Scalable Synthesis of N,N-Bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline for OLED Applications
Scalable Synthesis of N,N-Bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline for OLED Applications
The rapid advancement of organic optoelectronic devices, including organic photoelectric drums (OPC), organic electroluminescent displays (OLED), and organic solar cells (OPE), has created an urgent demand for high-performance charge transport materials. Among these, triphenylamine derivatives stand out due to their excellent hole-transporting properties and thermal stability. Specifically, the compound N,N-bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline exhibits superior performance characteristics attributed to its extended conjugation system, making it a critical component for double-layer photoconductors with high charge retention and low dark decay rates. The synthesis methodology detailed in patent CN101429131B represents a significant technological breakthrough in this field, offering a robust, scalable, and economically viable pathway for producing this high-value electronic chemical intermediate. This report provides a deep technical analysis of the patented process, evaluating its mechanistic advantages, operational feasibility, and commercial potential for global supply chains seeking reliable sources of advanced display materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the methodology described in CN101429131B, the synthesis of N,N-bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline was fraught with significant inefficiencies that hindered large-scale commercial adoption. As documented in earlier literature, such as the work by Yang Xiongwen et al. published in Fine Chemical Industry in 2005, conventional routes typically relied on multi-step sequences starting from diphenylbromomethane. These traditional pathways often necessitated the formation of 2-(4-methylphenyl)-1,1-diphenylethylene followed by complex reduction reactions to generate 4-(2,2-diphenyl)vinylaniline. Such processes were not only characterized by excessively long reaction times but also suffered from inherently low product yields and high operational costs due to the requirement for multiple purification stages and harsh reaction conditions. Furthermore, the accumulation of impurities through these extended synthetic sequences posed severe challenges for achieving the stringent purity specifications required for high-end electronic applications, rendering these methods unsuitable for modern industrial mass production.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach outlined in the patent introduces a streamlined, four-step synthetic strategy that dramatically enhances process efficiency and product quality. This innovative route begins with the conversion of diphenylmethanol to diethyl diphenylmethylphosphonate via a bromination and subsequent Arbuzov reaction, establishing a stable phosphonate precursor. The core scaffold is then constructed through a copper-catalyzed Ullmann-type coupling between aniline and p-iodotoluene, followed by a precise Vilsmeier-Haack formylation to introduce the aldehyde functionality. The final assembly is achieved through a Horner-Wadsworth-Emmons olefination, directly linking the phosphonate and aldehyde fragments to form the target vinyl aniline structure. This modular approach not only reduces the total number of unit operations but also allows for milder reaction conditions and easier control over stereochemistry and impurity profiles, thereby facilitating the cost reduction in electronic chemical manufacturing while ensuring consistent batch-to-batch reproducibility.
Mechanistic Insights into Cu-Catalyzed Coupling and HWE Olefination
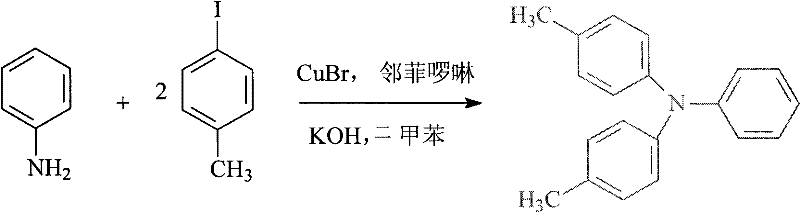
The success of this synthetic route hinges on the precise execution of two critical catalytic transformations: the formation of the triarylamine core and the construction of the stilbene-like double bond. The synthesis of N,N-bis(4-methylphenyl)aniline utilizes a modified Ullmann condensation mediated by cuprous bromide (CuBr) and 1,10-phenanthroline in the presence of potassium hydroxide. This catalytic system is pivotal as it facilitates the carbon-nitrogen bond formation under relatively moderate temperatures ranging from 100°C to 144°C, avoiding the extreme conditions often required for non-catalyzed variants. The mechanistic cycle likely involves the oxidative addition of the aryl iodide to the copper center, followed by coordination with the aniline nitrogen and subsequent reductive elimination to release the coupled product. The use of 1,10-phenanthroline as a ligand stabilizes the copper species, preventing aggregation and deactivation, which is crucial for maintaining high catalytic turnover and minimizing residual metal contamination in the final API intermediate.
