Advanced One-Pot Synthesis of Electrophilic Perfluoroalkylating Reagents for Industrial Scale
Introduction to Next-Generation Electrophilic Fluorination Technology
The introduction of patent CN107540655B marks a significant paradigm shift in the industrial preparation of electrophilic perfluoroalkylating agents, specifically S-(perfluoroalkyl)-dibenzothiophene triflates. These compounds, often referred to as Umemoto-type reagents, are critical tools for introducing trifluoromethyl and other perfluoroalkyl groups into complex organic scaffolds, a transformation essential for enhancing the metabolic stability and lipophilicity of modern pharmaceuticals and agrochemicals. Traditionally, the synthesis of these valuable reagents has been plagued by multi-step procedures, harsh conditions, and the generation of substantial chemical waste, creating bottlenecks for reliable fluorochemical supplier networks aiming for green chemistry compliance. This patent discloses a remarkably efficient one-pot methodology that utilizes readily available biphenyl compounds, perfluoroalkyl sulfinates, trifluoroacetic anhydride, and trifluoromethanesulfonic acid to construct the dibenzothiophenium core directly. By streamlining the synthetic route and enabling the recovery of key starting materials, this technology offers a robust solution for cost reduction in API manufacturing while maintaining the high purity standards required by global regulatory bodies.
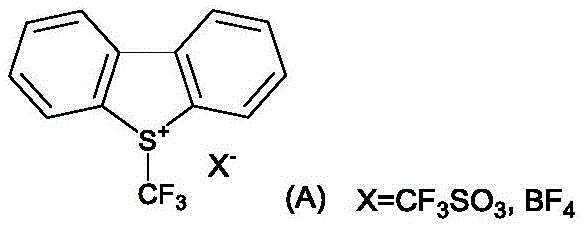
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of S-(perfluoroalkyl)-dibenzothiophene salts has relied on laborious multi-step syntheses starting from dibenzothiophene or its derivatives. A major drawback of these conventional routes is the reliance on expensive and hazardous reagents such as trifluoromethanesulfonic anhydride in stoichiometric excess, which drastically inflates the raw material costs and complicates waste disposal protocols. Furthermore, traditional methods often suffer from poor regioselectivity, yielding mixtures of isomers that are difficult to separate. For instance, when synthesizing fluorinated derivatives, the formation of the thermally unstable 2,6-isomer alongside the desired 2,8-isomer necessitates energy-intensive purification techniques like column chromatography, which are impractical for commercial scale-up of complex fluorinated intermediates. Additionally, the by-product dibenzothiophene generated during the actual fluorination application of these reagents was historically treated as waste, representing a loss of atomic economy and increasing the environmental footprint of the overall process.
The Novel Approach
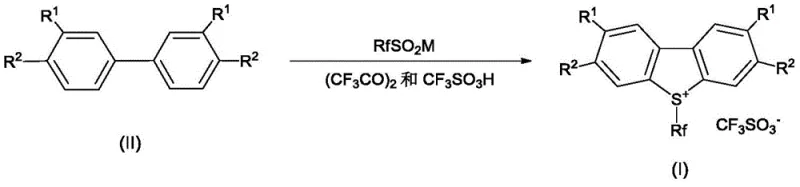
The innovative process described in the patent overcomes these hurdles by employing a direct cyclization strategy starting from substituted biphenyls. Instead of building the sulfur-containing ring first, the method constructs the S-(perfluoroalkyl)-dibenzothiophenium core in a single reactor vessel through a cascade of reactions involving perfluoroalkyl sulfinates and trifluoroacetic anhydride. This approach not only bypasses the need for expensive sulfonic anhydrides but also allows for precise control over the substitution pattern on the aromatic rings. By selecting specific biphenyl precursors, manufacturers can directly access halogenated or alkylated variants of the reagent with high specificity. Crucially, the process is designed with sustainability in mind; the reaction conditions facilitate the easy isolation of the product via simple filtration and washing, eliminating the need for complex chromatographic separations. This represents a substantial advancement for any organization seeking a reliable agrochemical intermediate supplier capable of delivering high-volume fluorination reagents with consistent quality.
