Advanced Rong-Phos Iridium Complexes for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of the chiral bidentate nitrogen phosphine ligand Rong-Phos iridium complex, as detailed in patent CN111499666B. This groundbreaking technology addresses long-standing challenges in constructing stable nitrogen chiral centers, a feat that has historically eluded many synthetic methodologies due to the inherent instability of nitrogen inversion. By successfully stabilizing both carbon and nitrogen chiral centers within a single iridium coordination sphere, this innovation offers unprecedented control over stereoselectivity in hydrogenation reactions. For R&D directors and process chemists, this represents a pivotal shift towards more reliable and versatile catalytic systems capable of handling complex substrate scopes. The ability to generate four distinct optical isomers from a single ligand framework provides a strategic advantage in developing robust synthetic routes for high-value pharmaceutical intermediates. Furthermore, the demonstrated stability of these complexes under various reaction conditions suggests a high potential for seamless technology transfer from laboratory discovery to commercial manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to asymmetric hydrogenation have predominantly relied on ligands possessing only carbon-centered chirality, which often limits the diversity of accessible stereochemical outcomes. In many conventional systems, the inability to stabilize nitrogen chirality restricts the catalyst's ability to differentiate between subtle steric environments, leading to lower enantiomeric excess values in challenging transformations. Moreover, existing methods frequently require distinct ligand syntheses to access opposite enantiomers of a target product, thereby doubling the development time and increasing the overall cost of goods sold. The instability of nitrogen centers in prior art often results in racemization during the catalytic cycle, compromising the optical purity of the final active pharmaceutical ingredient. These limitations create significant bottlenecks for procurement managers seeking cost-effective solutions that do not compromise on quality or regulatory compliance standards. Consequently, the industry has been in urgent need of a catalytic system that can overcome these thermodynamic barriers while maintaining high turnover frequencies.
The Novel Approach
The Rong-Phos iridium complex introduces a paradigm shift by leveraging a dual-chirality system that stabilizes the nitrogen center through kinetic and thermodynamic control upon metal coordination. This novel approach allows for the precise construction of nitrogen chiral centers with high enantioselectivity, enabling the catalyst to distinguish between pro-chiral faces of substrates with exceptional accuracy. As illustrated in the reaction scheme below, this methodology facilitates the asymmetric hydrogenation of cyclic unsaturated carbonyl compounds with remarkable efficiency and selectivity.
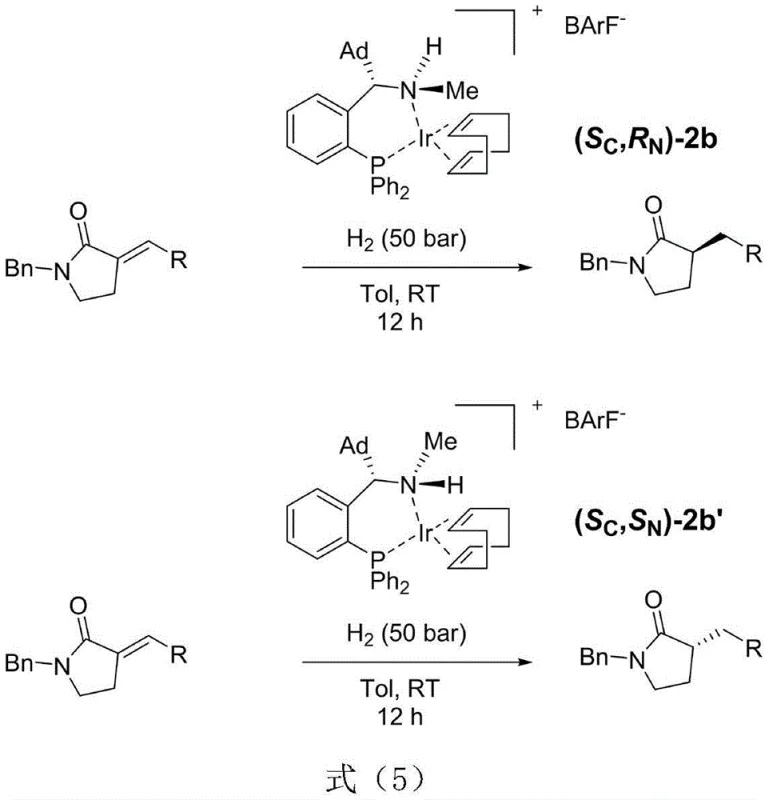
By utilizing a pair of diastereomer catalysts with different nitrogen center chiralities, manufacturers can access both R and S configuration products using the same ligand backbone, drastically simplifying the supply chain logistics. This flexibility reduces the need for multiple custom ligand syntheses, thereby streamlining the procurement process and enhancing overall operational agility. The robustness of this system under standard hydrogenation pressures and temperatures further underscores its viability for large-scale industrial applications where consistency is paramount.
Mechanistic Insights into Rong-Phos Iridium Complex Catalysis
The core innovation lies in the unique structural architecture of the Rong-Phos ligand, which integrates a bidentate nitrogen-phosphine motif capable of forming stable ionic iridium complexes. As depicted in the structural formulas, the ligand coordinates with the iridium metal center to create a rigid chiral environment that effectively transmits stereochemical information during the catalytic cycle. The presence of both carbon and nitrogen chiral centers generates four possible isomeric forms, each offering a distinct spatial arrangement around the active metal site. This structural diversity allows for fine-tuning of the catalyst's electronic and steric properties to match specific substrate requirements, ensuring optimal performance across a wide range of cyclic unsaturated carbonyl compounds.