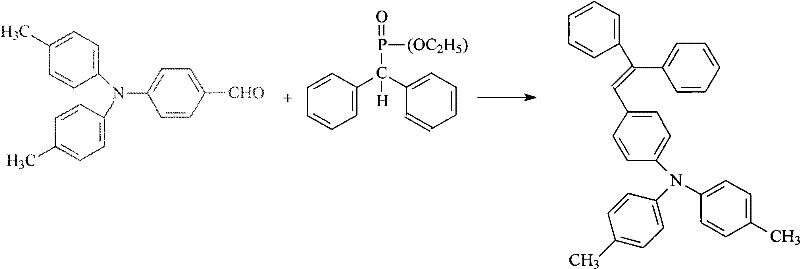
Following the construction of the amine scaffold, the introduction of the formyl group via the Vilsmeier-Haack reaction and the final carbon-carbon bond formation via the Horner-Wadsworth-Emmons (HWE) reaction are executed with high precision. The formylation step employs phosphorus oxychloride and dimethylformamide to generate the reactive chloroiminium salt in situ, which electrophilically attacks the electron-rich aromatic ring para to the amino group. Subsequently, the HWE reaction couples the resulting aldehyde with diethyl diphenylmethylphosphonate under basic conditions using sodium methoxide. This base deprotonates the alpha-carbon of the phosphonate to generate a carbanion, which nucleophilically attacks the aldehyde carbonyl. The resulting betaine intermediate collapses to eliminate diethyl phosphate, driving the formation of the thermodynamically stable trans-alkene product. This sequence ensures the extended conjugation necessary for optimal charge transport properties in the final OLED material.
Impurity control is rigorously managed throughout the mechanistic pathway through specific workup and purification protocols integrated into each step. For instance, in the Ullmann coupling stage, the reaction mixture is treated with toluene and water to facilitate the filtration and recovery of the o-phenanthroline-copper complex, effectively removing the bulk of the transition metal catalyst before it can contaminate downstream processes. Similarly, the aqueous phase is processed to reclaim potassium iodide, turning a potential waste stream into a recoverable resource. In the final olefination step, the addition of deionized water precipitates the crude product, allowing for the removal of soluble phosphate byproducts and excess base. Recrystallization from methanol further refines the solid, eliminating trace organic impurities and isomers. This multi-layered approach to purification ensures that the final high-purity OLED material meets the rigorous standards demanded by the display industry, with HPLC analysis confirming contents consistently above 98%.
How to Synthesize N,N-Bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process is divided into four distinct stages, each requiring specific temperature controls and molar ratios to drive the reactions to completion while minimizing side products. Operators must pay particular attention to the exothermic nature of the Vilsmeier reagent formation and the moisture sensitivity of the phosphonate coupling step. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate this high-efficiency route in a commercial setting. Detailed standardized synthesis steps are provided in the guide below.
- Synthesize diethyl diphenylmethylphosphonate by reacting diphenylmethanol with hydrobromic acid followed by triethyl phosphite at 120-156°C.
- Perform Ullmann-type coupling of aniline and p-iodotoluene using CuBr and phenanthroline catalysts in xylene at 135-144°C to form N,N-bis(4-methylphenyl)aniline.
- Conduct Vilsmeier-Haack formylation using POCl3 and DMF in toluene at 0-80°C to generate N,N-bis(4-methylphenyl)-4-aminobenzaldehyde.
