Revolutionizing Deuterated API Intermediates via Green Electrochemical Deoxydeuteration
Revolutionizing Deuterated API Intermediates via Green Electrochemical Deoxydeuteration
In the rapidly evolving landscape of pharmaceutical development, the demand for high-purity deuterated compounds has surged due to their ability to improve metabolic stability and pharmacokinetic profiles through the kinetic isotope effect. A groundbreaking advancement in this field is detailed in Chinese Patent CN114438532A, which discloses a novel electrochemical method for the deoxydeuteration reduction of aldehyde and ketone compounds into corresponding gem-di-deuterated saturated hydrocarbons. This technology represents a paradigm shift from traditional stoichiometric chemical reductions to a catalytic, electricity-driven process that operates under mild conditions. By leveraging simple electrode materials and abundant deuterium sources, this invention offers a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their synthetic routes for deuterated active pharmaceutical ingredients (APIs).
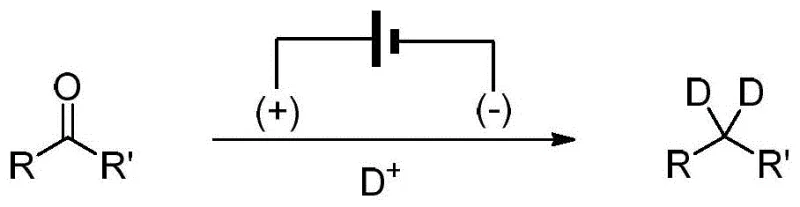
The significance of this patent lies in its ability to address long-standing challenges in deuterium incorporation, specifically the replacement of carbonyl oxygen atoms with two deuterium atoms to form CD2 groups. Traditional approaches often suffer from harsh reaction conditions, poor atom economy, or the requirement for exotic reagents. In contrast, the method described in CN114438532A utilizes a straightforward electrochemical cell setup where the substrate, an electrolyte, and a deuterated protic acid are dissolved in a solvent system. Upon applying a constant current at room temperature, the carbonyl group is efficiently reduced, yielding the target gem-di-deuterated product with exceptional isotopic purity. This innovation not only streamlines the synthesis of complex deuterated molecules but also aligns perfectly with the industry's growing emphasis on green chemistry and sustainable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gem-di-deuterated hydrocarbons from carbonyl precursors has relied heavily on chemical reduction strategies that present significant operational and economic hurdles for large-scale manufacturing. One common approach involves the use of powerful reducing agents such as lithium aluminum deuteride (LiAlD4), which, while effective, is pyrophoric, moisture-sensitive, and generates substantial amounts of aluminum waste that requires complex disposal protocols. Another prevalent method employs catalytic hydrogenation using deuterium gas (D2) over precious metal catalysts like palladium on carbon (Pd/C); however, this necessitates the use of high-pressure reactors and expensive noble metals, introducing safety risks and inflating the cost of goods sold (COGS). Furthermore, multi-step chemical syntheses utilizing deuterated alkylating agents like CD3I are often limited by the availability of specific starting materials and can lead to scrambling of the deuterium label during subsequent transformation steps, compromising the isotopic integrity of the final drug substance.
The Novel Approach
The electrochemical deoxydeuteration method disclosed in the patent data offers a transformative solution by eliminating the need for hazardous chemical reductants and high-pressure infrastructure. By employing a simple undivided cell equipped with a zinc anode and a graphite felt cathode, the process facilitates the direct reduction of the carbonyl group using electrons as the primary reagent. The deuterium source is derived from inexpensive and readily available deuterated hydrochloric acid (DCl) in heavy water (D2O), which serves both as the proton donor and the solvent component. This approach operates at ambient temperature and atmospheric pressure, drastically simplifying the engineering requirements for the reaction vessel. Moreover, the absence of transition metal catalysts removes the risk of heavy metal contamination in the final product, a critical quality attribute for pharmaceutical intermediates, thereby reducing the need for extensive purification steps and enhancing the overall process efficiency.
Mechanistic Insights into Electrochemical Deoxydeuteration
The core of this technology relies on the precise control of electron transfer at the electrode-solution interface to drive the reductive cleavage of the carbon-oxygen bond. In the proposed mechanism, the carbonyl substrate undergoes reduction at the cathode surface, accepting electrons to form a radical anion or dianion intermediate depending on the specific potential applied. Simultaneously, the deuterated protic acid in the solution provides a rich reservoir of deuterons (D+) that rapidly quench the reduced intermediates. This concerted sequence of electron transfer and deuteronation effectively replaces the oxygen atom with two deuterium atoms, resulting in the formation of the gem-di-deuterated methylene group. The use of a sacrificial zinc anode helps maintain the charge balance and may also play a role in scavenging oxygen or stabilizing the electrolyte environment, ensuring a consistent and reproducible reaction outcome across different substrate classes.
Understanding the mechanistic nuances is vital for controlling impurity profiles, particularly regarding the retention of the deuterium label. The high deuteration rates (>99%) reported in the patent examples suggest that the exchange rate of deuterons from the solvent system is significantly faster than any potential back-exchange with residual protons. This kinetic preference ensures that once the deuterium is incorporated into the carbon framework, it remains stable throughout the workup and isolation phases. For R&D teams, this implies that the process is highly forgiving regarding minor variations in moisture content, provided the molar excess of the deuterated acid is maintained. The robustness of this mechanism allows for the processing of sensitive functional groups that might otherwise be compromised by strong chemical reducing agents, thereby expanding the scope of accessible deuterated scaffolds for drug discovery programs.
