Advanced Electrochemical Manufacturing of Eight-Membered Selenium-Containing Benzoazacycles for Global Pharma Supply Chains
Introduction to Next-Generation Selenium Heterocycle Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access complex heterocyclic scaffolds, particularly those containing selenium, which is increasingly recognized for its potent biological activities ranging from antioxidant properties to anticancer potential. A groundbreaking development in this field is detailed in Chinese Patent CN115652344A, which discloses a novel method for preparing eight-membered selenium-containing benzoazacycle compounds. This technology represents a significant leap forward by leveraging electrochemical driving forces to generate selenyl radicals, thereby enabling a radical coupling-cyclization cascade that constructs the challenging eight-membered ring system without the need for transition metal catalysts or stoichiometric chemical oxidants. The ability to synthesize these medium-sized nitrogen-selenium heterocycles efficiently opens new doors for drug discovery teams looking to explore underrepresented chemical space with high-purity intermediates.
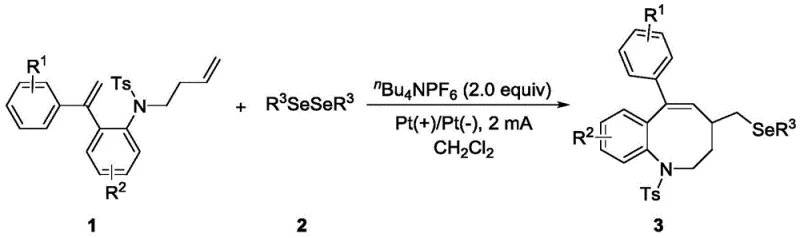
The core innovation lies in the use of electricity as a clean reagent to drive the transformation of N-(but-3-en-1-yl)-4-methyl-N-(2-(1-phenylvinyl)phenyl)benzenesulfonamide derivatives. By employing an undivided cell setup with platinum electrodes, the process achieves anodic oxidation of diselenides to generate reactive selenyl radical species in situ. These radicals then engage in a precise sequence of intermolecular addition and intramolecular cyclization with the alkene-tethered substrate. This approach not only streamlines the synthetic route but also aligns perfectly with the principles of green chemistry, offering a reliable pharmaceutical intermediate supplier with a distinct competitive edge in sustainability and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of medium-sized heterocyclic rings, such as eight-membered benzoazacycles, has been fraught with synthetic challenges that hinder large-scale adoption. Conventional methodologies often rely heavily on transition metal catalysis, utilizing precious metals like palladium, platinum, or rhodium to facilitate C-H activation or cross-coupling reactions. While effective in small-scale laboratory settings, these metal-dependent routes introduce significant bottlenecks for commercial manufacturing. The primary concern is the stringent regulatory requirement for residual metal limits in active pharmaceutical ingredients (APIs), necessitating complex and costly purification steps to remove trace metal contaminants. Furthermore, traditional methods frequently require stoichiometric amounts of harsh chemical oxidants to regenerate the active catalytic species, generating substantial quantities of toxic waste streams that complicate environmental compliance and increase disposal costs.
Another critical limitation of existing protocols is the difficulty in controlling regioselectivity and preventing polymerization or oligomerization during the formation of larger ring systems. The entropy penalty associated with forming eight-membered rings often leads to low yields and poor atom economy when using thermal or purely chemical activation methods. Additionally, the sensitivity of many organometallic catalysts to moisture and oxygen requires rigorous exclusion of air and the use of expensive anhydrous solvents, further escalating the operational expenditure. For procurement managers and supply chain heads, these factors translate into longer lead times, higher raw material costs, and increased risk of batch-to-batch variability, making the reliable sourcing of such complex intermediates a persistent challenge.
The Novel Approach
In stark contrast, the electrochemical methodology described in patent CN115652344A offers a transformative solution by replacing chemical oxidants with traceless electrons. This metal-free strategy utilizes a constant current electrolysis setup to drive the reaction, effectively bypassing the need for expensive transition metal catalysts entirely. The process operates under mild conditions, typically at room temperature and under an air atmosphere, which drastically reduces energy consumption and eliminates the need for specialized inert gas handling equipment. By generating selenyl radicals directly at the anode surface, the reaction achieves high efficiency in the radical coupling-cyclization step, leading to the formation of the eight-membered ring with impressive selectivity. This novel approach not only simplifies the reaction workflow but also enhances the overall safety profile by avoiding the use of potentially explosive or hazardous chemical oxidants.
