Advanced S-Flurbiprofen Intermediate Manufacturing: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust manufacturing pathways for nonsteroidal anti-inflammatory drugs (NSAIDs), particularly for single-enantiomer formulations that offer superior therapeutic profiles. Patent CN113045532A discloses a groundbreaking methodology for preparing S-flurbiprofen, a potent analgesic where the S-enantiomer provides the primary cyclooxygenase inhibiting activity while minimizing gastrointestinal adverse effects associated with the R-isomer. This technical insight report analyzes the novel four-step synthesis route that bypasses the limitations of traditional enzymatic or resolution-based methods. By leveraging a chiral pool strategy using L-tartaric diesters, the disclosed process achieves exceptional optical purity and yield stability. The strategic implementation of this chemistry allows for the production of high-purity pharmaceutical intermediates suitable for global regulatory standards. 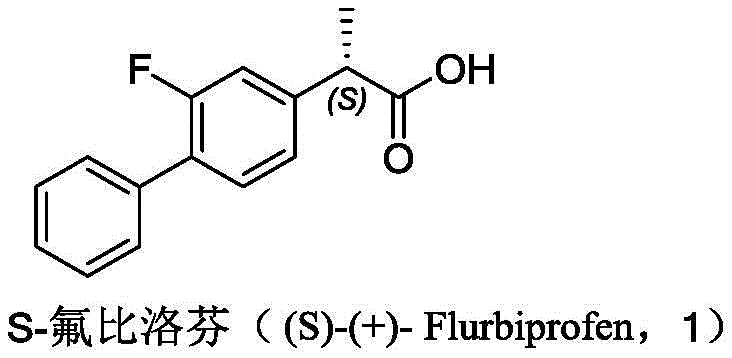
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of S-flurbiprofen has been plagued by significant economic and technical inefficiencies that hinder cost-effective mass production. Traditional routes often rely on the resolution of racemic flurbiprofen using chiral amines, a process inherently limited by a maximum theoretical yield of 50% for the desired isomer, necessitating complex recycling streams for the unwanted R-enantiomer. Alternative approaches involving enzymatic kinetic resolution, such as those utilizing CAL-A lipase, introduce substantial cost volatility due to the high price of biocatalysts and their sensitivity to reaction conditions. Furthermore, earlier asymmetric synthesis routes described in literature, such as those involving silver-catalyzed rearrangements or Grignard reactions followed by complex dehydration, often suffer from harsh reaction conditions, low overall yields, and the generation of hazardous waste streams. These legacy methods create bottlenecks in supply chain reliability and escalate the cost of goods sold (COGS) for downstream drug manufacturers. 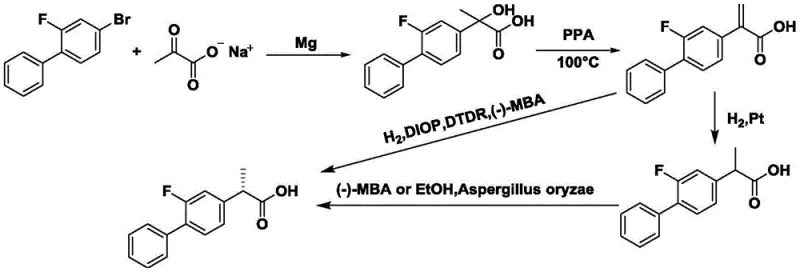
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by introducing a concise, four-step sequence that prioritizes atom economy and operational simplicity. This novel approach initiates with a condensation reaction between a biphenyl ketone and an L-tartaric diester, effectively installing the chiral center at the very beginning of the synthesis rather than attempting to separate it at the end. By avoiding the use of expensive enzymes or precious metal asymmetric catalysts, the process drastically reduces raw material costs and simplifies the procurement landscape for production facilities. The reaction conditions are mild, typically operating between 60°C to 110°C in common aromatic hydrocarbon solvents like toluene, which facilitates easier heat management and safety protocols during scale-up. This streamlined workflow not only enhances the total yield to over 54% with optical purity reaching 99.8% but also ensures that the process is amenable to continuous manufacturing environments.
Mechanistic Insights into L-Tartrate Mediated Chiral Induction
The core innovation of this synthesis lies in the stereoselective ketalization step where the prochiral ketone reacts with the chiral diester to form a rigid dioxolane intermediate. This transformation is catalyzed by sulfonic acids or inorganic acids, which promote the formation of a cyclic structure that locks the stereochemistry derived from the naturally abundant L-tartaric acid. The steric bulk of the tartrate moiety directs the subsequent bromination to occur with high diastereoselectivity, ensuring that the chiral information is faithfully transferred through the synthetic sequence. This mechanism eliminates the need for external chiral catalysts that often require rigorous removal processes to meet residual metal specifications in active pharmaceutical ingredients (APIs). The robustness of this chiral auxiliary approach means that minor fluctuations in reaction parameters do not compromise the enantiomeric excess, providing a stable quality profile essential for regulatory filings. 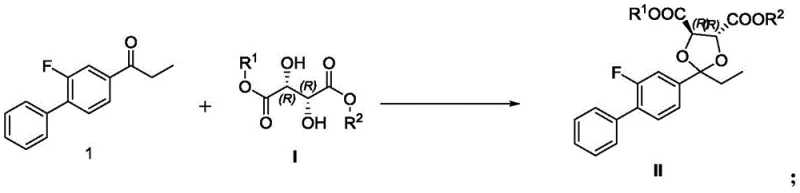
Following the establishment of the chiral center, the subsequent bromination and silver-mediated transfer reactions proceed with high fidelity to preserve the stereochemical integrity of the molecule. The use of silver tetrafluoroborate as a transfer catalyst in the presence of water facilitates a clean conversion of the bromo-intermediate into the corresponding ester without racemization. This step is critical as it sets up the final hydrolysis, where mild alkaline conditions cleave the auxiliary groups to release the free carboxylic acid. The impurity profile generated during these steps is significantly cleaner compared to racemic resolution, as the chiral pool starting material prevents the formation of the opposite enantiomer entirely. Consequently, downstream purification requirements are minimized, reducing solvent consumption and processing time while maximizing the recovery of the valuable S-enantiomer product.