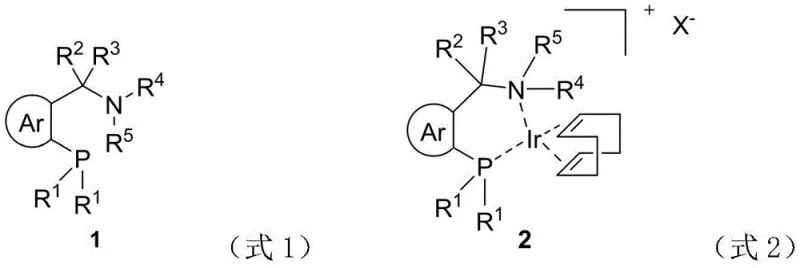
From an impurity control perspective, the high stability of the nitrogen chiral center prevents unwanted racemization pathways that typically plague asymmetric synthesis. This inherent stability ensures that the optical purity of the product remains intact throughout the reaction duration, minimizing the formation of undesired enantiomeric impurities that are difficult to remove downstream. For quality assurance teams, this translates to a cleaner reaction profile and reduced burden on purification processes such as chromatography or crystallization. The mechanistic robustness of the Rong-Phos system thus provides a reliable foundation for producing high-purity intermediates that meet stringent regulatory specifications for pharmaceutical applications.
How to Synthesize Rong-Phos Efficiently
The synthesis of the Rong-Phos iridium complex involves a streamlined sequence starting from chiral sulfinamide precursors, followed by hydrolysis and metal coordination steps. Detailed standardized synthetic procedures, including specific solvent systems, temperature ranges, and molar ratios, are critical for reproducing the high enantioselectivity reported in the patent literature. Process engineers should note that the choice of anion, such as BARF or BF4, can influence the solubility and catalytic activity of the final complex, necessitating careful optimization during scale-up. The following guide outlines the key stages required to achieve consistent batch-to-batch quality.
- Prepare the chiral bidentate nitrogen phosphine ligand Rong-Phos via hydrolysis of sulfinamide precursors followed by substitution or reductive amination.
- React the purified Rong-Phos ligand with [Ir(COD)Cl]2 dimer in dry dichloromethane under inert atmosphere.
- Add sodium salt (NaBARF or NaBF4) and water to finalize the ionic iridium complex structure with specific nitrogen chirality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the Rong-Phos iridium complex offers substantial strategic benefits beyond mere technical performance. The ability to access both enantiomers from a single ligand platform significantly reduces the complexity of raw material sourcing and inventory management. This consolidation of supply requirements leads to a more resilient supply chain, less vulnerable to disruptions associated with managing multiple specialized reagents. Furthermore, the high efficiency of the catalyst minimizes waste generation and solvent consumption, aligning with modern sustainability goals and reducing environmental compliance costs. These factors collectively contribute to a more predictable and cost-efficient manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of the need for separate ligand syntheses for opposite enantiomers results in significant savings in research and development expenditures. By utilizing a unified ligand framework, companies can leverage economies of scale in precursor purchasing, driving down the unit cost of the catalyst. Additionally, the high turnover number and enantioselectivity reduce the amount of catalyst required per batch, further lowering the overall material cost. This economic efficiency is crucial for maintaining competitiveness in the margin-sensitive pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: The robust nature of the Rong-Phos complex ensures consistent performance across different production batches, mitigating the risk of campaign failures due to catalyst variability. Suppliers can maintain smaller safety stocks of raw materials since the same ligand serves multiple product configurations, optimizing working capital utilization. This reliability fosters stronger partnerships between chemical manufacturers and their clients, as delivery schedules become more predictable and dependable. Ultimately, a stable supply of high-quality catalysts supports uninterrupted production lines for critical drug substances.
- Scalability and Environmental Compliance: The reaction conditions described in the patent utilize common organic solvents and moderate hydrogen pressures, facilitating easy scale-up from pilot plant to commercial production volumes. The high selectivity of the process reduces the formation of by-products, simplifying waste treatment protocols and lowering the environmental footprint of the manufacturing site. This alignment with green chemistry principles enhances the corporate social responsibility profile of the organization while ensuring compliance with increasingly strict environmental regulations. Such scalability ensures that the technology can grow alongside market demand without requiring major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of Rong-Phos iridium complexes in industrial settings. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a factual basis for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this technology into their existing process workflows. The answers reflect the practical implications of the dual-chirality mechanism on product quality and operational efficiency.
Q: What makes the Rong-Phos iridium complex unique compared to traditional phosphine ligands?
A: Unlike traditional ligands that rely solely on carbon chirality, the Rong-Phos iridium complex features both carbon-centered and nitrogen-centered chirality. This dual-chirality system allows for the construction of four distinct optical isomers, enabling precise control over the stereochemical outcome of asymmetric hydrogenation reactions.
Q: Can this catalyst be used for large-scale production of chiral amines?
A: Yes, the patent demonstrates excellent stability and high enantioselectivity (up to 99% ee) across various cyclic unsaturated carbonyl compounds. The robust nature of the nitrogen chiral center ensures consistent performance during scale-up, making it suitable for industrial manufacturing of pharmaceutical intermediates.
Q: How does the nitrogen chirality affect the reaction selectivity?
A: The chirality at the nitrogen atom creates different chiral environments within the catalytic cycle. By selecting specific diastereomers of the iridium complex, manufacturers can access either R or S configuration products using the same ligand backbone, significantly simplifying the supply chain for enantiomeric pairs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rong-Phos Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Rong-Phos iridium complex in advancing the synthesis of chiral pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst meets the highest international standards. Our commitment to quality ensures that your downstream processes remain robust and compliant with global regulatory requirements.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. By engaging with us, you can obtain specific COA data and route feasibility assessments that demonstrate the tangible benefits of adopting this advanced catalytic technology. Let us partner with you to optimize your synthesis routes and secure a competitive advantage in the global market for high-value chiral chemicals.