From a commercial perspective, this shift to electrochemical synthesis provides substantial advantages in terms of cost reduction in pharmaceutical intermediate manufacturing. The elimination of precious metal catalysts removes a major cost driver and simplifies the downstream processing, as there is no need for extensive metal scavenging procedures. The use of simple electrolytes like nBu4NPF6 and common solvents like dichloromethane ensures that raw materials are readily available and affordable. Moreover, the scalability of electrochemical processes is well-established, allowing for seamless transition from milligram-scale discovery to kilogram or ton-scale production. This robustness ensures a stable supply chain, reducing the risk of disruptions caused by the scarcity of specific catalytic reagents and providing a more predictable manufacturing timeline for high-purity selenium-containing heterocycles.
Mechanistic Insights into Electrochemical Radical Coupling-Cyclization
The mechanistic pathway of this transformation is a fascinating example of how electrochemical potential can be harnessed to control radical reactivity with high precision. The process initiates at the anode, where the diselenide reagent undergoes single-electron oxidation to generate a selenyl radical cation, which rapidly fragments to yield the neutral selenyl radical species. This electrophilic radical then selectively attacks the terminal double bond of the butenyl chain on the sulfonamide substrate, forming a carbon-centered radical intermediate. This step is crucial as it sets the stage for the subsequent ring-closing event. The resulting alkyl radical is positioned in close proximity to the internal alkene moiety attached to the aromatic ring, facilitating a rapid intramolecular addition. This cyclization step forms the new carbon-carbon bond that establishes the eight-membered ring framework, generating a new benzylic radical intermediate.
Following the cyclization, the final step involves the oxidation of the benzylic radical intermediate, likely occurring at the anode or via interaction with another radical species, followed by deprotonation to restore aromaticity or establish the final double bond within the ring system. Simultaneously, at the cathode, protons derived from the solvent or substrate are reduced to evolve hydrogen gas, completing the electrical circuit without generating harmful byproducts. This elegant mechanism ensures that the overall redox balance is maintained solely by the flow of electrons. For R&D directors, understanding this mechanism is vital for optimizing reaction parameters such as current density and electrode material. The use of platinum electrodes is preferred due to their stability and efficiency in promoting the initial oxidation of the diselenide without undergoing corrosion themselves. The choice of supporting electrolyte, specifically nBu4NPF6, is also critical to ensure sufficient conductivity in the organic solvent while remaining chemically inert under the oxidative conditions.
Impurity control is inherently superior in this electrochemical system due to the absence of metal catalysts that often promote side reactions like homocoupling or beta-hydride elimination. The radical nature of the reaction is highly selective for the specific alkene geometries present in the substrate, minimizing the formation of regioisomers. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups that might be present on the aromatic rings, such as halogens or methoxy groups. This high level of chemoselectivity results in a cleaner crude reaction mixture, which significantly eases the burden on purification teams. The ability to tolerate a wide range of substituents on both the sulfonamide substrate and the diselenide reagent demonstrates the versatility of this mechanistic platform, making it a powerful tool for generating diverse libraries of selenium-containing analogs for biological screening.
How to Synthesize Eight-Membered Selenium Heterocycles Efficiently
The practical implementation of this electrochemical synthesis is straightforward and amenable to standard laboratory equipment, making it accessible for process development teams. The general procedure involves dissolving the N-(but-3-en-1-yl)-4-methyl-N-(2-(1-phenylvinyl)phenyl)benzenesulfonamide substrate and the chosen diselenide reagent in dichloromethane, followed by the addition of the supporting electrolyte nBu4NPF6. The mixture is then subjected to constant current electrolysis using platinum plates as both anode and cathode. Monitoring the reaction progress via thin-layer chromatography (TLC) allows for precise determination of the endpoint, typically ensuring complete consumption of the starting material. Upon completion, the reaction is quenched with a mild base such as saturated sodium bicarbonate solution, and the product is extracted into the organic phase. The final isolation is achieved through standard flash column chromatography, yielding the pure eight-membered selenium-containing benzoazacycle.
