Advanced Oxidative Rearrangement Strategy for High-Purity Tetrahydrocarboline Pharmaceutical Intermediates
Introduction to Novel Tetrahydrocarboline Scaffold Synthesis
The pharmaceutical industry constantly seeks efficient pathways to construct complex heterocyclic systems that serve as privileged structures in medicinal chemistry. Patent CN110835341A introduces a groundbreaking methodology for the preparation of oxidative coupling rearrangement products containing a tetrahydrocarboline skeleton. This technology addresses the longstanding challenge of functionalizing tetrahydrocarboline derivatives at the late stage of synthesis, a critical step for generating diverse libraries of bioactive molecules. By leveraging a mild peroxide rearrangement condition, this invention enables the direct coupling of oxidized tetrahydrocarboline intermediates with indole derivatives. The resulting compounds, with molecular weights around 500, represent a novel class of chemical entities with demonstrated potential in oncology. For R&D teams focused on antitumor agents, this patent provides a robust platform for accessing unique chemical space that was previously difficult to explore due to reactivity control issues inherent to the tetrahydrocarboline core.
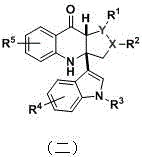
The significance of this technology extends beyond mere academic interest; it offers a practical solution for the commercial scale-up of complex pharmaceutical intermediates. Traditional methods often suffer from low selectivity or require hazardous reagents, but this approach utilizes relatively benign oxidants and rearrangement promoters. The ability to introduce diverse substituents (R groups) on both the tetrahydrocarboline and indole moieties allows for extensive structure-activity relationship (SAR) studies. This flexibility is paramount for reliable pharmaceutical intermediate supplier networks aiming to support drug discovery pipelines. The patent explicitly highlights the simplicity of the preparation method, noting that it does not require high temperature, high pressure, or strong acidic/alkaline conditions, which translates directly to reduced operational risks and lower infrastructure costs for manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of tetrahydrocarboline alkaloids has been plagued by significant synthetic hurdles. The peculiar skeleton structure of these derivatives dictates that their chemical reactivity is not easy to control, often leading to unpredictable side reactions or decomposition under standard conditions. Conventional approaches to modifying these cores frequently necessitate harsh reaction environments, such as elevated temperatures exceeding 100 °C or the use of corrosive strong acids and bases, which can degrade sensitive functional groups present on the molecule. Furthermore, traditional oxidative coupling strategies often rely on expensive transition metal catalysts that require rigorous removal steps to meet stringent purity specifications for pharmaceutical applications. These legacy methods typically result in lower overall yields, sometimes struggling to exceed 50%, which drastically impacts the cost of goods sold (COGS) and limits the availability of material for preclinical testing. The inability to efficiently perform late-stage functionalization also restricts the speed at which medicinal chemists can iterate on lead compounds, slowing down the entire drug discovery timeline.
The Novel Approach
In stark contrast, the methodology described in CN110835341A represents a paradigm shift towards greener and more efficient synthesis. The novel approach utilizes tert-butyl hydroperoxide for the initial oxidation, followed by a coupling with indoles and a subsequent rearrangement mediated by mild reagents like ascorbic acid (Vitamin C) or methanesulfonic acid. This sequence operates effectively at room temperature or mild heating (up to 60 °C), significantly reducing energy consumption compared to thermal-intensive legacy processes. The use of ascorbic acid as a rearrangement promoter is particularly innovative, as it is a non-toxic, inexpensive, and readily available commodity chemical, eliminating the need for precious metal catalysts. This shift not only simplifies the workup procedure—often requiring only filtration and drying—but also enhances the environmental profile of the synthesis. For procurement managers, this means cost reduction in pharmaceutical intermediate manufacturing is achieved through both raw material savings and simplified processing. The high yields reported, ranging generally above 60% and reaching up to 99% in optimized cases, ensure that material throughput is maximized, supporting robust supply chains for downstream API production.
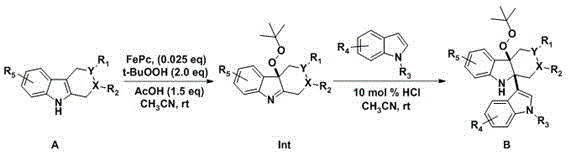
Mechanistic Insights into FePc-Catalyzed Oxidation and Peroxide Rearrangement
The core of this technological advancement lies in the precise control of oxidative species and their subsequent rearrangement. The process initiates with the oxidation of the tetrahydrocarboline precursor using iron phthalocyanine (FePc) as a catalyst in the presence of tert-butyl hydroperoxide (t-BuOOH). FePc is a robust, biomimetic catalyst known for its ability to activate peroxides selectively, generating reactive oxygen species that functionalize the electron-rich positions of the tetrahydrocarboline ring without causing over-oxidation or ring cleavage. This step forms a crucial peroxide intermediate (Int), which retains the integrity of the complex polycyclic system while introducing the necessary reactivity for the next stage. The subsequent coupling with indole derivatives occurs under acidic conditions (e.g., HCl in acetonitrile), where the electrophilic nature of the oxidized intermediate facilitates nucleophilic attack by the indole. This creates a new carbon-carbon bond linking the two heterocyclic systems, forming the precursor B. The final rearrangement step is the most critical, where reagents like ascorbic acid induce a skeletal reorganization. This likely involves the reduction of the peroxide linkage and a concomitant migration of bonds to establish the stable, fused tetrahydrocarboline-indole architecture found in the final products. Understanding this mechanism is vital for R&D directors aiming to replicate or modify the pathway for analog synthesis.
