Advanced Manufacturing of (S)-(+)-1-Indanyl Isocyanate: A Breakthrough in Chiral Pharmaceutical Intermediates
The pharmaceutical industry continuously demands more efficient and safer pathways for synthesizing complex chiral intermediates, particularly those serving as building blocks for neuroprotective and anticancer agents. Patent CN116396189A introduces a groundbreaking preparation method for (S)-(+)-1-indanyl isocyanate, a critical chiral intermediate with the CAS number 745783-81-7. This compound serves as a pivotal precursor for 1-aminoindan derivatives, which are increasingly utilized in the treatment of traumatic brain injury, tinnitus, and hypoxia. The disclosed technology addresses long-standing inefficiencies in existing synthetic routes by employing a non-traditional Lewis acid catalyst, tris(pentafluorophenyl)borane, to streamline the dehydration process. This innovation not only enhances the chemical yield but also significantly improves the operational safety profile by eliminating the need for hazardous reagents often associated with conventional methodologies. For global supply chain leaders and R&D directors, this patent represents a substantial leap forward in the reliable manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-aminoindan derivatives has been plagued by cumbersome multi-step procedures that hinder efficient commercial production. As illustrated in prior art, such as the route described in Tetrahedron Asymmetry (2003), the process typically begins with 1-indanone and (R)-phenylglycine amide to form a Schiff base, followed by high-pressure hydrogenation using Raney Nickel. This approach presents severe safety risks due to the pyrophoric nature of Raney Nickel, requiring specialized equipment and rigorous safety protocols that inflate operational costs. Furthermore, subsequent steps involve toxic phosphorus oxychloride for cyano group formation and elimination reactions that generate significant waste streams. Another reported method utilizing enzymatic resolution with Candida antarctic lipase suffers from low yields and poor resolution efficiency, making it economically unviable for large-scale applications. These legacy processes create bottlenecks in the supply chain, leading to extended lead times and inconsistent quality control for downstream API manufacturers.
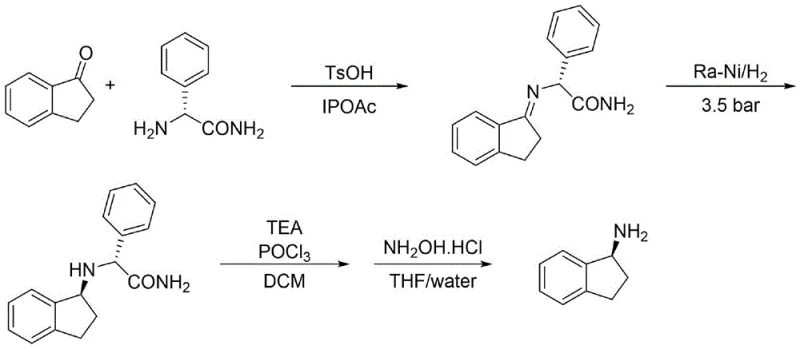
The Novel Approach
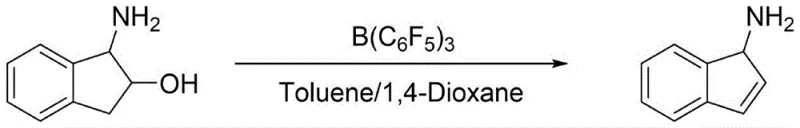
In stark contrast to these archaic methods, the novel approach detailed in CN116396189A utilizes 1-amino-2-indanol as a readily available starting material, bypassing the need for dangerous metal catalysts and complex protection groups. The core innovation lies in the direct dehydration of 1-amino-2-indanol to 1-aminoindene using tris(pentafluorophenyl)borane under reflux conditions with water separation. This catalytic system operates under mild conditions compared to traditional strong acid dehydrations, minimizing side reactions and degradation of the sensitive amino functionality. Following the formation of the indene intermediate, the process employs standard Pd/C catalytic hydrogenation, a robust and scalable technology familiar to most chemical manufacturing facilities. The subsequent chiral resolution using (S)-(-)-3-cyclohexenecarboxylic acid offers superior stereoselectivity compared to enzymatic methods, ensuring high optical purity without the need for expensive biocatalysts. This streamlined workflow drastically reduces the number of unit operations, directly translating to lower capital expenditure and improved throughput for industrial partners.
Mechanistic Insights into B(C6F5)3-Catalyzed Dehydration
The mechanistic elegance of this synthesis centers on the unique properties of tris(pentafluorophenyl)borane, a potent Lewis acid that facilitates the elimination of water from the beta-amino alcohol substrate. Unlike traditional Brønsted acids which might protonate the amine and inhibit reactivity or cause polymerization, this boron-based catalyst coordinates selectively with the hydroxyl group, activating it for elimination while tolerating the nucleophilic amine. The reaction proceeds through a concerted mechanism where the boron center stabilizes the transition state, allowing for the formation of the carbon-carbon double bond in 1-aminoindene with high regioselectivity. The use of a toluene and 1,4-dioxane solvent system further optimizes the reaction kinetics by facilitating the azeotropic removal of water, driving the equilibrium towards the product. This catalytic cycle is highly efficient, requiring only a molar ratio of 1:0.02 to 1:0.05 relative to the substrate, which minimizes catalyst loading costs and simplifies downstream purification. Understanding this mechanism is crucial for process chemists aiming to replicate these results, as it highlights the importance of maintaining anhydrous conditions during the initial mixing to prevent catalyst deactivation.
