Scalable Two-Step Synthesis of Diarylfluorene Intermediates for Advanced OLED Manufacturing
Scalable Two-Step Synthesis of Diarylfluorene Intermediates for Advanced OLED Manufacturing
The rapid evolution of the organic optoelectronics sector demands intermediates that balance high spectral stability with manufacturability. Patent CN101643381A introduces a transformative preparation method for diarylfluorene intermediates, specifically targeting the needs of organic electroluminescence and photovoltaic applications. This technology addresses the critical bottleneck of morphological stability in OLED materials by utilizing a non-planar 9,9-diarylfluorene structure that effectively prevents fluorescence quenching caused by π-system stacking. Unlike legacy synthetic routes, this innovation leverages a direct condensation strategy between arylamines and fluorenone derivatives, bypassing the hazardous and cumbersome preparation of Grignard reagents. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more cost-efficient supply chains for high-performance display materials. 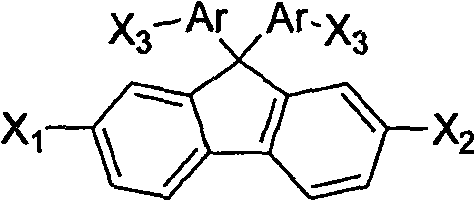
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9,9-diarylfluorenes has relied heavily on the traditional Grignard reagent-Friedel-Crafts two-step methodology. This conventional pathway necessitates the preparation of aromatic Grignard reagents, which are notoriously sensitive to moisture and oxygen, requiring rigorous anhydrous conditions and specialized equipment that increases capital expenditure. Furthermore, the reaction yields a tertiary alcohol intermediate that is notoriously difficult to purify, often requiring complex separation techniques to remove unreacted magnesium salts and side products. The subsequent acid-catalyzed Friedel reaction to close the ring adds another layer of complexity, often resulting in lower overall yields and significant waste generation. For large-scale manufacturers, these factors translate into extended lead times, higher safety risks, and inflated production costs that hinder the commercial viability of next-generation organic electronic devices.
The Novel Approach
The patented methodology offers a streamlined alternative by employing a direct acid-catalyzed condensation between arylamines (or their hydrochloride salts) and fluorenone derivatives. This approach fundamentally alters the reaction landscape by utilizing stable, solid-state reagents that do not require the stringent exclusion of water inherent to organometallic chemistry. The process operates effectively at elevated temperatures between 120-220°C, facilitating a robust reaction kinetics profile that is easier to control in large reactors. By eliminating the unstable Grignard step, the process drastically simplifies the workflow to a manageable two-step sequence involving condensation followed by diazotization. This not only enhances operator safety but also improves the atom economy of the synthesis, ensuring that a higher proportion of raw materials are converted into the valuable final product, thereby aligning perfectly with green chemistry principles and modern sustainability mandates.
Mechanistic Insights into Acid-Catalyzed Condensation and Diazotization
The core of this synthetic breakthrough lies in the efficient formation of the C-C bonds at the 9-position of the fluorene backbone through an acid-catalyzed mechanism. In the first stage, arylamine and arylamine hydrochloride react with fluorenone under thermal conditions, where the acid catalyst activates the carbonyl group of the fluorenone towards nucleophilic attack by the amine. This results in the formation of a high-purity diarylaminofluorene intermediate after a straightforward workup involving alkalization and filtration. The use of arylamine hydrochlorides in a 3-8 fold molar excess serves a dual purpose: it acts as both a reactant and an acid source, driving the equilibrium forward while suppressing side reactions. This mechanistic elegance ensures that the intermediate obtained is of sufficient purity to proceed directly to the next step without extensive chromatographic purification, a common bottleneck in fine chemical synthesis. 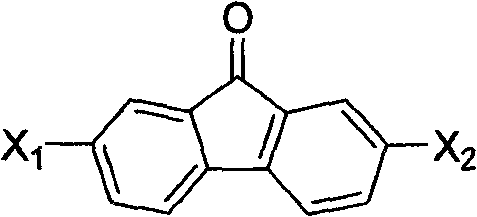
The second mechanistic phase involves a diazotization-reduction sequence using alkyl nitrites in a dichloromethane solvent system. The diarylaminofluorene intermediate undergoes diazotization at mild temperatures ranging from 0-50°C, generating a diazonium species that is subsequently reduced to yield the final 9,9-diarylfluorene structure. This step is critical for removing the amino functionality and establishing the final aromatic character of the molecule. The choice of solvent and the controlled addition of alkyl nitrite allow for precise regulation of the reaction exotherm, minimizing the formation of tar-like byproducts. Following the reaction, a simple extraction and recrystallization protocol using petroleum ether and dichloromethane mixtures effectively removes residual impurities and colored byproducts. This rigorous control over the reaction environment ensures that the final product meets the stringent purity specifications required for electronic grade materials, where trace metal or organic impurities can severely degrade device performance and lifetime.
