Advanced CuI-Catalyzed Synthesis of Functionalized Iodo-Aryl Thioethers for Commercial Scale
Advanced CuI-Catalyzed Synthesis of Functionalized Iodo-Aryl Thioethers for Commercial Scale
The pharmaceutical and agrochemical industries are constantly seeking efficient, scalable routes to complex organic scaffolds that serve as critical building blocks for active ingredients. A significant breakthrough in this domain is detailed in patent CN110015981B, which discloses a novel synthetic method for preparing (2-iodo-2-aryl)ethyl aryl sulfide derivatives. This technology leverages a transition metal-catalyzed three-component coupling reaction involving styrene derivatives, diaryl disulfides, and N-iodosuccinimide (NIS). Unlike conventional methods that struggle with selectivity and over-oxidation, this approach utilizes a copper(I) iodide (CuI) catalytic system to achieve high-yielding bifunctionalization under remarkably mild conditions. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-purity organic synthesis intermediates.
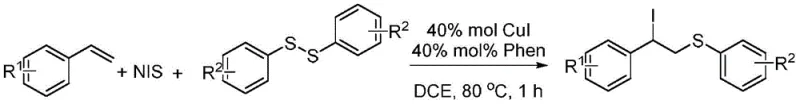
The core innovation lies in the simultaneous construction of carbon-sulfur and carbon-iodine bonds on the ethyl backbone of the styrene derivative. This dual functionalization creates a highly versatile intermediate; the alkyl iodide moiety serves as an excellent leaving group for subsequent nucleophilic substitutions or transition metal-catalyzed cross-coupling reactions, while the thioether linkage provides essential structural motifs found in numerous bioactive molecules. By enabling the direct transformation of readily available styrenes and disulfides into these valuable precursors, the patent offers a streamlined pathway that significantly reduces the step count compared to traditional multi-step syntheses. This efficiency is crucial for reducing lead time for high-purity API precursors in a competitive market environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organothioether compounds has relied heavily on radical oxidative coupling strategies involving thiophenols or diphenyl disulfides with alkenes. While conceptually straightforward, these conventional free-radical pathways suffer from significant drawbacks regarding selectivity and product distribution. Under typical radical oxidation conditions, the benzyl radical intermediate generated after the addition of a sulfur radical to the styrene double bond is highly susceptible to further oxidation by oxygen or other oxidants present in the system. This uncontrolled oxidation frequently leads to the formation of unwanted byproducts such as beta-hydroxy sulfides, beta-carbonyl sulfides, beta-hydroxy sulfones, and beta-carbonyl sulfones. Consequently, isolating the desired beta-iodo thioether becomes exceedingly difficult, often requiring extensive purification efforts that drive up costs and reduce overall process efficiency. Furthermore, many traditional methods require harsh conditions or expensive noble metal catalysts that are not ideal for large-scale commercial operations.
The Novel Approach
In stark contrast, the methodology described in patent CN110015981B introduces a green, transition metal-catalyzed solution that effectively circumvents the pitfalls of uncontrolled radical oxidation. By employing a specific catalytic system comprising 40 mol% CuI and 40 mol% 1,10-phenanthroline (Phen) in 1,2-dichloroethane (DCE), the reaction proceeds with exceptional regioselectivity and chemoselectivity. The process operates at a moderate temperature of 80°C and completes within just 1 hour, demonstrating a dramatic improvement in reaction kinetics compared to slower, less efficient legacy methods. This novel approach not only suppresses the formation of over-oxidized impurities but also ensures that the iodine atom is installed precisely at the benzylic position, creating a stable and reactive handle for downstream chemistry. For a reliable pharma intermediate supplier, adopting such a robust and rapid protocol translates directly into enhanced production capacity and consistent product quality.
Mechanistic Insights into CuI-Catalyzed Three-Component Coupling
The success of this synthesis hinges on the intricate interplay between the copper catalyst, the ligand, and the iodine source. Mechanistically, the CuI/Phen complex is believed to facilitate the homolytic cleavage of the disulfide bond or activate the N-iodosuccinimide to generate the necessary radical species in a controlled manner. Unlike uncontrolled free-radical processes, the presence of the copper center likely mediates the transfer of the sulfur and iodine groups to the alkene substrate through a coordinated cycle that stabilizes the reactive intermediates. This coordination prevents the benzyl radical from reacting with ambient oxygen or undergoing further oxidation to sulfones, thereby channeling the reaction pathway exclusively towards the desired (2-iodo-2-aryl)ethyl aryl sulfide product. The use of phenanthroline as a bidentate nitrogen ligand is critical, as it stabilizes the copper species and enhances its catalytic activity, allowing the reaction to proceed efficiently even with electron-deficient styrene substrates.
