Scalable Solid-Phase Synthesis of 1,2,4-Oxadiazolines for Commercial API Manufacturing
Scalable Solid-Phase Synthesis of 1,2,4-Oxadiazolines for Commercial API Manufacturing
In the rapidly evolving landscape of pharmaceutical and agrochemical development, the efficient construction of heterocyclic scaffolds remains a critical bottleneck for R&D teams striving to bring new active ingredients to market. Patent CN1202094C introduces a transformative solid-phase synthesis methodology for generating 3-aryl-substituted 1,2,4-oxadiazolines, a privileged structure known for its diverse biological activities ranging from antimicrobial and anti-inflammatory properties to antitumor effects. This technology addresses the longstanding challenges of traditional solution-phase synthesis by leveraging the inherent advantages of polymer-supported chemistry, offering a pathway to high-purity intermediates with significantly streamlined processing. For procurement and supply chain leaders, this represents a viable strategy for cost reduction in pharmaceutical intermediate manufacturing, as it replaces labor-intensive purification protocols with simple filtration and washing steps. The following analysis details the mechanistic robustness and commercial viability of this approach for establishing a reliable supply of complex heterocyclic building blocks.
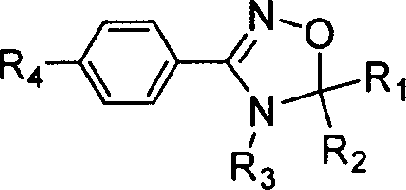
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-oxadiazoline derivatives has relied heavily on solution-phase techniques such as 1,3-dipolar cycloaddition or reactions involving nitro compounds and imines. While these methods are chemically valid, they suffer from significant operational inefficiencies when applied to the generation of diverse compound libraries or large-scale production. Conventional routes often require expensive reagents, harsh reaction conditions, and extensive downstream processing to isolate the desired product from complex reaction mixtures. The purification of intermediates typically involves multiple extraction, drying, and chromatographic separation steps, which not only consume vast quantities of organic solvents but also lead to substantial material loss, resulting in unfavorable overall yields. Furthermore, the time-consuming nature of isolating and characterizing each intermediate makes it nearly impossible to rapidly synthesize large arrays of structurally diverse precursors, thereby slowing down the drug discovery pipeline and increasing the cost per gram of the final active pharmaceutical ingredient.
The Novel Approach
The solid-phase synthesis method disclosed in the patent data offers a paradigm shift by anchoring the synthetic sequence to an insoluble polymer support, such as Merrifield or Wang resin. This approach allows for the use of excess reagents to drive reactions to completion without complicating the purification process, as impurities and excess reagents are simply washed away. The process operates under remarkably mild conditions, with key cyclization steps occurring at room temperature (20-30°C), which minimizes thermal degradation and energy consumption. By utilizing in-situ generated nitrile oxides and imines, the method avoids the stability issues associated with isolating reactive intermediates. The result is a highly efficient workflow where the crude product purity can exceed 80% after mere filtration, and final isolated yields in specific embodiments reach as high as 96% with purities up to 98%. This efficiency makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, ensuring a consistent and high-quality supply chain.
Mechanistic Insights into Solid-Phase Heterocycle Construction
The core of this technology lies in the sequential functionalization of the resin-bound substrate, beginning with the loading of a substituted aromatic aldehyde onto the polymer support. Under alkaline conditions in an organic solvent at elevated temperatures (typically 80°C), the aldehyde is covalently attached to the resin, forming a stable anchor for subsequent transformations. The next critical step involves the condensation of this resin-bound aldehyde with hydroxylamine hydrochloride to form a resin-loaded oxime. This oxime serves as the precursor for the nitrile oxide species, which is generated in situ upon treatment with an N-halosuccinimide (such as N-chlorosuccinimide or N-bromosuccinimide). The use of N-halosuccinimides is particularly advantageous as they are stable, easy-to-handle solids that facilitate the smooth conversion of oximes to the reactive nitrile oxide intermediates directly on the solid support.
Following the generation of the nitrile oxide, the system undergoes a [3+2] cycloaddition with an externally added imine. The imine, prepared separately from an aldehyde and a primary amine in the presence of activated molecular sieves, acts as the dipolarophile. The reaction proceeds in the presence of an organic base, such as triethylamine or diisopropylethylamine, at room temperature to form the 1,2,4-oxadiazoline ring. The final step involves the cleavage of the product from the resin using a low-boiling primary amine (e.g., ethylamine) or a sodium alkoxide solution. This cleavage step is designed to be mild yet effective, releasing the final 3-aryl-1,2,4-oxadiazoline derivative into the solution while leaving the polymer support intact for potential recycling or disposal. The entire sequence is characterized by its modularity, allowing for the easy variation of R1, R2, R3, and R4 groups to generate a wide array of analogues for biological screening.