How to Synthesize Gem-Di-Deuterated Hydrocarbons Efficiently
Implementing this electrochemical protocol requires careful attention to the composition of the electrolyte solution and the configuration of the electrode system to maximize current efficiency. The standard procedure involves dissolving the aldehyde or ketone substrate along with a supporting electrolyte like sodium chloride in a mixed solvent system of acetonitrile and heavy water. Following the addition of deuterated hydrochloric acid, the reaction is initiated by applying a constant current, typically ranging from 30 mA for small-scale optimization to 20 A for pilot-scale production. The reaction progress is monitored until the starting material is fully consumed, after which standard aqueous workup and chromatographic purification yield the high-purity deuterated product. For a detailed breakdown of the specific parameters and troubleshooting tips, please refer to the standardized synthesis guide below.
- Dissolve the aldehyde or ketone substrate, an electrolyte such as sodium chloride, and a deuterated protic acid in a solvent mixture of acetonitrile and heavy water within an electrochemical cell.
- Insert a zinc anode and a graphite felt cathode into the reaction mixture and connect to a power supply.
- Apply a constant current at room temperature for approximately 1 to 5 hours until the reaction is complete, then isolate the product via extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this electrochemical technology offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility in the deuterated chemicals market. By shifting the dependency from scarce precious metals and hazardous reagents to commodity chemicals and electricity, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The elimination of expensive catalysts like palladium and platinum not only lowers the raw material bill but also mitigates the supply risk associated with these geopolitically sensitive metals. Furthermore, the simplified equipment requirements—standard glass reactors or steel vessels lined with inert materials instead of high-pressure autoclaves—reduce capital expenditure (CAPEX) for new production lines and allow for faster deployment of manufacturing capacity to meet market demand.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of high-cost reagents with low-cost alternatives. Traditional deuterium incorporation often relies on reagents like LiAlD4 or D2 gas, which carry substantial price tags and require specialized handling infrastructure. In contrast, the electrochemical method utilizes zinc rods and graphite felt, which are inexpensive and widely available industrial materials. Additionally, the use of sodium chloride as an electrolyte and DCl/D2O as the deuterium source further drives down the variable costs per kilogram of product. This structural change in the cost base allows for more competitive pricing strategies and improved margin protection against fluctuations in the global chemical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and modularity of the electrochemical setup. Since the process does not rely on complex catalytic systems that may have long lead times or single-source suppliers, procurement teams can secure materials with greater ease. The ability to run reactions at room temperature and atmospheric pressure also reduces the logistical burden of transporting hazardous gases or managing high-energy chemical inventories. This operational flexibility ensures continuous production capabilities even during periods of resource constraint, making it a reliable choice for maintaining the continuity of supply for critical deuterated drug candidates.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis route is markedly lower than conventional methods, aligning with stringent global regulations on waste disposal and emissions. The absence of heavy metal waste streams simplifies effluent treatment and reduces the costs associated with environmental compliance. Moreover, the patent data explicitly demonstrates successful scale-up from milligram scales in three-neck flasks to multi-gram and potentially kilogram scales in 50-liter reactors, proving the commercial scale-up of complex deuterated intermediates is feasible. This scalability ensures that the technology can grow alongside the clinical development of the drug, from early-phase trials to commercial launch, without the need for disruptive process changes.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries based on the patent specifications. These questions address the practical aspects of implementation, including substrate compatibility, safety considerations, and quality control metrics. Understanding these details is crucial for integrating this electrochemical method into existing development pipelines and ensuring that the resulting deuterated compounds meet the rigorous standards required for clinical applications.
Q: What are the advantages of this electrochemical method over traditional chemical reduction?
A: Unlike traditional methods requiring expensive precious metal catalysts like palladium or hazardous reagents like lithium aluminum deuteride, this electrochemical approach uses inexpensive zinc and graphite electrodes under mild room temperature conditions, significantly reducing safety risks and raw material costs.
Q: Can this process be scaled for industrial production of deuterated drugs?
A: Yes, the patent demonstrates successful scale-up in a 50-liter glass reactor with a 20.0A constant current, proving the feasibility of commercial scale-up of complex pharmaceutical intermediates without the need for high-pressure equipment.
Q: What is the typical deuteration rate achieved with this technology?
A: The method consistently achieves a deuteration rate of greater than 99%, ensuring high isotopic purity which is critical for the metabolic stability and regulatory approval of deuterated pharmaceutical products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Di-Deuterated Hydrocarbons Supplier
As the pharmaceutical industry continues to embrace deuterated drugs for their enhanced therapeutic profiles, the need for a partner capable of delivering high-quality deuterated intermediates at scale has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging advanced electrochemical synthesis platforms to provide cost-effective and sustainable solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of deuterated material meets the highest isotopic and chemical purity standards required by global regulatory agencies.
We invite you to explore how our expertise in electrochemical deoxydeuteration can accelerate your drug development timeline and optimize your production costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets, highlighting potential efficiencies in your current supply chain. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects, ensuring a secure and efficient supply of high-purity deuterated building blocks for your next generation of therapies.