How to Synthesize S-Flurbiprofen Efficiently
The synthesis protocol described in the patent offers a clear pathway for laboratory and pilot-scale operations to reproduce the high-yield results consistently. Operators should focus on maintaining strict control over the molar ratios of the tartaric diester and the acid catalyst during the initial condensation phase to ensure complete conversion of the ketone starting material. The subsequent bromination step requires careful temperature management, preferably keeping the reaction between 10°C to 30°C to prevent side reactions that could lower the diastereomeric excess. Detailed standardized operating procedures for the workup, including quenching with sodium thiosulfate and extraction protocols, are essential to isolate the intermediates with the purity required for the final coupling steps. For comprehensive technical guidance on scaling this route, please refer to the standardized synthesis steps provided in the section below.
- Perform acid-catalyzed ketalization of 1-(2-fluoro-[1,1'-biphenyl]-4-yl)propan-1-one with L-tartaric diester in toluene.
- Execute bromination of the condensed carbonyl compound using bromine or HBr/Acetic acid mixture under nitrogen protection.
- Conduct silver-catalyzed transfer reaction with water to form the chiral carboxylic ester intermediate.
- Finalize with alkaline hydrolysis using lithium hydroxide to obtain high-purity S-flurbiprofen.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing route offers compelling advantages that directly address the pain points of cost volatility and supply discontinuity in the pharmaceutical intermediate market. By eliminating the dependency on proprietary enzymes or scarce chiral ligands, the production cost structure becomes more predictable and resilient against market fluctuations in specialty reagents. The reliance on commodity chemicals such as toluene, bromine, and tartaric acid derivatives ensures that raw material availability remains high, reducing the risk of production stoppages due to supplier shortages. Furthermore, the simplified purification train reduces the consumption of chromatography media and solvents, leading to substantial cost savings in waste treatment and utility usage. These factors combine to create a highly competitive cost position for manufacturers adopting this technology, allowing for more aggressive pricing strategies in the global API market.
- Cost Reduction in Manufacturing: The exclusion of high-cost biocatalysts and precious metal complexes significantly lowers the direct material expenses associated with each batch production cycle. The process utilizes inexpensive chiral pool materials that are produced on a massive industrial scale globally, ensuring long-term price stability and avoiding the premiums associated with custom-synthesized chiral auxiliaries. Additionally, the high overall yield of the four-step sequence means that less starting material is wasted per kilogram of final product, further driving down the effective cost per unit. This economic efficiency allows procurement teams to negotiate better terms with suppliers who utilize this advanced methodology, resulting in improved margins for the final drug product.
- Enhanced Supply Chain Reliability: The use of widely available commodity reagents mitigates the risk of supply chain disruptions that often plague processes dependent on single-source specialty chemicals. Since the reaction conditions are not overly sensitive to moisture or oxygen compared to organometallic alternatives, the manufacturing process can be executed in a broader range of facilities without requiring specialized infrastructure. This flexibility increases the pool of qualified contract development and manufacturing organizations (CDMOs) capable of producing the intermediate, thereby diversifying supply sources and enhancing business continuity. The robustness of the chemistry ensures consistent lead times, enabling procurement planners to maintain leaner inventory levels without compromising production schedules.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scalability in mind, utilizing standard reactor equipment and avoiding extreme pressures or temperatures that would require capital-intensive hardware investments. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering the compliance burden and disposal costs for manufacturing sites. The absence of heavy metal catalysts simplifies the validation process for equipment cleaning and reduces the risk of cross-contamination in multi-purpose plants. These environmental and operational benefits make the technology attractive for long-term capacity planning, ensuring that supply can be ramped up rapidly to meet market demand without encountering regulatory or engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route for S-flurbiprofen intermediates. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these nuances is critical for evaluating the feasibility of technology transfer and the potential return on investment for adopting this new manufacturing standard. We encourage stakeholders to review these points carefully when assessing supplier capabilities and process robustness.
Q: How does this route improve optical purity compared to traditional resolution?
A: By utilizing L-tartaric diester as a chiral auxiliary during the initial ketalization step, the process establishes stereochemistry early, achieving up to 99.8% optical purity without the 50% yield loss inherent in racemic resolution.
Q: Are the reagents used in this patent commercially available for large-scale production?
A: Yes, the protocol relies on inexpensive and readily available starting materials such as biphenyl ketones and tartaric esters, avoiding specialized enzymes or precious metal asymmetric catalysts.
Q: What are the environmental benefits of this synthesis method?
A: The process generates significantly less three-waste compared to prior art, operates under mild temperatures, and eliminates the need for complex heavy metal removal steps, ensuring better environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Flurbiprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic routes like the one described in CN113045532A to maintain competitiveness in the global pharmaceutical supply chain. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of S-flurbiprofen intermediate meets the highest international quality standards. We are committed to leveraging our technical expertise to optimize this chiral pool synthesis, delivering consistent quality and reliability to our partners.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent can enhance your supply chain efficiency. By collaborating with us, you gain access to a secure, scalable, and cost-effective source of high-value pharmaceutical intermediates that drive your product success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