Mechanistic Insights into the Cascade Cyclization Process
The chemical elegance of this invention lies in its three-stage mechanistic pathway occurring within a unified reaction medium. Initially, the perfluoroalkyl sulfinate salt reacts with trifluoroacetic anhydride to generate a highly reactive perfluoroalkyl sulfinyl trifluoroacetate intermediate. This species then acts as the electrophile in a subsequent Friedel-Crafts-type sulfinylation of the biphenyl substrate, directed by the electronic properties of the substituents on the aromatic rings. The resulting sulfinyl-biphenyl intermediate is not isolated but immediately subjected to cyclization conditions upon the addition of trifluoromethanesulfonic acid and further heating. This in-situ generation of reactive intermediates minimizes handling risks associated with unstable sulfinyl species and ensures high conversion rates. The presence of trifluoroacetic acid, generated as a by-product, serves a dual purpose as both a reaction medium and a proton source, facilitating the dehydration and aromatization steps required to close the central thiophene ring. Understanding this mechanism is vital for R&D teams optimizing reaction parameters, as the concentration of acid and the rate of anhydride addition directly influence the yield and purity of the final triflate salt.
A critical aspect of this mechanism is the control of regioisomers during the cyclization phase. The patent data reveals that temperature modulation is the key determinant in suppressing the formation of unwanted isomers. For example, in the synthesis of difluoro-substituted reagents, maintaining the reaction temperature within a specific window (e.g., 0°C to 45°C) favors the formation of the 2,8-difluoro isomer, which possesses superior thermal stability compared to the 2,6-isomer. If the temperature is too low, the cyclization kinetics are sluggish; if too high, thermodynamic equilibration may lead to impurity profiles that compromise the reagent's efficacy in downstream applications. This level of mechanistic control allows for the production of high-purity OLED material precursors or pharmaceutical intermediates where trace isomeric impurities could alter biological activity or material properties. The ability to tune the reaction outcome through thermal management rather than complex protecting group strategies underscores the practical utility of this method for industrial chemists.
How to Synthesize S-(Perfluoroalkyl)-Dibenzothiophene Triflate Efficiently
The operational simplicity of this one-pot protocol makes it highly attractive for pilot plant and commercial manufacturing environments. The process begins with the activation of the perfluoroalkyl source, followed by the sequential addition of the biphenyl substrate and acid catalysts under controlled thermal conditions. Detailed standard operating procedures regarding stoichiometry, solvent selection (such as nitromethane or sulfolane), and workup protocols are essential for reproducibility. The following guide outlines the critical phases of the synthesis based on the patented methodology, ensuring optimal yield and safety.
- React perfluoroalkyl sulfinate with trifluoroacetic anhydride to generate the active sulfinyl species.
- Add biphenyl substrate and trifluoromethanesulfonic acid at controlled low temperatures to form the sulfinyl-biphenyl intermediate.
- Heat the mixture to promote cyclization and isolate the final triflate salt via filtration and washing.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthesis route offers transformative benefits that extend beyond mere chemical efficiency. The shift from multi-step batch processes to a streamlined one-pot operation significantly reduces the consumption of solvents, energy, and labor hours, leading to drastic simplifications in the manufacturing workflow. By utilizing commercially available and inexpensive starting materials like sodium trifluoromethanesulfinate and substituted biphenyls, the dependency on scarce or volatile specialty reagents is minimized. This stability in raw material sourcing is crucial for maintaining continuous production schedules and mitigating the risks associated with supply chain disruptions common in the fine chemical sector. Furthermore, the elimination of column chromatography in favor of crystallization and filtration drastically lowers the cost of goods sold (COGS), making high-performance fluorinating agents more accessible for large-scale applications.
- Cost Reduction in Manufacturing: The replacement of expensive trifluoromethanesulfonic anhydride with the more economical trifluoroacetic anhydride represents a direct material cost saving that scales linearly with production volume. Additionally, the simplified workup procedure, which relies on washing and filtration rather than resource-intensive purification methods, reduces the demand for silica gel and organic solvents. This lean manufacturing approach allows for competitive pricing strategies without compromising on the quality of the final electrophilic reagent, providing a clear economic advantage for buyers sourcing bulk quantities of fluorination agents.
