Advanced Chiral Silicon Ligands for Scalable Asymmetric Catalysis in Fine Chemical Manufacturing
Introduction to Novel Chiral Ligand Technology
The landscape of asymmetric catalysis is continuously evolving with the introduction of robust scaffolds that enhance both selectivity and stability in industrial processes. Patent CN114315906B introduces a significant breakthrough with the disclosure of spirobis-dihydrobenzosilole phosphite compounds, represented generally by Formula (I). These compounds serve as highly efficient chiral ligands capable of catalyzing critical transformations such as the hydrosilylation of allenes and the hydroformylation of olefins. The unique spiro-silicon backbone provides a rigid structural framework that effectively transfers chirality to the reaction center, addressing long-standing challenges in producing optically pure pharmaceutical intermediates. This technology represents a pivotal advancement for manufacturers seeking reliable pharmaceutical intermediate supplier solutions that prioritize high enantiomeric excess and process reliability.
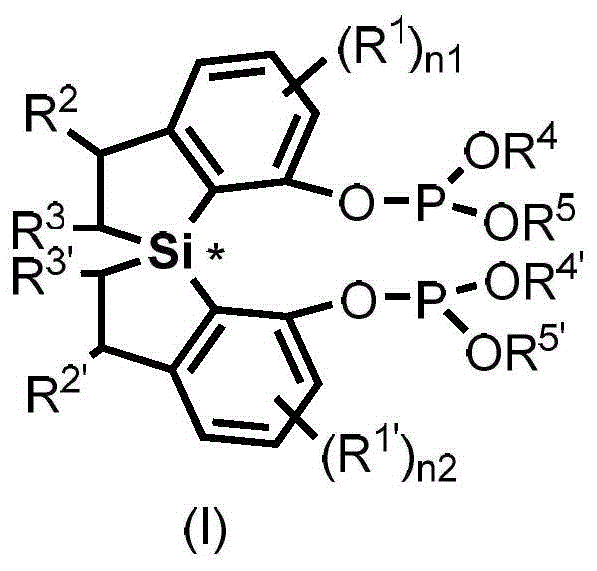
The structural versatility of Formula (I) allows for extensive modification of substituents R1 through R5, enabling fine-tuning of steric and electronic properties to match specific substrate requirements. This adaptability is crucial for R&D teams aiming to optimize reaction outcomes for complex drug molecules. By leveraging the inherent stability of the silicon-carbon bonds within the spirocyclic core, these ligands demonstrate superior resistance to hydrolysis compared to traditional carbon-based phosphites, thereby extending catalyst lifetime and reducing the frequency of catalyst replenishment in continuous flow systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphite ligands often suffer from inherent instability under reaction conditions, particularly in the presence of moisture or elevated temperatures, which can lead to rapid decomposition and loss of catalytic activity. Furthermore, many conventional chiral backbones lack the necessary rigidity to enforce strict stereocontrol, resulting in moderate enantioselectivity that necessitates costly downstream purification steps such as recrystallization or chiral chromatography. The reliance on flexible biaryl axes in older ligand generations can also lead to conformational ambiguity, where multiple active species form in situ, complicating process validation and scale-up efforts for commercial manufacturing. These limitations frequently translate into inconsistent batch-to-batch quality and increased production costs due to lower overall yields and higher waste generation.
The Novel Approach
The novel spirobis-dihydrobenzosilole architecture overcomes these deficiencies by locking the chiral environment into a fixed three-dimensional geometry that minimizes conformational freedom. This rigidity ensures that the metal center interacts with the substrate in a highly predictable manner, consistently delivering high enantiomeric excess values often exceeding 94% in hydrosilylation reactions. The incorporation of the silicon atom not only enhances thermal stability but also modifies the electronic density at the phosphorus centers, boosting catalytic turnover frequencies. This approach facilitates cost reduction in pharmaceutical intermediate manufacturing by minimizing the need for expensive chiral resolving agents and reducing the environmental footprint associated with solvent-intensive purification protocols.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Hydrosilylation
The efficacy of these ligands is best exemplified in the nickel-catalyzed hydrosilylation of allenes, where the chiral information is transferred from the ligand to the forming carbon-silicon bond with remarkable precision. The mechanism involves the coordination of the nickel center to the phosphite oxygen atoms, creating a chiral pocket that directs the approach of the allene substrate and the silane reagent. The rigid spiro-framework prevents unfavorable rotational isomers from participating in the catalytic cycle, effectively filtering out pathways that would lead to racemic byproducts. This level of control is essential for producing high-purity OLED material precursors or active pharmaceutical ingredients where trace impurities can compromise product safety and efficacy.
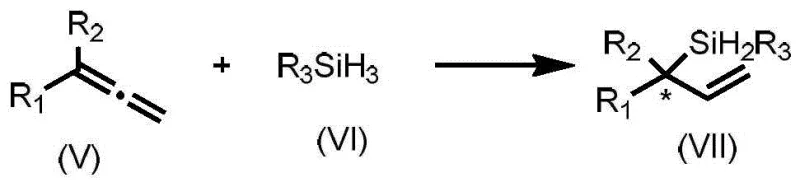
Impurity control is further enhanced by the high chemoselectivity of the catalyst system, which tolerates various functional groups on the allene substrate without promoting side reactions such as polymerization or isomerization. The patent data indicates that substituents on the aromatic rings of the ligand can be adjusted to modulate the steric bulk around the active site, allowing chemists to suppress the formation of specific regioisomers. This tunability is a powerful tool for process chemists who need to adapt a single ligand family to a diverse library of substrates, streamlining the development timeline for new synthetic routes and ensuring robust supply chain continuity for key building blocks.