How to Synthesize 9,9-Diarylfluorene Efficiently
Implementing this synthesis route requires careful attention to temperature control and stoichiometry to maximize yield and purity. The process begins with the thermal condensation of the amine and fluorenone components, followed by a distinct diazotization step that converts the amino-intermediate into the target hydrocarbon. Detailed standard operating procedures regarding reagent grades, specific heating rates, and crystallization parameters are essential for reproducible results in a GMP environment. The following guide outlines the critical operational phases derived from the patent examples to assist technical teams in process validation.
- React arylamine and arylamine hydrochloride with fluorenone derivatives under acid catalysis at 120-220°C for 2-8 hours to form the diarylaminofluorene intermediate.
- Purify the intermediate via alkalization, filtration, and ethanol washing to remove pigments and impurities.
- React the purified intermediate with alkyl nitrite in dichloromethane under reflux (0-50°C) for 0.5-3 hours, followed by recrystallization to obtain the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages beyond mere technical feasibility. The elimination of Grignard reagents removes the dependency on high-purity magnesium turnings and dry ether solvents, which are subject to volatile pricing and strict transportation regulations due to flammability. Instead, the process relies on commodity chemicals like aniline and fluorenone, which are produced globally at massive scales, ensuring a stable and resilient supply base. This shift significantly de-risks the supply chain against disruptions caused by specialty reagent shortages. Furthermore, the simplified workup procedure, which relies on filtration and washing rather than complex distillation or chromatography, reduces the energy consumption and solvent usage per kilogram of product. This efficiency translates directly into a lower cost of goods sold (COGS), allowing downstream device manufacturers to remain competitive in the rapidly evolving display market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive, hazard-prone organometallic reagents with inexpensive arylamine salts. By avoiding the need for specialized anhydrous reactors and the associated safety infrastructure, capital investment for production facilities is substantially lowered. Additionally, the high yield and simplicity of purification mean that less raw material is wasted, and labor hours spent on complex separations are minimized. This cumulative effect results in a drastic reduction in manufacturing overheads, making the production of high-value OLED intermediates more economically sustainable without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term product lifecycles in the electronics industry. Since the key starting materials—fluorenones and arylamines—are widely available from multiple global suppliers, the risk of single-source bottlenecks is effectively mitigated. The robustness of the reaction conditions also means that production can be scaled up or down rapidly in response to market demand without the need for lengthy process re-optimization. This flexibility ensures consistent delivery schedules for clients, supporting just-in-time manufacturing models and reducing the need for excessive inventory buffering of finished goods.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a cleaner profile compared to traditional routes. The absence of heavy metal catalysts and the reduction in hazardous waste streams simplify wastewater treatment and disposal protocols. The ability to recover and recycle arylamine hydrochloride from the filtrate further enhances the atom economy and reduces the environmental footprint. These factors facilitate easier regulatory compliance in jurisdictions with strict environmental laws, smoothing the path for commercial scale-up from pilot plants to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding scalability, purity, and substrate scope, providing clarity on how this technology meets the rigorous demands of the optoelectronic industry. These insights are derived directly from the experimental data and claims within the patent documentation.
Q: How does this novel synthesis method improve upon traditional Grignard routes?
A: Unlike traditional methods requiring moisture-sensitive Grignard reagents and complex tertiary alcohol purification, this patented process utilizes stable arylamine hydrochlorides and fluorenones. It eliminates the need for strict anhydrous conditions and simplifies the workup to basic filtration and washing, significantly reducing operational complexity and safety risks associated with organometallic reagents.
Q: What represents the key advantage for scaling production of OLED materials?
A: The process employs cheap, commercially available raw materials such as aniline derivatives and fluorenone, avoiding expensive catalysts. The reaction conditions (150-190°C) are robust, and the purification relies on standard recrystallization rather than column chromatography, making it highly suitable for multi-kilogram to ton-scale manufacturing required by the display industry.
Q: Can this method produce halogenated derivatives for further functionalization?
A: Yes, the patent explicitly demonstrates the successful synthesis of 2-bromo and 2,7-dibromo substituted diarylfluorenes. These halogenated intermediates are critical building blocks for subsequent cross-coupling reactions (like Suzuki or Buchwald-Hartwig) used to construct complex emissive layers in high-performance organic light-emitting diodes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylfluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of diarylfluorene intermediate meets the exacting standards required for high-performance OLED and photovoltaic applications. Our commitment to quality assurance guarantees that the materials you receive will perform reliably in your final devices, minimizing failure rates and maximizing efficiency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific product requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this greener, more efficient route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global organic electronics market through superior supply chain solutions.