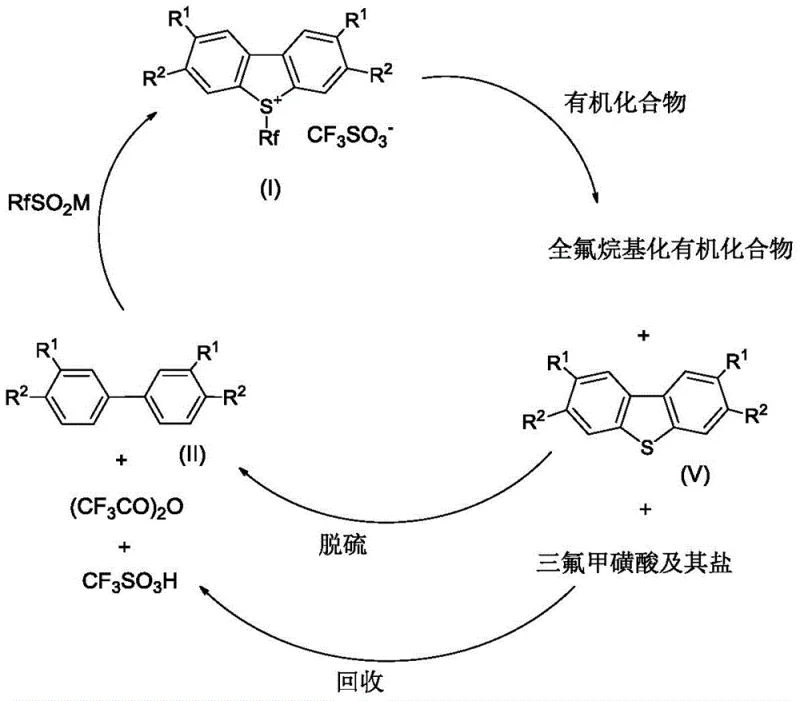
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, ensures high batch-to-b consistency, a critical factor for long-term supply contracts. The ability to recover and recycle the dibenzothiophene by-product back into the biphenyl starting material creates a circular economy model that insulates the supply chain from raw material price fluctuations. This closed-loop capability means that the effective yield of the process is maximized, and the environmental burden is minimized, aligning with the sustainability goals of modern multinational corporations seeking responsible partners.
- Scalability and Environmental Compliance: The one-pot nature of the reaction inherently reduces the number of unit operations, thereby shrinking the physical footprint required for production and lowering the potential for fugitive emissions. The use of recoverable solvents like trifluoroacetic acid and the generation of manageable salt by-products simplify waste treatment protocols, ensuring compliance with stringent environmental regulations. This scalability ensures that the transition from laboratory gram-scale to multi-ton commercial production can be achieved with minimal process re-engineering, facilitating rapid market entry for new fluorinated products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical aspects of adopting this method for industrial fluorination projects.
Q: How does this novel method improve isomer purity compared to traditional Umemoto reagent synthesis?
A: By strictly controlling the reaction temperature during the cyclization step (specifically between 0°C and 45°C), the process favors the formation of the thermally stable 2,8-isomer over the less stable 2,6-isomer, significantly reducing purification burdens.
Q: Can the dibenzothiophene by-product be recycled in this process?
A: Yes, the dibenzothiophene by-product generated during the perfluoroalkylation of organic substrates can be recovered and converted back to the biphenyl starting material via desulfurization with Raney Nickel, creating a sustainable closed-loop system.
Q: What are the primary cost drivers eliminated by this one-pot methodology?
A: This method eliminates the need for expensive trifluoromethanesulfonic anhydride as a primary reagent, replacing it with more economical trifluoroacetic anhydride, and removes complex column chromatography steps through simple filtration workups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(Perfluoroalkyl)-Dibenzothiophene Triflate Supplier
The technological advancements detailed in patent CN107540655B represent a significant opportunity for the fine chemical industry to enhance the efficiency of fluorination workflows. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent discoveries into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot synthesis are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of S-(perfluoroalkyl)-dibenzothiophene triflate meets the exacting standards required for pharmaceutical and electronic material applications, providing our clients with peace of mind regarding quality and consistency.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cost-effective and sustainable synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can accelerate your project timelines and reduce your overall development costs.