How to Synthesize Spirobis-dihydrobenzosilole Phosphite Efficiently
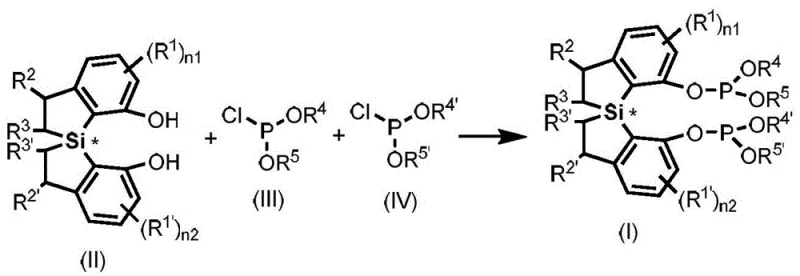
The preparation of these advanced ligands follows a straightforward substitution protocol that is amenable to large-scale production without requiring exotic reagents or extreme conditions. The process begins with the activation of a spirodihydrobenzosilole chiral diphenol precursor, which is reacted with phosphorus chlorides in the presence of a tertiary amine base to scavenge generated acid. The reaction is typically conducted in ether solvents like tetrahydrofuran at controlled temperatures to manage exotherms and ensure complete conversion. This synthetic route is designed for operational simplicity, utilizing commercially available starting materials that support a stable and reliable supply chain for high-volume manufacturing needs.
- Prepare the reaction vessel under nitrogen and dissolve spirodihydrobenzosilole chiral diphenol in tetrahydrofuran with triethylamine base.
- Add the phosphorus chloride solution dropwise at 0°C to control exothermic reaction and ensure proper substitution.
- Stir overnight at room temperature, filter through silica gel, and purify via column chromatography to isolate the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this ligand technology offers substantial strategic benefits by simplifying the sourcing of critical chiral catalysts. The synthesis relies on standard organic building blocks and common solvents, which mitigates the risk of supply disruptions often associated with specialized or proprietary reagents. This accessibility ensures that manufacturing partners can maintain consistent production schedules without being bottlenecked by raw material shortages, thereby enhancing overall supply chain reliability for downstream clients. Furthermore, the robustness of the ligand reduces the frequency of catalyst replacement, leading to significant operational efficiencies and lower total cost of ownership for catalytic processes.
- Cost Reduction in Manufacturing: The high enantioselectivity achieved by these ligands drastically reduces the burden on downstream purification units, eliminating the need for expensive chiral separation columns or multiple recrystallization steps. By maximizing the yield of the desired enantiomer directly from the reactor, manufacturers can significantly lower solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable production model. The elimination of transition metal scavenging steps, often required with less stable catalysts, further streamlines the workflow and reduces material costs.
- Enhanced Supply Chain Reliability: The use of stable silicon-based scaffolds ensures that the ligands have an extended shelf life, allowing for bulk purchasing and inventory buffering without degradation concerns. This stability is critical for maintaining uninterrupted production lines, especially for long-term contracts where consistent quality is paramount. The ability to synthesize these ligands using widely available chemical feedstocks means that procurement teams are not dependent on single-source suppliers for niche intermediates, fostering a more resilient and competitive supply network.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as operating at near-ambient temperatures and using common organic solvents, facilitate easy scale-up from laboratory to commercial tonnage without complex engineering modifications. This scalability supports the commercial scale-up of complex pharmaceutical intermediates while adhering to strict environmental regulations regarding energy consumption and volatile organic compound emissions. The high atom economy of the hydrosilylation reaction further aligns with green chemistry principles, reducing the overall environmental impact of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and handling of spirobis-dihydrobenzosilole phosphite ligands in industrial settings. These insights are derived directly from the experimental data and embodiments provided in the patent documentation to assist technical teams in evaluating feasibility. Understanding these parameters is essential for integrating this technology into existing manufacturing workflows effectively.
Q: What are the primary advantages of spirobis-dihydrobenzosilole phosphite ligands?
A: These ligands offer high catalytic activity and exceptional enantioselectivity in nickel-catalyzed allene hydrosilylation reactions, often achieving yields above 90% with ee values exceeding 94%.
Q: Can these ligands be used for hydroformylation reactions?
A: Yes, the patent demonstrates their efficacy in cobalt-catalyzed olefin hydroformylation, providing a versatile platform for synthesizing aldehyde intermediates under mild pressure conditions.
Q: What are the typical reaction conditions for synthesizing these ligands?
A: Synthesis typically occurs in ether solvents like THF at temperatures between 0°C and 25°C using organic bases such as triethylamine, ensuring operational safety and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirobis-dihydrobenzosilole Phosphite Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into practical industrial solutions, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to adapt the synthesis of spirobis-dihydrobenzosilole phosphite compounds to meet stringent purity specifications required by global regulatory bodies. With state-of-the-art rigorous QC labs, we ensure that every batch of chiral ligand delivered meets the highest standards of optical purity and chemical integrity, providing our partners with the confidence needed to accelerate their drug development pipelines.
We invite potential collaborators to engage with our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and target molecules. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible economic benefits of switching to this next-generation ligand technology. Let us help you optimize your catalytic processes and secure a competitive advantage in the global market for high-value fine chemicals.
