Advanced Organocatalytic Synthesis of Potent Naphthoquinopyran Anticancer Intermediates
The landscape of oncology drug discovery is constantly evolving, driven by the urgent need for more effective and less toxic therapeutic agents. A significant breakthrough in this domain is documented in Chinese patent CN108864110B, which details the synthesis and application of novel naphthoquinopyran derivatives. These compounds, characterized by their fused ring systems incorporating naphthoquinone and pyran moieties, have demonstrated remarkable inhibitory activity against aggressive cancer cell lines, including triple-negative breast cancer (MDA-MB-231), triple-positive breast cancer (MCF-7), and human lung cancer (A549). The patent highlights a sophisticated yet practical synthetic methodology that leverages organocatalysis to construct these complex pharmacophores efficiently. For pharmaceutical developers and procurement specialists, this technology represents a pivotal shift towards greener, more scalable, and cost-effective manufacturing of high-value anticancer intermediates.
The core innovation lies in the structural versatility of the derivatives, specifically those defined by formulas 7 and 9. These molecules are not merely theoretical constructs but have undergone rigorous biological evaluation, revealing potent anticancer properties with a favorable safety profile compared to existing chemotherapeutics. The ability to fine-tune the substituents on the aromatic rings allows for precise modulation of biological activity, targeting specific cancer pathways while minimizing off-target effects on healthy tissue. This level of molecular precision is critical for the next generation of targeted cancer therapies.
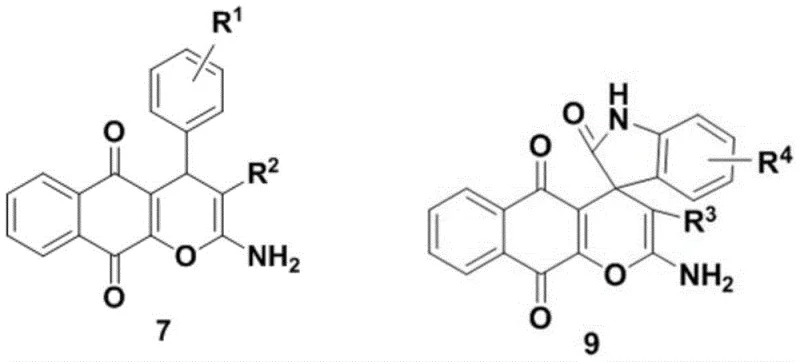
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of naphthoquinone-based heterocycles has been plagued by significant operational challenges that hinder commercial viability. Traditional protocols often rely on harsh reaction conditions, including the use of strong bases, high temperatures, or toxic transition metal catalysts such as iron oxide nanoparticles or zinc complexes. These methods frequently suffer from low atom economy, requiring stoichiometric amounts of reagents that generate substantial chemical waste. Furthermore, the removal of residual heavy metals from the final product is a costly and technically demanding step, essential for meeting stringent pharmaceutical purity standards. The reliance on volatile organic solvents and extended reaction times further exacerbates the environmental footprint and production costs, creating bottlenecks in the supply chain for these critical intermediates.
The Novel Approach
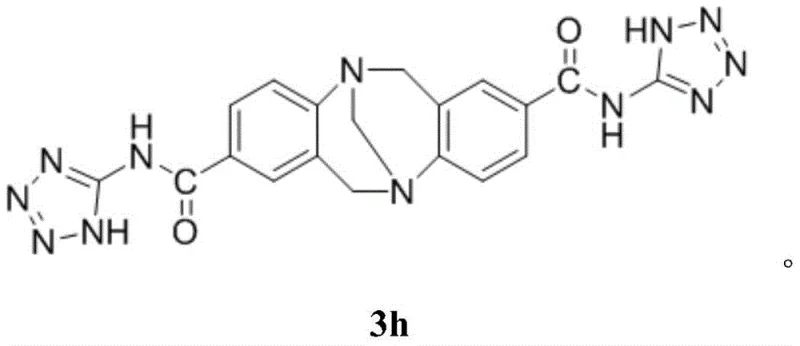
In stark contrast, the methodology disclosed in patent CN108864110B introduces a paradigm shift through the use of a specialized amide-triazabicyclodecene derivative, specifically compound 3h, as an efficient organocatalyst. This approach facilitates a one-pot multicomponent reaction involving 2-hydroxy-1,4-naphthoquinone, an aromatic aldehyde or substituted isatin, and an active methylene compound like benzothiazole-2-acetonitrile. The reaction proceeds smoothly in ethanol, a green and inexpensive solvent, under mild reflux conditions. This eliminates the need for hazardous reagents and simplifies the workup process significantly. The result is a high-yielding transformation that produces complex spiro or fused heterocyclic systems with excellent regioselectivity, directly addressing the purity and efficiency concerns that have long plagued this chemical class.
