Advanced Asymmetric Catalysis for Commercial Scale Ruxolitinib Production
Advanced Asymmetric Catalysis for Commercial Scale Ruxolitinib Production
The pharmaceutical landscape for Janus kinase (JAK) inhibitors continues to evolve, driven by the critical need for efficient, scalable, and cost-effective manufacturing processes for high-value active pharmaceutical ingredients (APIs). Patent CN114456181A discloses a groundbreaking preparation method for luccotinib, widely known as Ruxolitinib, which represents a significant technological leap over prior art synthesis routes. This novel methodology leverages advanced asymmetric catalysis to construct the crucial chiral center early in the synthetic sequence, thereby bypassing the traditional bottlenecks associated with racemic resolution. By shifting from separation-based chirality introduction to direct asymmetric synthesis, this process offers a robust pathway for producing high-purity intermediates that meet the stringent quality standards required for global regulatory submission. The structural integrity and stereochemical purity of the final compound are paramount for its efficacy as a selective JAK1/JAK2 tyrosine kinase inhibitor used in treating myelofibrosis.
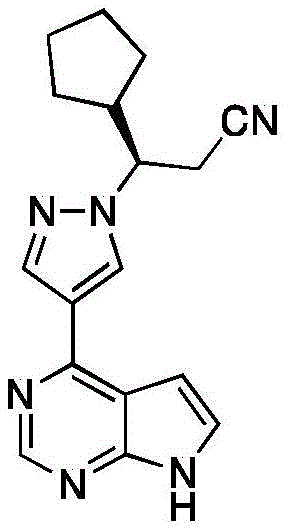
Ruxolitinib, chemically defined as (R)-3-(4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-pyrazol-1-yl)-3-cyclopentylpropanenitrile, has established itself as a cornerstone therapy for intermediate or high-risk myelofibrosis. The commercial success of this molecule, with global sales reaching billions, underscores the immense market demand for reliable supply chains capable of delivering consistent quality. However, the complexity of its molecular architecture, particularly the stereogenic center adjacent to the nitrile group, has historically posed significant challenges for process chemists. The invention detailed in CN114456181A addresses these challenges by introducing a streamlined three-step sequence starting from cyclopentyl acrylonitrile. This approach not only simplifies the operational workflow but also fundamentally alters the economic model of production by removing low-efficiency purification steps that have long plagued the manufacturing of this critical oncology and hematology medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ruxolitinib has been hindered by reliance on inefficient chiral separation techniques that drastically reduce overall yield and increase production costs. For instance, the route described in patent WO2007070514A1 utilizes a Horner-Wadsworth-Emmons reaction followed by a chiral preparative column to isolate the desired enantiomer. This reliance on chromatographic resolution is inherently problematic for industrial scale-up due to the low throughput of chiral columns, the high consumption of expensive chiral stationary phases, and the significant solvent waste generated during the process. Furthermore, another common approach reported in patent CN108699063B involves the chemical resolution of racemic intermediates using D-tartaric acid. While chemical resolution is a classic technique, it suffers from a theoretical maximum yield of 50% for the desired isomer unless dynamic kinetic resolution is employed, which adds further complexity. These traditional methods result in poor atom economy, extended processing times, and a substantial environmental footprint, making them less attractive for modern, sustainability-focused pharmaceutical manufacturing.
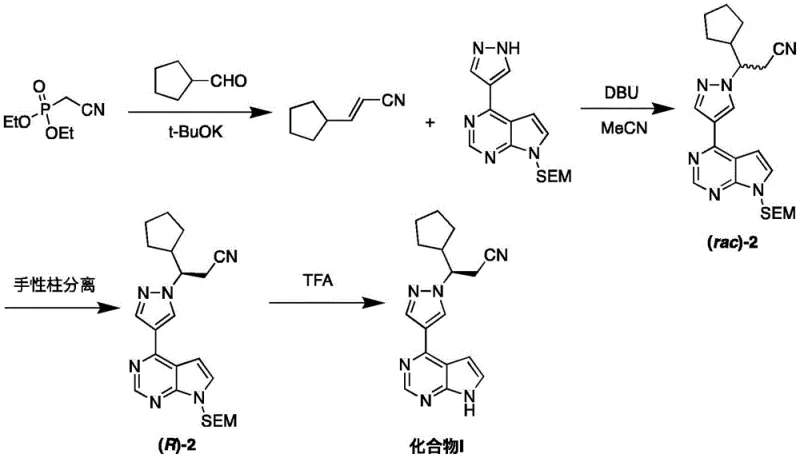
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in CN114456181A employs a direct asymmetric catalytic strategy that establishes chirality with high precision from the outset. The core innovation lies in the copper-catalyzed asymmetric borylation of cyclopentyl acrylonitrile, which generates a chiral organoboron intermediate that is subsequently oxidized to the corresponding chiral alcohol. This transformation is achieved using specialized chiral ferrocenyl phosphine ligands in conjunction with cuprous salts, enabling enantiomeric excess (ee) values exceeding 98% without the need for downstream resolution. By avoiding the formation of racemic mixtures entirely, this route effectively doubles the theoretical yield compared to resolution-based strategies and eliminates the capital expenditure associated with chiral separation infrastructure. The subsequent coupling of this chiral fragment with the heterocyclic core proceeds smoothly under mild basic conditions, preserving the stereochemical integrity throughout the synthesis and delivering the final API with purity levels greater than 99%.
