Advanced CuI-Catalyzed Synthesis of Functionalized Thioether Intermediates for Pharmaceutical Scale-Up
Introduction to Patent CN110015981B
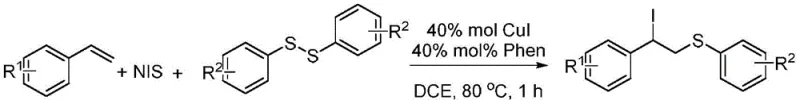
The landscape of organic synthesis intermediates is constantly evolving, driven by the need for more efficient, selective, and scalable methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN110015981B, which discloses a robust synthetic method for (2-iodo-2-aryl) ethyl aryl ether derivatives. This technology represents a pivotal advancement for the fine chemical industry, particularly for those seeking a reliable pharmaceutical intermediate supplier capable of delivering complex scaffolds. The core innovation lies in a transition metal-catalyzed three-component reaction that seamlessly integrates styrene derivatives, diaryl disulfides, and N-iodosuccinimide (NIS). Unlike traditional methods that struggle with selectivity, this approach leverages a copper(I) iodide catalytic system to achieve high yields under remarkably mild conditions. For R&D directors and procurement managers alike, this patent offers a compelling solution for accessing versatile building blocks that contain both sulfur and iodine functionalities, which are critical for subsequent nucleophilic substitutions or cross-coupling reactions in drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds via the addition of thiols or disulfides to alkenes has relied heavily on free radical oxidative coupling strategies. While conceptually straightforward, these conventional pathways suffer from significant drawbacks that hinder their utility in high-value manufacturing. Under typical radical oxidation conditions, the benzyl radical intermediate generated after the sulfur radical adds to the styrene derivative is highly susceptible to further oxidation. This lack of control frequently leads to the formation of undesirable byproducts such as β-hydroxyl sulfides, β-carbonyl sulfides, β-hydroxyl sulfones, and β-carbonyl sulfones. For a procurement manager focused on cost reduction in API manufacturing, these side reactions are disastrous, as they drastically reduce the overall yield and complicate the purification process, often requiring extensive chromatography that is not feasible on a multi-ton scale. Furthermore, introducing an iodine atom specifically at the beta-position using these oxidative methods is exceptionally difficult, limiting the downstream utility of the resulting thioethers for further functionalization.
The Novel Approach
In stark contrast to the limitations of radical oxidation, the methodology described in CN110015981B introduces a green, transition metal-catalyzed coupling reaction that effectively circumvents these issues. By employing a catalytic system comprising 40 mol% CuI and 40 mol% 1,10-phenanthroline (Phen) in 1,2-dichloroethane (DCE), the reaction achieves a highly selective three-component coupling. The process operates at a moderate temperature of 80 °C and reaches completion within just 1 h, demonstrating exceptional kinetic efficiency. This novel approach not only prevents the over-oxidation of the intermediate radicals but also ensures the precise installation of the iodine atom alongside the thioether moiety. The result is a clean reaction profile that delivers the target (2-iodo-2-aryl) ethyl aryl ether derivatives with considerable benefits in terms of purity and isolated yield. This shift from uncontrolled radical chemistry to a regulated metal-catalyzed process marks a substantial improvement in process reliability and scalability.
Mechanistic Insights into CuI-Catalyzed Three-Component Coupling
The success of this synthetic route can be attributed to the unique role of the copper catalyst in mediating the interaction between the disulfide, the alkene, and the iodine source. Mechanistically, the CuI/Phen complex likely facilitates the homolytic cleavage of the disulfide bond or activates the N-iodosuccinimide to generate the necessary reactive species without triggering the uncontrolled radical chain reactions seen in non-catalyzed systems. The presence of the phenanthroline ligand stabilizes the copper center, ensuring that the catalytic cycle proceeds efficiently to couple the sulfur and iodine fragments across the double bond of the styrene derivative. This controlled environment is crucial for maintaining the integrity of the sensitive alkyl iodide functionality, which might otherwise be lost to elimination or substitution side reactions in harsher conditions. For technical teams evaluating route feasibility, understanding this mechanism highlights the importance of the specific catalyst loading and ligand choice in achieving the reported high yields.
From an impurity control perspective, this mechanism offers a distinct advantage by suppressing the formation of oxygenated byproducts. In traditional radical pathways, the presence of trace oxygen or the inherent oxidizing nature of the reagents often leads to the aforementioned hydroxyl and carbonyl impurities. The CuI-catalyzed system appears to channel the reactivity exclusively towards the desired iodination and thiolation, effectively shutting down the oxidation pathways that lead to sulfones and ketones. This high level of chemoselectivity simplifies the downstream purification process significantly, as the crude reaction mixture contains fewer structurally similar impurities that are difficult to separate. Consequently, the final product meets stringent purity specifications with less intensive workup, directly translating to lower production costs and higher throughput for supply chain operations.