Impurity control is another critical aspect where this novel mechanism excels, particularly regarding the preservation of the amino group integrity throughout the synthesis. In conventional routes involving harsh acidic or basic conditions, the amino group is prone to oxidation or unwanted alkylation, leading to difficult-to-remove impurities that compromise the final API quality. The mild Lewis acid catalysis ensures that the amino group remains largely unreactive during the dehydration step, resulting in a crude product with GC purity exceeding 99%. Subsequent hydrogenation over Pd/C is carefully controlled at moderate pressures (40-45 Psi) to prevent over-reduction of the aromatic ring, a common side reaction in indan synthesis. The final resolution step acts as a powerful purification gate, where the specific interaction between the chiral acid and the amine enantiomers effectively filters out any remaining racemic impurities or structural analogs. This multi-layered approach to impurity management ensures that the final (S)-(+)-1-indanyl isocyanate meets the stringent purity specifications required for regulatory submission in pharmaceutical applications.
How to Synthesize (S)-(+)-1-Indanyl Isocyanate Efficiently
The synthesis of this high-value chiral intermediate is achieved through a logical sequence of four distinct chemical transformations, each optimized for maximum yield and safety. The process begins with the catalytic dehydration of 1-amino-2-indanol, followed by hydrogenation to saturate the double bond, then chiral resolution to isolate the desired enantiomer, and finally conversion to the isocyanate. Each step has been rigorously tested to ensure reproducibility and scalability, providing a clear roadmap for manufacturing teams. The detailed standardized synthesis steps, including specific reagent quantities, temperature profiles, and workup procedures, are outlined in the comprehensive guide below to assist technical teams in process validation.
- Dehydration of 1-amino-2-indanol using tris(pentafluorophenyl)borane catalyst to form 1-aminoindene.
- Catalytic hydrogenation of 1-aminoindene using Pd/C to produce racemic 1-aminoindan.
- Chiral resolution of 1-aminoindan using (S)-(-)-3-cyclohexenecarboxylic acid followed by dissociation.
- Reaction of resolved (S)-(+)-1-aminoindan with diphosgene to yield the final isocyanate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. By eliminating the reliance on pyrophoric Raney Nickel and toxic phosphorus oxychloride, the process significantly reduces the regulatory burden and safety compliance costs associated with handling hazardous materials. The use of commercially available 1-amino-2-indanol as a starting material ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty chemical markets. Furthermore, the simplified operational workflow, characterized by fewer reaction steps and easier purification protocols, allows for faster batch turnover times. This efficiency gain directly contributes to cost reduction in pharmaceutical intermediate manufacturing, enabling suppliers to offer more competitive pricing without compromising on quality. The robustness of the Pd/C hydrogenation and crystallization resolution steps also ensures consistent product quality across different production batches, a critical factor for long-term supply agreements with multinational pharmaceutical companies.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as Raney Nickel and phosphorus oxychloride leads to substantial savings in raw material costs and waste disposal fees. Additionally, the high catalytic efficiency of tris(pentafluorophenyl)borane means that lower catalyst loadings are required, further reducing the bill of materials. The streamlined process flow reduces energy consumption by minimizing the number of heating and cooling cycles, while the high purity of intermediates reduces the need for extensive chromatographic purification, which is often a major cost driver in fine chemical production. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), providing a strong competitive advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of stable, non-hazardous starting materials that are widely available from multiple global vendors. The avoidance of specialized equipment required for high-pressure hydrogenation with pyrophoric catalysts means that production can be easily transferred between different manufacturing sites without significant capital investment. This flexibility enhances supply continuity, ensuring that customers receive their orders on time even in the face of regional disruptions. The robust nature of the chemical steps also means that production schedules are less likely to be delayed by unexpected technical failures or safety incidents, providing procurement teams with greater confidence in their supply planning and inventory management.
- Scalability and Environmental Compliance: The process is inherently designed for green chemistry principles, generating minimal waste and avoiding the use of persistent organic pollutants. The aqueous workup steps and the ability to recycle solvents like toluene and dichloromethane align with modern environmental regulations, reducing the ecological footprint of the manufacturing process. Scalability is ensured by the use of standard reactor types and operating conditions that are easily replicated from pilot scale to multi-ton commercial production. This ease of scale-up allows manufacturers to respond quickly to increasing market demand for (S)-(+)-1-indanyl isocyanate derivatives, supporting the rapid development of new drug candidates without the typical delays associated with process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (S)-(+)-1-indanyl isocyanate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline or manufacturing process.
Q: What are the advantages of using tris(pentafluorophenyl)borane over traditional dehydration methods?
A: Traditional methods often require multiple protection and deprotection steps or harsh conditions. The use of tris(pentafluorophenyl)borane allows for direct reflux dehydration with water separation, significantly simplifying the operation, reducing step count, and improving overall yield and safety profile for industrial scale-up.
Q: How is high chiral purity achieved in this synthesis route?
A: High enantiomeric excess (ee > 99%) is achieved through a highly efficient crystallization resolution step using (S)-(-)-3-cyclohexenecarboxylic acid. This resolving agent forms a diastereomeric salt with the target amine, which can be purified via filtration and subsequent dissociation, ensuring stringent purity specifications required for API intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed with scalability in mind. It utilizes readily available raw materials like 1-amino-2-indanol and avoids pyrophoric catalysts like Raney Nickel found in older literature. The operational simplicity and robust purification steps make it highly suitable for commercial scale-up from pilot plants to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-(+)-1-Indanyl Isocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the successful development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our expertise in handling complex chiral amines and isocyanates allows us to navigate the intricacies of this specific synthesis route, guaranteeing a consistent supply of (S)-(+)-1-indanyl isocyanate that meets international regulatory standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing process can reduce your overall project costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your requirements, ensuring a seamless integration of our high-purity intermediates into your supply chain.