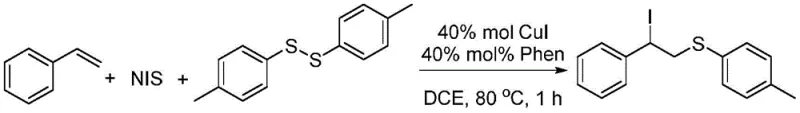
From an impurity control perspective, this mechanism offers substantial advantages for manufacturing high-purity organic synthesis intermediates. The suppression of beta-hydroxy and beta-carbonyl side products means that the crude reaction mixture is significantly cleaner, simplifying the downstream workup and purification processes. In the reported examples, simple extraction followed by column chromatography yields the target compounds with high purity, as evidenced by the clean NMR spectra provided in the patent data. For quality control teams, this implies a more predictable impurity profile and reduced risk of carrying over difficult-to-remove contaminants into the final API. The ability to tolerate a wide range of functional groups—including nitro, fluoro, chloro, and bromo substituents—without compromising the integrity of the catalytic cycle further underscores the robustness of this chemical transformation for diverse synthetic applications.
How to Synthesize (2-Iodo-2-aryl)ethyl Aryl Sulfides Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and requires standard equipment. The general procedure involves charging a reaction vessel with the styrene derivative, the diaryl disulfide, and N-iodosuccinimide in a molar ratio optimized for maximum conversion, typically around 4:2:1. The catalyst system, consisting of CuI and phenanthroline, is added along with the solvent 1,2-dichloroethane. The detailed standardized synthesis steps, including precise stoichiometry and workup parameters, are outlined below to ensure reproducibility and safety during scale-up operations.
- Combine styrene derivatives, diaryl disulfides, and N-iodosuccinimide (NIS) in 1,2-dichloroethane (DCE) with CuI and phenanthroline catalysts.
- Heat the reaction mixture to 80°C and stir magnetically for 1 hour to ensure complete conversion.
- Extract with ethyl acetate, concentrate under reduced pressure, and purify via column chromatography using petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CuI-catalyzed technology offers compelling economic and logistical benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; styrenes and diaryl disulfides are commodity chemicals available in bulk quantities from multiple global suppliers, ensuring supply continuity and mitigating the risk of single-source dependency. Furthermore, the elimination of expensive noble metal catalysts like palladium or platinum in favor of abundant copper significantly lowers the raw material cost base. This shift contributes to substantial cost savings in pharmaceutical manufacturing, allowing companies to maintain healthy margins even in price-sensitive markets. The short reaction time of just 1 hour also maximizes asset utilization, enabling manufacturing facilities to produce more batches per week and respond rapidly to fluctuating market demands.
- Cost Reduction in Manufacturing: The economic impact of switching to this copper-catalyzed protocol is profound. By replacing costly reagents and minimizing reaction times, the overall cost of goods sold (COGS) is significantly reduced. The use of inexpensive CuI and phenanthroline eliminates the need for expensive metal scavenging steps often required to meet strict residual metal limits in pharmaceutical products. Additionally, the high selectivity of the reaction reduces solvent consumption during purification, as less material is lost to side-product separation. These factors combine to create a leaner, more cost-efficient production process that enhances the competitiveness of the final intermediate in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable, shelf-stable reagents that do not require specialized storage conditions like cryogenic temperatures or inert atmospheres beyond standard practice. The robustness of the reaction conditions (80°C in DCE) means that the process is less sensitive to minor variations in operational parameters, leading to consistent batch-to-batch quality. This reliability is critical for maintaining uninterrupted production schedules for downstream API synthesis. Moreover, the broad substrate scope allows for flexibility in sourcing; if a specific substituted styrene becomes unavailable, alternative analogs can often be substituted with minimal process re-optimization, ensuring that production timelines remain intact.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the simplicity of the operation and the absence of hazardous exotherms typically associated with uncontrolled radical reactions. The use of 1,2-dichloroethane, while requiring proper handling, is a well-understood solvent in industrial chemistry with established recovery and recycling protocols. The high atom economy of the three-component coupling minimizes waste generation, aligning with modern green chemistry principles and environmental regulations. This ease of scale-up ensures that the commercial scale-up of complex thioethers can be achieved rapidly, bridging the gap between laboratory discovery and industrial production without the usual teething problems associated with process intensification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in patent CN110015981B, offering clarity on the practical aspects of adopting this technology for your specific manufacturing needs.
Q: What are the advantages of this CuI-catalyzed method over traditional radical oxidation?
A: Traditional radical oxidation often leads to over-oxidation byproducts like sulfones or carbonyl sulfides. This CuI-catalyzed method selectively installs the iodine and sulfur groups, preventing over-oxidation and ensuring high purity of the beta-iodo thioether product.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process utilizes inexpensive copper catalysts and common solvents like DCE. The short reaction time (1 hour) and mild temperature (80°C) make it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the substrate scope for this synthesis?
A: The method demonstrates excellent tolerance for various substituents on the styrene ring, including electron-withdrawing groups like nitro, fluoro, chloro, and bromo, allowing for diverse functionalization of the final thioether scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2-Iodo-2-aryl)ethyl Aryl Sulfides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our team of expert chemists has thoroughly analyzed the potential of the CuI-catalyzed three-component coupling described in CN110015981B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of (2-iodo-2-aryl)ethyl aryl sulfides we deliver meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your project timelines and enhance your product's market competitiveness. Let us be your partner in turning innovative chemical methodologies into commercial reality.