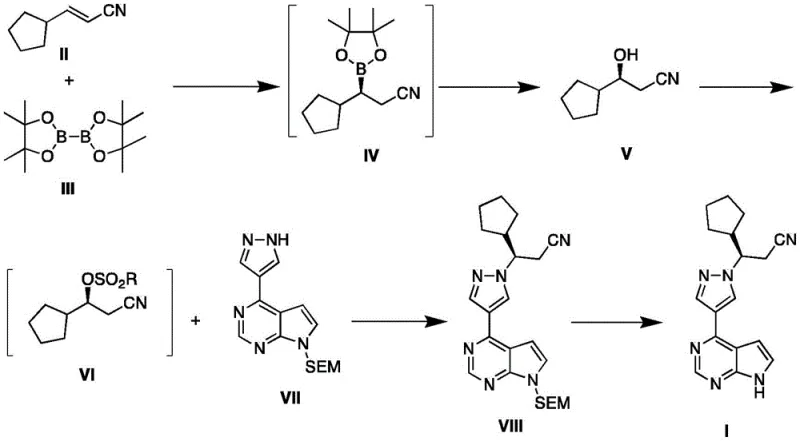
Mechanistic Insights into Copper-Catalyzed Asymmetric Borylation
The heart of this innovative synthesis is the enantioselective borylation reaction, which relies on a finely tuned catalytic cycle involving copper(I) species and bulky chiral ligands. The mechanism initiates with the formation of an active copper-boryl species through the reaction of a cuprous salt, such as cuprous iodide or cuprous acetate, with bis(pinacolato)diboron in the presence of a base like potassium tert-butoxide. The chiral ligand, typically a ferrocenyl-based phosphine such as (S)-(-)-1-[(R)-2-diphenylphosphino]ferrocenyl ethyldicyclohexyl phosphine, creates a sterically demanding environment around the metal center. This chiral pocket dictates the facial selectivity of the addition of the copper-boryl bond across the carbon-carbon double bond of cyclopentyl acrylonitrile. The steric bulk of the ligand ensures that the boron atom adds to the less hindered face of the alkene while the copper adds to the more hindered face, or vice versa depending on the specific ligand geometry, resulting in the preferential formation of one enantiomer of the organoboron intermediate. This step is critical as it sets the absolute configuration of the molecule, which is maintained throughout the subsequent oxidative workup where the carbon-boron bond is converted to a carbon-oxygen bond using oxidants like sodium perborate or Oxone.
Impurity control in this process is inherently superior due to the high stereoselectivity of the initial catalytic step. In traditional resolution routes, the unwanted enantiomer is often carried through several steps before being discarded, leading to the accumulation of difficult-to-remove diastereomeric impurities in later stages. In this catalytic route, the generation of the wrong enantiomer is suppressed at the source, with ee values consistently reported above 94% and often reaching 98% or higher in optimized examples. The subsequent activation of the chiral alcohol via mesylation or tosylation, followed by nucleophilic substitution with the protected pyrazole-pyrimidine fragment, proceeds with inversion or retention of configuration depending on the specific conditions, but critically, without racemization. The final deprotection step using reagents like boron trifluoride etherate or lithium fluoroborate cleanly removes the SEM protecting group to reveal the free NH of the pyrrole ring. The rigorous control over stereochemistry at the very first step ensures that the final crystallization yields a product with an ee value of 99.5% or greater, meeting the strict impurity profiles required for oncology drugs where chiral purity is directly linked to safety and efficacy.