- Prepare the reaction mixture by combining N-(but-3-en-1-yl)-4-methyl-N-(2-(1-phenylvinyl)phenyl)benzenesulfonamide substrate, diselenide reagent, and nBu4NPF6 electrolyte in dichloromethane (DCM) solvent.
- Conduct constant current electrolysis at 2 mA using Platinum (Pt) electrodes under air atmosphere at room temperature until the starting material is fully consumed.
- Quench the reaction with saturated sodium bicarbonate, extract with dichloromethane, concentrate the organic layer, and purify the residue via flash column chromatography to isolate the target heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology translates into tangible strategic benefits that go beyond mere technical novelty. The most significant advantage is the drastic simplification of the supply chain for raw materials. By eliminating the dependence on scarce and price-volatile precious metal catalysts, manufacturers can insulate themselves from market fluctuations and geopolitical supply risks associated with mining and refining these metals. The reagents used in this process, such as diselenides and simple sulfonamides, are commodity chemicals that can be sourced from multiple vendors, ensuring a robust and competitive supply base. This diversification of suppliers enhances negotiation leverage and secures long-term availability of critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free electrochemical process is profound. The removal of expensive transition metal catalysts directly lowers the bill of materials, while the absence of stoichiometric oxidants reduces waste disposal costs. Furthermore, the simplified workup procedure, which does not require specialized metal scavenging resins or complex filtration steps, reduces labor time and consumable usage. The energy efficiency of running electrolysis at low constant currents also contributes to lower utility costs compared to energy-intensive thermal processes. These cumulative savings allow for a more competitive pricing structure for the final high-purity pharmaceutical intermediates, enhancing margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Operational reliability is significantly improved due to the robustness of the electrochemical setup. The reaction tolerates air and moisture, removing the need for expensive glovebox operations or rigorous solvent drying protocols that often cause delays in production scheduling. The scalability of the process means that production capacity can be increased simply by adding more electrolytic cells or increasing electrode surface area, without the need for redesigning the entire chemical process. This flexibility ensures that supply can be ramped up quickly to meet sudden increases in demand, reducing lead time for high-purity selenium-containing heterocycles and preventing stockouts that could halt downstream drug formulation.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology offers a clear path to sustainable manufacturing. The generation of hydrogen gas as the only byproduct at the cathode is benign and easily managed, contrasting sharply with the toxic heavy metal waste generated by traditional methods. This aligns with increasingly strict global environmental regulations and corporate sustainability goals. The ease of waste treatment and the reduced hazard profile of the reagents simplify permitting and compliance processes, accelerating the time to market for new products. The ability to scale this green process commercially demonstrates a commitment to responsible chemistry, which is a valuable asset for brand reputation and regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of eight-membered selenium-containing benzoazacycles. These answers are derived directly from the experimental data and beneficial effects reported in the underlying patent literature, providing clarity for technical stakeholders evaluating this technology for integration into their supply chains.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed synthesis?
A: This method eliminates the need for expensive transition metal catalysts and external chemical oxidants, utilizing traceless electrons instead. This significantly simplifies downstream purification by removing heavy metal residues and reduces the environmental footprint associated with stoichiometric oxidant waste.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (room temperature, constant current) and uses readily available substrates. The absence of sensitive metal catalysts and the use of simple electrolytic cells facilitate easier scale-up from laboratory to industrial manufacturing scales.
Q: What is the substrate scope for this selenium-containing heterocycle synthesis?
A: The method demonstrates a wide substrate tolerance, accommodating various substituents on the benzene rings including halogens (Cl, Br, F), alkyl groups (methyl), and electron-donating groups (methoxy), as well as different diselenide reagents, allowing for diverse library generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eight-Membered Selenium Heterocycle Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115652344A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity eight-membered selenium heterocycles that empower your drug discovery and development programs with reliable quality and consistency.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this metal-free route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of these valuable selenium-containing building blocks for your next generation of therapeutic agents.