Mechanistic Insights into Amide-Triazabicyclodecene Catalyzed Cyclization
The efficacy of this synthetic route is rooted in the unique bifunctional nature of the catalyst 3h. As a strong organic base, it effectively activates the acidic protons of the active methylene component, generating a nucleophilic species capable of attacking the electrophilic centers of the naphthoquinone and aldehyde/isatin substrates. Simultaneously, the amide functionality within the catalyst framework likely participates in hydrogen bonding interactions, stabilizing the transition states and orienting the reactants for optimal cyclization. This dual activation mechanism lowers the activation energy of the reaction, allowing it to proceed rapidly at lower temperatures compared to uncatalyzed thermal methods. The robustness of this catalytic cycle ensures consistent performance across a wide range of substrate electronic properties, from electron-deficient nitro-substituted aldehydes to electron-rich methoxy derivatives.
From an impurity control perspective, the mildness of the reaction conditions is a decisive advantage. Harsh basic conditions often lead to the decomposition of the sensitive naphthoquinone core or the formation of polymeric byproducts, which are difficult to separate and can compromise the safety profile of the final drug substance. By operating in ethanol at reflux with a controlled amount of organic catalyst, the process minimizes side reactions such as over-alkylation or oxidative degradation. The resulting crude product is typically of high purity, requiring only standard column chromatography for final isolation. This streamlined purification pathway not only reduces solvent consumption but also enhances the overall throughput of the manufacturing process, ensuring a reliable supply of high-quality intermediates for preclinical and clinical studies.
How to Synthesize Naphthoquinopyran Derivatives Efficiently
The practical implementation of this technology involves a straightforward procedure that is easily adaptable to various scales of production. The process begins with the preparation of the reaction mixture in a dry vessel, ensuring that moisture does not interfere with the catalytic activity. The specific molar ratios of the starting materials are optimized to drive the equilibrium towards the desired product while minimizing the formation of unreacted starting materials. The use of ethanol as the solvent aligns with modern green chemistry principles, reducing the environmental burden associated with solvent disposal and recovery. Following the reaction, the isolation of the product is achieved through simple evaporation and purification techniques, making this route highly attractive for process chemists looking to optimize manufacturing workflows.
- Combine 2-hydroxy-1,4-naphthoquinone, aromatic aldehyde or substituted isatin, and an acetonitrile derivative in a dry round-bottom flask.
- Add the specific amide-triazabicyclodecene catalyst (e.g., compound 3h) and ethanol as the green solvent medium.
- Heat the mixture to reflux under stirring until reaction completion is confirmed by TLC, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere chemical efficiency. The shift from metal-catalyzed to organocatalytic processes fundamentally alters the cost structure and risk profile of producing these valuable intermediates. By eliminating the dependency on scarce or expensive transition metals, manufacturers can insulate themselves from volatile commodity markets and supply disruptions. Furthermore, the simplified purification requirements translate directly into reduced operational expenditures, as fewer processing steps mean lower labor, energy, and equipment utilization costs. This efficiency gain is crucial for maintaining competitiveness in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated heavy metal scavenging steps leads to substantial cost savings. Additionally, the use of ethanol, a commodity solvent, replaces more expensive and hazardous organic solvents, further driving down raw material costs. The high yields reported in the patent minimize waste generation, ensuring that a greater proportion of input materials are converted into saleable product, thereby optimizing the overall cost of goods sold (COGS) for these complex intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 2-hydroxy-1,4-naphthoquinone and various substituted benzaldehydes, are commercially available in bulk quantities from multiple suppliers. This diversity in the supply base reduces the risk of single-source dependency and ensures continuity of supply even during market fluctuations. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality or environmental factors, providing a stable and predictable manufacturing output that supports long-term planning.
- Scalability and Environmental Compliance: The one-pot nature of the reaction significantly simplifies scale-up from laboratory to pilot and commercial production. There is no need for complex intermediate isolation or sensitive handling procedures, which often pose challenges during technology transfer. Moreover, the reduced generation of hazardous waste and the use of a green solvent facilitate compliance with increasingly stringent environmental regulations. This proactive approach to sustainability not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their R&D pipelines. The following questions address common inquiries regarding the stability, scalability, and specific applications of these naphthoquinopyran derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making.
Q: What are the primary advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: This method eliminates the need for toxic transition metals and harsh conditions, utilizing a benign ethanol solvent and a reusable organic catalyst, which simplifies downstream purification and reduces environmental impact.
Q: Which specific derivatives demonstrated the highest selectivity for breast cancer cells?
A: Compound 7u exhibited high specificity against triple-positive breast cancer cells (MCF-7) with negligible toxicity to normal human bronchial epithelial cells, making it a prime candidate for further development.
Q: Is this synthesis protocol suitable for large-scale commercial production?
A: Yes, the one-pot nature of the reaction, combined with the use of common solvents like ethanol and standard reflux conditions, makes it highly amenable to scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoquinopyran Derivative Supplier
The promising biological activity of compounds like 7u and 9k underscores the immense potential of naphthoquinopyran derivatives in the fight against cancer. However, translating this potential into reality requires a manufacturing partner with the technical expertise to execute complex syntheses with precision and consistency. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of organocatalytic reactions, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards necessary for pharmaceutical applications.
We invite research teams and procurement leaders to explore the possibilities of collaborating with us on the development and supply of these advanced anticancer intermediates. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your journey from discovery to market is supported by a reliable and innovative supply chain partner.