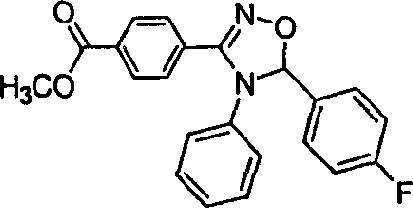
How to Synthesize 3-Aryl-1,2,4-Oxadiazolines Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable heterocycles with minimal operational complexity. The process is divided into five distinct stages: resin loading, oxime formation, imine preparation, cyclization, and cleavage. Each step is optimized to maximize yield and purity while minimizing the need for intermediate isolation. The use of common solvents like dichloromethane, DMF, and THF ensures compatibility with standard laboratory and pilot-plant equipment. Detailed below is the strategic overview of the synthesis, highlighting the critical parameters for success.
- Load substituted aromatic aldehyde onto resin (Merrifield/Wang) in organic solvent with alkali at 50-100°C.
- React resin-bound aldehyde with hydroxylamine hydrochloride and organic base at room temperature to form resin-bound oxime.
- Prepare imine solution separately by reacting aldehyde with primary amine under molecular sieve activation.
- Halogenate the resin-bound oxime with N-halosuccinimide, then react with the imine solution and base at room temperature for annulation.
- Cleave the final product from the resin using low-boiling primary amine or sodium alkoxide in organic solvent at 25-60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid-phase methodology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the workup procedure. Unlike solution-phase synthesis which often requires multiple liquid-liquid extractions and column chromatography, this method relies on simple filtration and washing. This reduction in unit operations translates directly into lower labor costs and reduced solvent waste, aligning with modern green chemistry principles and environmental compliance standards. Furthermore, the ability to use excess reagents to drive reactions to completion without affecting product isolation ensures consistently high conversion rates, minimizing the risk of batch failures and supply disruptions.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive purification steps and the use of cost-effective raw materials. The process utilizes inexpensive functionalized polystyrene resins and common organic bases, avoiding the need for precious metal catalysts or specialized reagents often found in alternative heterocycle synthesis routes. By achieving high crude purity (>80%) directly from the reactor, the downstream processing burden is significantly lightened, leading to substantial cost savings in terms of both material consumption and processing time. The high yields reported, such as 96% in specific embodiments, further enhance the overall process economics by maximizing the output from every kilogram of starting material.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness and reproducibility of the solid-phase protocol. The reactions are performed under mild conditions, often at room temperature, which reduces the dependency on specialized heating or cooling infrastructure and lowers the risk of thermal runaway incidents. The modular nature of the synthesis allows for the rapid substitution of different aldehydes and amines, enabling manufacturers to quickly adapt to changing demand for specific analogues without retooling the entire production line. This flexibility ensures that a reliable pharmaceutical intermediate supplier can maintain steady delivery schedules even when facing fluctuations in the demand for specific API precursors.
- Scalability and Environmental Compliance: Scaling this process from gram-scale library synthesis to multi-kilogram commercial production is facilitated by the simplicity of the solid-handling operations. The use of filtration for separation is inherently easier to scale than complex distillation or crystallization processes required for some solution-phase intermediates. Additionally, the reduction in solvent usage and the avoidance of heavy metal contaminants contribute to a cleaner environmental footprint. This aligns with increasingly stringent global regulations on industrial emissions and waste disposal, making the technology a sustainable choice for long-term cost reduction in API manufacturing and ensuring compliance with international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-phase synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear understanding of the method's capabilities and limitations for potential partners and stakeholders.
Q: What are the purity levels achievable with this solid-phase method?
A: The patent data indicates that crude product purity can reach above 80% simply through filtration and washing, with specific optimized embodiments achieving final purities between 92% and 98% after cleavage and concentration, significantly reducing the need for complex chromatographic purification.
Q: How does this solid-phase route compare to traditional solution-phase synthesis in terms of cost?
A: This method utilizes inexpensive functionalized polystyrene resins and common solvents. By eliminating multiple extraction and purification steps required in solution-phase chemistry, it drastically simplifies the workflow and reduces solvent consumption and labor costs associated with intermediate isolation.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is highly amenable to automation using solid-phase synthesis instruments. The simple operational steps of filtration and washing, combined with mild reaction conditions (often room temperature), facilitate easy scale-up from gram-scale library generation to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Oxadiazoline Supplier
The technological advancements detailed in patent CN1202094C underscore the immense potential of solid-phase synthesis in modernizing the production of high-value heterocyclic intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency at every stage of production.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective synthesis route for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how our optimized processes can reduce your overall cost of goods sold. We encourage you to contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis needs. Let us help you secure a stable, high-quality supply of 1,2,4-oxadiazoline derivatives to accelerate your drug development timelines.