Impurity control is inherently built into this mechanistic design. The use of specific catalysts like FePc at low loading (0.025 equivalents) minimizes the formation of metal-associated impurities, which are often difficult to purge in later stages. Furthermore, the mild conditions of the rearrangement step prevent the formation of thermal degradation products that commonly plague high-temperature syntheses. The patent data indicates that the reaction can be monitored easily, and the products are often solids that can be purified by simple filtration or column chromatography. This high level of chemoselectivity ensures that the final high-purity pharmaceutical intermediates meet the rigorous standards required for biological testing. For quality assurance teams, the predictability of the impurity profile reduces the burden on analytical method development and validation. The ability to tolerate various substituents (halogens, alkyl groups, alkoxy groups) on both the indole and tetrahydrocarboline rings without compromising the rearrangement efficiency further underscores the robustness of this chemical transformation.
How to Synthesize Tetrahydrocarboline Derivatives Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the oxidant and the choice of rearrangement promoter. The general protocol involves dissolving the starting tetrahydrocarboline material in a solvent like acetonitrile, followed by the slow addition of the iron catalyst and acetic acid. Temperature control is critical during the oxidation phase, typically maintained around 5 °C to prevent runaway exotherms, before adding the aqueous tert-butyl hydroperoxide. Once the intermediate is formed and isolated, it is reacted with the chosen indole derivative. The final transformation to the target scaffold can be tuned by selecting different promoters; for instance, using ascorbic acid in methanol at room temperature yields one class of products, while using methanesulfonic acid in acetonitrile may favor alternative rearrangement pathways or rates. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these results accurately.
- Oxidize complex tetrahydrocarboline precursors using tert-butyl hydroperoxide and iron phthalocyanine catalyst to form a peroxide intermediate.
- Couple the oxidized intermediate with various substituted indoles under acidic conditions to generate the coupled precursor material.
- Perform the final rearrangement reaction using ascorbic acid, methanesulfonic acid, or sodium borohydride in methanol or acetonitrile to yield the target scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits. The reliance on commodity chemicals such as ascorbic acid, methanesulfonic acid, and sodium borohydride eliminates dependency on scarce or geopolitically sensitive reagents. This diversification of the raw material base enhances supply security, ensuring that production schedules are not disrupted by shortages of specialized catalysts. Moreover, the operational simplicity of the process—often involving stir-and-filter operations without the need for complex distillation or cryogenic cooling—allows for faster batch turnover times. This efficiency is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling faster delivery of materials to clients engaged in urgent drug discovery programs. The high yields observed across a broad substrate scope mean that less starting material is wasted, directly contributing to a more sustainable and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of mild reaction conditions significantly lower the operational expenditure. By avoiding high-pressure reactors and extreme temperatures, facilities can utilize standard glass-lined or stainless-steel equipment, reducing capital investment requirements. The high atom economy and minimal waste generation further decrease disposal costs, aligning with modern green chemistry principles while improving the bottom line.
- Enhanced Supply Chain Reliability: The reagents used in this process, including tert-butyl hydroperoxide and various indoles, are widely available from multiple global suppliers. This redundancy mitigates the risk of single-source dependency. Additionally, the stability of the intermediates and the robustness of the reaction conditions allow for flexible scheduling and inventory management, ensuring consistent availability of critical building blocks for downstream API synthesis.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful synthesis of numerous derivatives in the patent examples. The mild conditions reduce the generation of hazardous byproducts, simplifying wastewater treatment and废气 (exhaust gas) management. This ease of compliance with environmental regulations facilitates smoother regulatory approvals for manufacturing sites, accelerating the path from laboratory bench to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative coupling technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this chemistry into their existing portfolios.
Q: What are the typical reaction yields for this oxidative rearrangement process?
A: According to patent CN110835341A, the reaction yields are generally above 60%, with specific optimized examples achieving yields as high as 99%, demonstrating excellent efficiency for complex scaffold construction.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method utilizes mild conditions (room temperature to 60 °C) and avoids high pressure or strong acids/bases, making it highly scalable and safer for industrial manufacturing compared to traditional harsh protocols.
Q: What is the biological significance of these tetrahydrocarboline derivatives?
A: These compounds exhibit significant antitumor activity against breast cancer (MCF-7), lung cancer (A549), and cervical cancer (Hela) cell lines, serving as potent lead structures for oncology drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrocarboline Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative academic research into commercial reality. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to manufacture these complex tetrahydrocarboline intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative chemistry, ensuring strict adherence to safety and quality protocols. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for preclinical and clinical development. Our team of expert process chemists can further optimize the route described in CN110835341A to maximize yield and minimize cost for your specific project needs.
We invite you to collaborate with us to accelerate your oncology drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Let us be your partner in bringing these promising antitumor candidates from the lab to the clinic.
