Revolutionizing Chiral Separation with Advanced Monochiral Metal-Organic Frameworks for Industrial Scale
The landscape of chiral separation in the fine chemical and pharmaceutical industries is undergoing a significant transformation driven by the need for higher purity and more sustainable processes. A pivotal advancement in this domain is documented in patent CN103193831A, which details a preparation method for a monochiral metal-organic framework (MOF) material possessing dual functions of chiral separation and photoinduction. This technology represents a paradigm shift from traditional chromatographic methods, offering a solid-state solution that combines the tunability of organic ligands with the stability of inorganic nodes. For R&D directors and procurement specialists, understanding the implications of this material is crucial, as it promises to streamline the purification of critical intermediates such as mandelic acid esters. The ability to achieve enantiomeric excess (ee) values exceeding 90% through simple adsorption mechanisms underscores the potential for this technology to become a cornerstone in modern asymmetric synthesis workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of chiral molecules, particularly in the production of high-purity pharmaceutical intermediates, has relied heavily on derivatized cyclodextrin columns. While effective to a degree, these traditional materials suffer from inherent structural and economic deficiencies that hinder large-scale adoption. The synthesis of cyclodextrin derivatives is often laden with trivial yet costly steps, requiring precise functionalization that drives up the overall manufacturing expense. Furthermore, the packing of these derivatives into chromatographic columns is a complicated process that can lead to inconsistencies in flow dynamics and separation efficiency. From a supply chain perspective, the fragility of these organic-only structures limits their operational lifespan, necessitating frequent replacement and generating substantial chemical waste. The inability of these conventional columns to effectively separate certain challenging substrates, such as mandelate compounds, further restricts their utility in complex synthetic routes where high stereochemical purity is non-negotiable.
The Novel Approach
In stark contrast, the novel approach utilizing monochiral metal-organic frameworks introduces a robust hybrid architecture that addresses the stability and efficiency gaps of legacy technologies. By constructing a porous skeleton through the coordination of chiral organic ligands with metal ions like dysprosium, this method creates a rigid, amphiphilic environment tailored for specific guest molecule recognition. The solvothermal synthesis employed is remarkably straightforward, operating at moderate temperatures around 100°C, which significantly lowers the energy barrier compared to high-pressure alternatives. This structural integrity allows the material to maintain its chiral pores even under repeated usage cycles, ensuring consistent separation performance over time. Moreover, the integration of photoinduction capabilities adds a layer of functionality, enabling the material to not just separate but also induce asymmetric reactions, thereby consolidating multiple processing steps into a single unit operation.
Mechanistic Insights into Solvothermal Coordination and Ligand Design
The core of this technological breakthrough lies in the precise design of the chiral ligand (S)-H4L and its subsequent coordination with metal centers. The synthesis begins with a Suzuki coupling reaction, where a chiral biphenyl backbone is functionalized with carboxylate groups to facilitate metal binding. This step is critical as it establishes the steric environment necessary for chiral recognition; the bulky tert-butyl groups on the biphenyl ring create a confined space that selectively accommodates one enantiomer over the other. Following the coupling, a hydrolysis step converts the ester groups into free carboxylic acids, which are essential for coordinating with the dysprosium and sodium ions. The resulting framework forms a one-dimensional metal chain structure that extends into a three-dimensional porous network, with pore sizes approximately 1.81nm, perfectly sized to trap target molecules like mandelic acid esters through host-guest interactions.
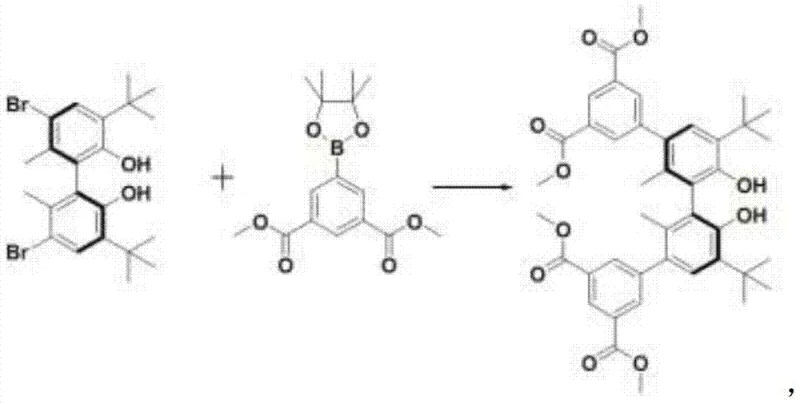
Understanding the coordination chemistry is vital for assessing the material's stability and performance in industrial settings. The dysprosium and sodium atoms adopt distorted octahedral coordination configurations, bridged by the carboxylate groups of the ligand to form stable secondary building units. These metal chains are further linked by the rigid biphenyl ligands, creating a channel system that exhibits strong chirality. The bond distances, ranging from 2.245(6) to 2.358(6) Å for Dy-O and 2.262(5) to 2.419(6) Å for Na-O, fall within rational scopes that ensure structural rigidity without compromising the flexibility needed for guest inclusion. This precise atomic arrangement is what allows the material to achieve high enantiomeric selectivity, as the chiral pockets within the framework interact differently with the R and S enantiomers of the substrate, effectively filtering out the unwanted isomer.
