3-catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and potentially toxic transition metal catalysts, utilizing inexpensive cesium carbonate instead. It operates at room temperature with significantly shorter reaction times (approx. 10 minutes), reducing energy consumption and simplifying downstream metal removal processes.
Q: What yields can be expected for the benzothienopyrrole derivatives using this protocol?
A: The patent data demonstrates consistently high isolated yields ranging from 87% to 93% across various substituted sulfur ylides, indicating robust reaction efficiency and excellent substrate tolerance for both electron-withdrawing and electron-donating groups.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the use of common solvents like THF, mild room temperature conditions, and short reaction times makes this process highly scalable. The simple workup procedure involving standard extraction and chromatography facilitates easy adaptation to kilogram or ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothieno Polysubstituted Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN114591348A for the production of advanced heterocyclic intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the benchtop to the reactor. Our state-of-the-art facilities are equipped to handle the specific solvent and reagent requirements of this cesium carbonate-catalyzed process, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-quality benzothieno polysubstituted pyrrole derivatives that meet the exacting standards of the global pharmaceutical industry, providing you with a secure and efficient source for your critical building blocks.
We invite you to engage with our technical team to explore how this innovative synthesis can be tailored to your specific molecular targets. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this mild catalytic system for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our superior manufacturing capabilities and deep technical expertise.