How to Synthesize Ruxolitinib Efficiently
The synthesis of Ruxolitinib via this patented method involves a logical sequence of transformations designed for operational simplicity and high yield. The process begins with the asymmetric borylation of cyclopentyl acrylonitrile to generate the chiral alcohol intermediate, followed by activation and coupling with the heterocyclic core, and concludes with deprotection and crystallization. This streamlined workflow minimizes unit operations and reduces the potential for yield loss associated with multiple isolation steps. For process chemists and manufacturing teams looking to implement this technology, the detailed standardized synthetic procedures, including specific reagent ratios, temperature controls, and workup protocols, are outlined in the comprehensive guide below.
- Perform asymmetric borylation of cyclopentyl acrylonitrile using a chiral copper catalyst and pinacol borane, followed by oxidation to obtain the chiral alcohol intermediate.
- Activate the chiral alcohol with sulfonyl chloride and couple it with the protected pyrrolo-pyrimidine-pyrazole core under basic conditions.
- Remove the SEM protecting group using a fluoride source or Lewis acid, followed by recrystallization to yield final Ruxolitinib with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric catalytic route offers transformative advantages in terms of cost structure and supply reliability. The elimination of chiral chromatography and chemical resolution steps removes two of the most significant cost drivers in the traditional manufacturing of Ruxolitinib. Chiral columns are expensive consumables with limited lifespans, and resolving agents like tartaric acid require stoichiometric amounts and generate substantial waste streams that must be treated. By replacing these batch-intensive separation processes with a catalytic transformation, the overall material cost is significantly reduced, and the throughput of the manufacturing facility is drastically increased. This efficiency gain translates directly into a more competitive pricing structure for the final API, allowing pharmaceutical companies to better manage their cost of goods sold (COGS) while maintaining healthy margins in a competitive therapeutic area.
- Cost Reduction in Manufacturing: The shift from resolution-based synthesis to direct asymmetric catalysis fundamentally improves the atom economy of the process. In traditional routes, nearly half of the synthesized material (the unwanted enantiomer) is discarded, representing a total loss of raw materials and processing energy. The new catalytic route utilizes nearly all starting materials to form the desired product, leading to substantial cost savings on raw materials. Furthermore, the avoidance of specialized chiral separation equipment reduces capital expenditure and maintenance costs, while the use of common solvents like methanol, ethyl acetate, and tetrahydrofuran simplifies solvent recovery and recycling operations. These factors combine to create a leaner, more cost-effective manufacturing process that is resilient to fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications like Ruxolitinib, and this new route enhances reliability by simplifying the supply chain for key intermediates. The starting material, cyclopentyl acrylonitrile, is a commodity chemical that is readily available from multiple global suppliers, reducing the risk of single-source bottlenecks. Additionally, the robustness of the catalytic system, which tolerates mild reaction conditions and standard laboratory glassware, means that production can be easily scaled or transferred between different manufacturing sites without the need for highly specialized infrastructure. This flexibility ensures that manufacturers can respond rapidly to changes in market demand, preventing shortages and ensuring consistent availability of the drug for patients worldwide.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern green chemistry principles. The reaction conditions are mild, typically operating between 0°C and 50°C, which reduces energy consumption compared to processes requiring cryogenic cooling or high-temperature reflux. The absence of heavy metal catalysts (using copper instead of precious metals like palladium or rhodium in some alternative routes) and the elimination of large volumes of chromatographic solvent waste significantly lower the environmental footprint of the synthesis. This makes the process easier to permit and operate within strict environmental regulations, facilitating smoother scale-up from pilot plant to commercial tonnage production without encountering regulatory hurdles related to waste disposal or emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and benefits of the technology for potential partners and licensees.
Q: How does the new asymmetric catalytic route improve upon traditional resolution methods?
A: The new route utilizes a chiral copper catalyst to directly establish stereocenters with high enantiomeric excess (>98% ee), completely eliminating the need for inefficient chiral chromatographic separation or chemical resolution with tartaric acid, thereby significantly improving atom economy and yield.
Q: What are the critical reaction conditions for the key borylation step?
A: The key borylation step operates under mild conditions (0-50°C) using cuprous salts and specialized ferrocenyl phosphine ligands in methanol, ensuring safety and ease of scale-up without requiring cryogenic temperatures or high-pressure equipment.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process avoids hazardous reagents and complex purification steps like chiral HPLC, utilizing standard organic solvents and readily available starting materials, making it highly robust for multi-kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruxolitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic methodologies to secure a competitive edge in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the asymmetric catalytic route described in CN114456181A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess values that exceed industry standards. We are committed to delivering high-purity Ruxolitinib intermediates and API that support the development of next-generation JAK inhibitor therapies.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Together, we can optimize the manufacturing of this vital medicine, ensuring affordability and availability for patients while driving value for your organization through superior process chemistry.