- Execute Horner-Wadsworth-Emmons reaction between the aldehyde and phosphonate ester using sodium methoxide in DMF to yield the final vinyl aniline product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers substantial benefits that extend beyond mere technical feasibility, directly impacting the bottom line and supply chain resilience. The streamlined nature of the process, characterized by fewer unit operations and higher per-step yields, translates into a significantly reduced cost of goods sold (COGS) compared to legacy methods. By eliminating the need for complex reduction steps and utilizing readily available industrial-grade raw materials such as aniline, p-iodotoluene, and diphenylmethanol, manufacturers can mitigate the risks associated with volatile raw material pricing and supply shortages. Furthermore, the ability to recover and reuse key catalysts and solvents, such as the copper-phenanthroline complex and xylene, creates a closed-loop system that minimizes waste disposal costs and environmental liabilities, aligning with modern sustainability mandates.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and efficient resource utilization. By avoiding the use of expensive noble metal catalysts often found in cross-coupling reactions and instead relying on abundant copper salts, the direct material costs are drastically lowered. Additionally, the high yields reported in each step—ranging from approximately 60% to 85%—mean that less raw material is required to produce a kilogram of the final product, compounding the savings across the entire production volume. The simplified purification protocols, which rely on standard crystallization techniques rather than costly chromatography, further reduce processing time and solvent consumption, leading to substantial cost savings in the overall manufacturing budget without compromising product quality.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical electronic chemicals is paramount for downstream device manufacturers, and this synthesis route enhances reliability through the use of commoditized starting materials. Unlike specialized precursors that may have single-source suppliers or long lead times, the inputs for this process are produced globally at massive scales for various industries, ensuring continuous availability. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, also contributes to supply continuity by reducing the likelihood of batch failures or production stoppages. This operational stability allows suppliers to maintain consistent inventory levels and meet tight delivery schedules, reducing lead time for high-purity electronic chemical intermediates essential for just-in-time manufacturing models.
- Scalability and Environmental Compliance: The transition from laboratory benchtop to commercial scale-up of complex organic intermediates is often a bottleneck, but this process is explicitly designed for industrial scalability. The reaction temperatures are moderate, and the exotherms are manageable, allowing for safe operation in large-scale reactors without requiring exotic cooling systems or high-pressure equipment. Moreover, the process generates minimal hazardous waste; the recovery of potassium iodide and copper complexes reduces the load on wastewater treatment facilities. The use of common organic solvents like toluene and methanol, which have well-established recycling infrastructures, ensures that the facility can maintain strict environmental compliance while scaling production capacity from pilot plants to multi-ton annual outputs seamlessly.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial viability of this synthesis method, we have compiled the following answers based on the detailed data provided in patent CN101429131B. These responses clarify the advantages of this route over prior art, the potential for resource recovery, and the expected quality metrics of the final product. Understanding these factors is essential for stakeholders evaluating the integration of this material into their supply chains or product formulations.
Q: How does this synthesis method improve upon previous techniques reported in literature?
A: Unlike the method reported by Yang Xiongwen et al. which involves long reaction steps and low yields, this patented process utilizes a direct Wittig-Horner approach that significantly simplifies the synthetic route, reduces reaction time, and achieves higher overall yields suitable for industrial production.
Q: Can the solvents and catalysts be recovered to reduce production costs?
A: Yes, the process is designed for industrial applicability with specific provisions for recycling. For instance, the o-phenanthroline-copper complex can be filtered and recovered, and potassium iodide can be reclaimed from the aqueous phase, substantially lowering raw material consumption.
Q: What purity levels can be expected from this manufacturing process?
A: The patented method consistently produces high-purity intermediates and final products. Through recrystallization steps using solvents like petroleum ether, ethyl acetate, and methanol, the final N,N-bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline achieves HPLC purity levels between 98% and 99.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline Supplier
As the global demand for high-performance organic electronics continues to surge, partnering with a technically proficient manufacturer is critical for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in process chemistry to deliver superior electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of market fluctuations. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of N,N-Bis(4-methylphenyl)-4-(2,2-diphenylvinyl)aniline meets the exacting standards required for next-generation OLED and OPC applications.
We invite you to collaborate with us to optimize your material sourcing strategy and accelerate your product development timelines. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our efficient synthesis route can lower your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics and reliable supply capabilities.