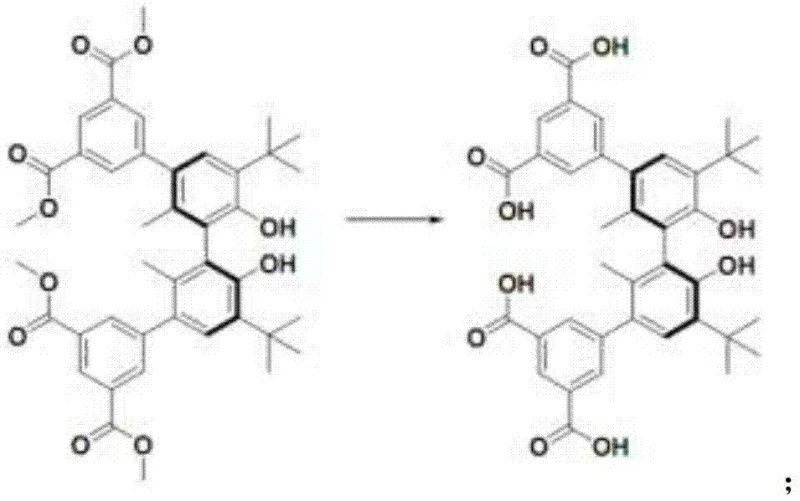
Impurity control in this synthesis is inherently managed through the crystallization process itself. As the MOF forms during the 3-day heating period at 100°C, the thermodynamic stability of the correct crystal lattice favors the exclusion of unreacted ligands or mismatched metal species. The washing steps with ether and subsequent drying remove surface-adsorbed impurities, leaving behind a highly pure crystalline product. For R&D teams, this means that the final material requires minimal post-synthesis purification, reducing the overall process mass intensity (PMI). The ability to tune the pore size and chemical environment by simply adjusting the ligand structure or metal salt ratio offers a versatile platform for adapting this technology to various chiral separation challenges beyond just mandelates, potentially extending to other pharmaceutical intermediates requiring stringent optical purity.
How to Synthesize Chiral MOF Material Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and temperature control to ensure the formation of the desired crystalline phase. The process leverages common laboratory and industrial solvents, making the transition from bench scale to pilot plant relatively seamless. Detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and reaction conditions necessary to replicate the high ee values reported in the patent data. Adhering to these parameters is essential for maintaining the structural integrity of the MOF and ensuring consistent batch-to-batch performance in separation applications.
- Synthesize the monochiral ligand (S)-H4L via Suzuki coupling followed by hydrolysis.
- Mix the ligand with dysprosium trichloride and sodium chloride in DMF/water solvent.
- Heat the mixture at 100°C for 3 days to form the crystalline MOF structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this monochiral MOF technology translates into tangible strategic advantages regarding cost structure and operational reliability. The primary driver for cost reduction lies in the elimination of expensive, single-use chromatography media and the associated solvents required for regenerating traditional columns. By utilizing a solid-state adsorbent that can be reused multiple times without significant loss in efficiency, manufacturers can drastically simplify their downstream processing workflows. This reduction in consumable dependency not only lowers direct material costs but also minimizes the logistical burden of sourcing specialized separation columns, thereby enhancing overall supply chain resilience against market fluctuations.
- Cost Reduction in Manufacturing: The synthesis of this MOF relies on readily available raw materials such as dysprosium salts, sodium chloride, and common organic solvents like DMF and ethanol, which are commoditized and easy to source globally. Unlike proprietary enzymatic catalysts or complex chiral columns that command premium pricing, the precursors for this material are standard fine chemicals, leading to substantial cost savings in raw material procurement. Furthermore, the solvent-thermal method operates at atmospheric pressure and moderate temperatures, reducing the energy consumption associated with high-pressure reactors or cryogenic separation processes. The qualitative elimination of transition metal catalysts in the separation step itself means there is no need for expensive heavy metal scavenging procedures, further streamlining the cost profile of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of the metal-organic framework structure ensures a longer operational lifecycle compared to fragile organic polymers, reducing the frequency of material replacement and the risk of production stoppages. Since the synthesis does not depend on rare biological enzymes or highly specialized equipment, the supply risk is mitigated, allowing for more predictable production planning. The ability to regenerate the material through simple solvent exchange and drying processes means that inventory levels of the separation media can be kept lower, freeing up working capital. This reliability is crucial for maintaining continuous manufacturing lines, especially when dealing with high-value chiral intermediates where downtime can result in significant financial losses.
- Scalability and Environmental Compliance: The solvothermal synthesis is inherently scalable, as it utilizes standard reactor vessels and heating mantles familiar to any chemical manufacturing facility, facilitating a smooth transition from grams to tons. The process generates minimal hazardous waste, as the solvents used are recoverable and the metal salts are sequestered within the stable framework, aligning with increasingly strict environmental regulations. The high selectivity of the material reduces the need for multiple recrystallization steps, which typically generate large volumes of mother liquor waste. By improving the atom economy of the separation process, this technology supports sustainability goals while simultaneously improving the throughput capacity of existing manufacturing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral MOF technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this material into their current purification pipelines.
Q: What are the advantages of this MOF over cyclodextrin columns?
A: Unlike cyclodextrin derivatives which require complex synthesis and offer lower stability, this MOF features a robust inorganic-organic hybrid skeleton that withstands repeated use and offers superior thermal stability up to 100°C.
Q: Can this material be reused for multiple separation cycles?
A: Yes, the patent data indicates excellent reusability, maintaining enantiomeric excess (ee) values above 90% even after secondary adsorption cycles, significantly reducing operational waste.
Q: Is the synthesis process scalable for industrial production?
A: The solvothermal method utilizes common solvents like DMF and water at moderate temperatures (100°C), making it highly adaptable for large-scale reactor systems without requiring extreme pressure conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral MOF Material Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced separation technologies like the monochiral MOF described in patent CN103193831A for the global pharmaceutical supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards. We understand that adopting new materials requires confidence in consistency, and our manufacturing protocols are designed to deliver the high-performance chiral separation materials your R&D teams demand.
We invite you to collaborate with us to explore how this technology can optimize your specific manufacturing processes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and purity requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can drive efficiency and value for your organization.