How to Synthesize (2-Iodo-2-Aryl) Ethyl Aryl Ether Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and industrial production. The procedure involves a straightforward one-pot reaction where all reagents are combined in a single vessel, eliminating the need for complex addition protocols or intermediate isolation steps. The standardized protocol utilizes a molar ratio of styrene derivative to diaryl disulfide to N-iodosuccinimide of 4:2:1, ensuring that the limiting reagent is fully consumed while minimizing waste. Following the reaction period of 1 h at 80 °C, the workup involves a standard extraction with ethyl acetate followed by solvent removal. The final purification is achieved via column chromatography using a gradient of petroleum ether and ethyl acetate, typically ranging from 5:1 to 30:1 depending on the polarity of the specific substituents. Detailed standardized synthesis steps are provided in the guide below.
- Charge a reaction vessel with styrene derivatives, diaryl disulfide, N-iodosuccinimide (NIS), cuprous iodide (40 mol%), 1,10-phenanthroline (40 mol%), and 1,2-dichloroethane (DCE).
- Seal the reaction tube and maintain magnetic stirring at 80 °C for 1 hour to ensure complete conversion.
- Upon completion, extract with ethyl acetate, concentrate under reduced pressure, and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and supply chain continuity, the adoption of this patented technology offers several strategic advantages that go beyond mere chemical novelty. The process is engineered to address common pain points in the manufacturing of complex organic intermediates, specifically regarding cost, scalability, and environmental compliance. By utilizing earth-abundant copper catalysts instead of precious metals like palladium or platinum, the raw material costs are inherently lower, and the supply risk associated with volatile precious metal markets is mitigated. Furthermore, the short reaction time and mild conditions reduce energy consumption and increase reactor turnover rates, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and readily available starting materials. Styrene derivatives and diaryl disulfides are commodity chemicals produced on a massive scale globally, ensuring a stable supply base and competitive pricing. The elimination of expensive transition metal catalysts and the reduction in reaction time from days to merely 1 h significantly lowers the operational expenditure (OPEX) associated with utilities and labor. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, thereby improving the overall atom economy and reducing the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical for pharmaceutical manufacturers who cannot afford interruptions in their raw material supply. This synthesis route relies on robust chemistry that tolerates a wide range of functional groups, meaning that variations in the quality of feedstock or the need to switch between different substituted analogs can be accommodated without re-validating the entire process. The simplicity of the workup and purification steps also reduces the dependency on specialized equipment or hazardous reagents that might face regulatory scrutiny or logistical bottlenecks. This flexibility ensures a consistent and reliable flow of high-purity intermediates, safeguarding the production schedules of downstream drug manufacturers.
- Scalability and Environmental Compliance: As the industry moves towards greener manufacturing practices, the environmental footprint of chemical processes is under increasing scrutiny. This CuI-catalyzed method aligns well with green chemistry principles by avoiding toxic heavy metals and minimizing waste generation through high-yield transformations. The use of 1,2-dichloroethane (DCE) is a known solvent, and its recovery and recycling can be easily integrated into existing solvent management systems. The ability to scale this reaction from gram quantities in the lab to multi-ton batches in the plant without significant loss of efficiency demonstrates its maturity and readiness for commercial deployment, reducing the risk and time associated with technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these details is essential for making informed decisions about process integration and long-term sourcing strategies.
Q: What are the key advantages of this CuI-catalyzed method over traditional radical oxidation?
A: Traditional radical oxidation often leads to over-oxidation byproducts like beta-hydroxyl or beta-carbonyl sulfides. This novel CuI-catalyzed approach selectively installs the iodine atom while forming the C-S bond, avoiding these difficult-to-remove impurities and significantly improving yield.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes inexpensive catalysts (CuI), common solvents (DCE), and operates under mild conditions (80 °C) with a short reaction time (1 h), making it highly amenable to commercial scale-up and cost-effective manufacturing.
Q: What is the substrate scope for this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents on both the styrene and disulfide components, including electron-withdrawing groups like nitro, halogens (F, Cl, Br), and alkyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2-Iodo-2-Aryl) Ethyl Aryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the CuI-catalyzed three-component reaction described in CN110015981B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to full-scale market launch. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, providing you with the confidence needed to advance your drug candidates.
We invite you to collaborate with us to optimize this synthetic route for your specific needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this method or need specific COA data to verify the quality of our pilot batches, our technical procurement team is ready to assist. Please contact us today to request route feasibility assessments and discuss how we can support your supply chain with reliable, cost-effective, and high-purity (2-iodo-2-aryl) ethyl aryl ether derivatives.
